Uner Tan syndrome
Uner Tan syndrome also known as Unertan Syndrome (UTS) or cerebellar ataxia, mental retardation, and disequilibrium syndrome (CAMRQ), is better known as human quadrupedalism that is characterized by habitual diagonal-sequence quadrupedal gait on hands and feet, cerebellar hypoplasia, loss of balance and coordination, impaired intelligence, and rudimentary speech, with no conscious experience 1. Among cerebellar ataxias, Uner Tan syndrome is a unique syndrome with substantial differences from other balance disorders such as disequilibrium syndrome, Cayman ataxia, and Joubert syndrome 2. In this context, Guertin 3 emphasized that Uner Tan syndrome is a “recently identified and uniquely different neurological disorder.” Genetic studies showed Uner Tan syndrome to be a unique, genetically heterogeneous syndrome without infantile hypotonia 4.
In 2005, Uner Tan described the first Uner Tan syndrome family who lived in a small village near Iskenderun in southern Turkey (Family A) and then a second family with three affected individuals was found in Adana (Family B), also in southern Turkey 5. Next, a large family (Family C) was identified in a small village near Gaziantep in southern Turkey 6 and a fourth family (Family D) was found in a village near Canakkale in northern Turkey 7. All patients except those in family C were from low socioeconomic families 1.
Since the first families were found, three more affected children using the diagonal-sequence quadrupedal locomotion typical of Uner Tan syndrome have been discovered 1. There were no intrafamilial marriages in the first two cases, but there was in the third. Two children (males, aged 4 and 12 years) were identified in families (E and F) in Adana and Istanbul 8. Both boys started to walk normally after crawling in infancy; their cognitive functions were normal, they had conscious experience and could speak and understand, but they were unable to perform a tandem walk. They began to adopt quadrupedal locomotion at around three years old, but only for fast locomotion. This is much later than the adoption of quadrupedal locomotion in the earlier cases, which occurred before they were one year old.
More recently, a 12-year-old boy exhibiting symptoms of Uner Tan syndrome has been discovered 9. The boy seemed normal at birth, crawled normally and then walked upright, but with truncal ataxia. At the age of 10 years he began to adopt a facultative quadrupedal gait for fast locomotion. This is the first known case of a patient exhibiting late childhood quadrupedalism. He has never spoken.
Uner Tan syndrome cause
The exact cause of Uner Tan syndrome is currently unknown. Genetic causes of Uner Tan syndrome were reported to be heterogeneous 10, however, a mutation in very-low-density lipoprotein receptor (VLDLR) gene was said to be the cause of both the cerebellar hypoplasia and quadrupedal locomotion in at least some of those individuals with Uner Tan syndrome 11. To date, mutations in very-low-density lipoprotein receptor (VLDLR), WDR81, CA8, ATP8A2 and TUBB2B have been described as causative in Uner Tan syndrome 12. Because the presence of quadrupedalism is variable with respect to the identified genetic mutations, other researchers have argued that the use of quadrupedalism is not the direct effect of genetic mutation, but an adaptation to instability of the trunk caused by the genetically determined cerebellar dysfunction, combined with environmental conditions such as insufficient medical care 13; but some argue that human quadrupedalism is not an epiphenomenon caused by neurodevelopmental malformation and ataxia 14, 15. Although a recent paper acknowledged that “the genetic associations hitherto reported for the Uner Tan syndrome seem to have no or only minor explanatory power, if any, for the origins of human quadrupedalism” 16, and acknowledged the role of other factors such as socioeconomic status, researchers continue to make persistent (but unsupported) claims regarding the “primitive” and “primate-like” nature of the quadrupedalism 17.
Uner Tan syndrome genetics
Uner Tan syndrome is associated with intrafamilial marriages, which suggests an autosomal recessive transmission 1. Uner Tan syndrome may be regarded as a distinct entity among non – progressive autosomal recessive cerebellar ataxias, but there are overlapping symptoms with these forms of ataxia. For instance, Cayman cerebellar ataxia syndrome is caused by mutations in the ATCAY gene, and is associated with truncal ataxia, hypotonia, psychomotor delay, dysarthria, and cerebellar hypoplasia similar to Disequilibrium syndrome 18. These symptoms are present in 75-100% of patients with cerebellar ataxia, suggesting a broad overlap among congenital non-progressive ataxias, with a growing number of different genes responsible for these disorders 19.
Homozygosity was observed in affected individuals in families C and D, and was mapped to a region on chromosome 9p24 20. This region includes the very low density lipoprotein receptor gene (VLDLR), which plays a role in neuroblast migration within the brain. Ozcelik et al. 20 first identified mutations in the VLDLR gene in affected members of families C and D. These authors suggested mutations in the VLDLR gene may lead to VLDLR deficiency at a key stage of the developing brain, which may preclude normal neural migration during the formation of cerebrocerebellar structures critical for gait, resulting in quadrupedal locomotion instead of normally occurring bipedal locomotion. This hypothesis has not yet been tested, however. Despite the involvement of the same gene (VLDLR) in Disequilibrium syndrome and Uner Tan syndrome, their phenotypes are different, especially with regard to the locomotor system. This difference may depend upon the grades of VLDLR mutations in the two syndromes; that is, complete deletion of the VLDLR gene in Disequilibrium syndrome 21 but nonsense mutations in Uner Tan syndrome families C and D 20.
The effects of such mutations can be complex, and different mutations in a single gene may lead to different expressions of the same phenotype (allelic heterogeneity), so dissimilar mutations in the VLDLR gene of the various families may lead to the same phenotype, resulting in the symptoms of Uner Tan syndrome 22. There are many examples of mutations in a single gene leading to a wide range of phenotypes closely related to the type of mutation 23. It has also been stated that “similar genetic lesions can have entirely different phenotypes” 24. Given this, it is possible that different mutations in the VLDLR gene might be responsible for several types of autosomal recessive cerebellar ataxias. That is, different VLDLR mutations may be responsible for different types of locomotion in affected individuals, such as habitual quadrupedal locomotion in Uner Tan syndrome, ataxic bipedal locomotion in some individuals with cerebello-vermial hypoplasia, or no locomotion at all in Disequilibrium syndrome.
Mutations in the VLDLR gene may result in a disruption of bipedal locomotion and the re-emergence of diagonal-sequence quadrupedal locomotion since it provides the best adaptive solution of the motor system to compensate for the inability to maintain the upright posture during bipedal locomotion. But mutations in a single gene like VLDLR 20 cannot be solely responsible for the emergence of human quadrupedalism, since the syndrome is genetically heterogeneous, like many other syndromes.
Two key evolutionary features of humans are increased brain volume and the ability to walk upright, but mutations in a single gene can prevent the full development of both 25. A single gene may be implicated in quadrupedalism in members of families C and D, but other genes may also be implicated in habitual quadrupedalism, and the development of quadrupedal locomotion may be affected by multiple sources. For example, in the affected individuals of family A, chromosome 17p13 was involved, while 17p13 and 9p24 had no effect in family B, which suggests the syndrome is genetically heterogeneous 20.
Ewing and Green 26 estimated the number of human genes as 35,000, which cannot be sufficient for the observed complexity of the human nervous system to be hard coded in the genome 27. Moreover, genes alone cannot be sufficient to induce different kinds of information processing within the brain, and the functions of the neural circuits are not predetermined by genes 28.
Environmental factors
Many of the affected individuals lived in small Turkish villages near large cities they had never visited. Most were children of consanguineous families, who rarely had visitors. Family A was a poor family with 19 children, five of whom had Uner Tan syndrome. The affected individuals lived isolated at home, except one man who often walked far away from home, but frequently became lost. The mother had diabetes, but the father had no health problems. The parents did not attempt any treatment for the impaired children, saying: “God gave them to us.” The healthy sisters helped the impaired sisters.
Family B lived in a large city, Adana, but they were also poor. They had three children, but the parents also did not seek medical treatment to help the impaired children walk upright. Two male children were affected. One was bipedal ataxic and one adopted habitual quadrupedal locomotion. He begged for money near hospitals.
Family C were relatively well off, but they also lived in a small village and the extended family had intrafamilial marriages. One of the relatives in this family was a physician and attempted physical treatment of the affected children, but with no success. The mother attempted to force her daughter to walk upright by fastening stones on her legs for six years. As a result the daughter now walks upright, but she is ataxic. She is also mentally retarded, and has dysarthric speech, and still prefers quadrupedal locomotion at times. Family D was also a poor family living in a small village. Of the new cases, two lived in cities (Adana and Istanbul) and were both in families of high socioeconomic status, but the child with late-onset quadrupedalism belonged to a poor family.
These results suggest socioeconomic factors are not primary in the origins of walking on all four extremities, and there were no environmental factors that forced the affected individuals to walk on all fours 29, although some authors have suggested the environment has a strong determining effect on quadrupedality 18.
Uner Tan syndrome symptoms
The primary characteristic of Uner Tan syndrome is habitual quadrupedalism, which was also the gait of our early ancestors 30. There are two kinds of quadrupedal locomotion: lateral sequence and diagonal sequence. The lateral sequence quadrupedal locomotion is used by non-primates, and the diagonal sequence quadrupedal locomotion mainly by primates, although not exclusively 31. In diagonal sequence quadrupedal locomotion a foot touches the ground followed by the contralateral hand and vice versa, whereas in lateral-sequence quadrupedalism the foot touches the ground followed by the ipsilateral hand and vice versa 32. All of the individuals with Uner Tan syndrome exhibit diagonal-sequence gait 1 when using quadrupedal locomotion.
All individuals in the first four families (except one man in family B) could stand and remain upright as long as they did not try to move. Truncal balance was nearly normal during standing, which meant they did not fall down if standing upright. Their main difficulty was in initiating the necessary movement to take a step, and when they did so they immediately lost their balance. In the most recent case, the boy could easily stand and walk (albeit with truncal ataxia), but he could not remain upright with his feet together, his eyes being open or closed (Romberg’s negative) 9.
It was reported that gait initiation is a transient stage between upright posture and locomotion, including anticipatory anteroposterior and lateral body movements 33. This suggests that Uner Tan syndrome might be an asymmetric lateral balance disorder. In accord, Gramsbergen 34 emphasized the importance of postural control for the development of the walking patterns.
Despite their difficulty with balance, all individuals except the man in family B could walk upright, but with ataxia. When using quadrupedal locomotion all the affected individuals could walk and even run at great ease and with no imbalance. At least one person in each family preferred the impaired upright gait, and these individuals were better able to speak than those who preferred quadrupedal locomotion, although the most recently discovered 12-year-old boy does not speak 9. The bipedal ataxic patients were also easier to approach and to talk to than the habitual quadrupeds. This finding is consistent with Uner Tan’s psychomotor theory, which highlights the importance of the motor system in the emergence of the human mind, as an expressive characteristic of language 35.
Impaired intelligence
Intelligence was impaired in all of the affected individuals in the first four families, most of whom could not answer a single question from the Mini-Mental State Examination Test 1, standardized for uneducated Turkish people. This test aims to measure patient responses in the following major areas: date and location, immediate recall of three words, counting backwards, recall of three items, naming a few items, repeating a sentence, paper folding, and drawing a watch. All but one of the affected individuals scored zero on this test. The bipedal ataxic patient scored two out of 30 points in the test 36. The children in family E were rather bright, but the child in family F was mentally impaired and could not speak, but could make a single sound.
The patients in family A were dysarthric, communicating among themselves in a language their neighbors could not understand, but they could understand simple commands. Those in family B understood simple sentences and answered with a limited vocabulary expressed by a few barely understandable words 37. Four patients in family C could understand simple commands, and expressed wishes with dysarthric speech using only few words 6, while two others in the same family used a few sounds instead of words. In family D two of the affected individuals exhibited dysarthric speech with a limited vocabulary, and two used only one or two sounds to communicate with others 7. The children in family E could understand and speak fairly well, while the child in family F could understand simple commands, but not speak at all. Despite the impaired cognition, the head circumference was within normal ranges in all of the patients (unpublished observartions).
Vestibular system
Different parts of the vestibular system were found to be affected in the Uner Tan syndrome patients. In families A and B respectively, Barany’s caloric nystagmus test indicated the central and peripheral vestibular systems were impaired 6, while in family C the vestibular system was normal 38. This means there was no unique vestibular defect associated with the syndrome. In accord with this, experiments in rats have shown impairments in vestibular input may cause dramatic retardations in the patterns involved in postural control 34, so genetically based impairments of the peripheral or central vestibular system may contribute to limited balance in some affected individuals, but not all of them.
The two new cases in Adana and Istanbul 8 showed no neuropsychological signs or symptoms, except their inability to tandem walk, and a bilateral positive Babinsky sign. They made no uncoordinated movements, the nose-finger test was normal with no intension tremor; whereas the lateral balance during standing was perfect. In the most recent case 9 the boy exhibited a mild strabismus with no nystagmus, and his eye movements were normal. His reflexes were hypoactive in the upper and lower extremities. The Babinsky sign could not be elicited bilaterally. There were no signs of spasticity. His muscle tone was normal in the trunk and extremities.
Uner Tan syndrome diagnosis
Uner Tan syndrome is diagnosed clinically based on clinical history with additional tests and investigations.
Magnetic Resonance Imaging and Positron Emission Tomography (PET) scans
Brain magnetic resonance imaging (MRI) scans were first reported for a single affected person in family A 39, but no scans were taken from controls in the same family. Detailed MRI and Positron Emission Tomography (PET) scans were then performed on families A and B 6. In the MRI scans of family A the right and left cerebral areas, right and left cerebellar areas, vermian and callosal areas were all measured, and from this inspection it appeared that the basal ganglia, thalamus, bulbus, and pons were normal but the right and left cerebral and cerebellar, vermian and corpus callosum areas were significantly smaller in patients than in normal control subjects. The cerebellar and vermian areas were unrelated to body size (height and weight), but there were significant correlations with body size in other areas. The difference in mean callosal area between patients and non-affected individuals decreased when height was considered as a covariate.
In family B the cerebral and cerebello-vermian areas in the affected individuals (one quadruped man, one quadruped woman, and one biped ataxic man) were not significantly different from those of the unaffected individuals. The affected individuals of family B had an impaired peripheral vestibular system, which suggested that their balance disorder had a different origin (peripheral vestibular defect) compared to that observed in family A (central vestibular defect).
In family A the PET scans indicated a significantly lower glucose metabolic rate in the right and left cerebellum, including the cerebellar vermis, of the affected individuals compared to unaffected individuals. Cerebral regions other than the cerebellum showed no significant differences between the affected and unaffected family members, except a mild decrease in the metabolic activity of the cerebral cortex. In family B, PET scans showed no decreased glucose metabolism in the cerebellum, vermis, frontal cortex, parietal cortex, temporal cortex, or occipital cortex of the affected individuals compared to controls 6.
Brain MRI scans were not possible for the two new cases 8 because of the hyperactivity of the children. The brain MRI of the latest patient 9 showed cerebello-vermial hypoplasia. He had additional cerebellar signs such as limb dysdiachokinesia, dysmetria, past-pointing, excessive rebound, kinetic tremor, trunk titubation and inability to perform tandem gait. These results suggest there may be subgroups of Uner Tan syndrome, or a range of characteristics, with regard to its genetics and neural origins 20.
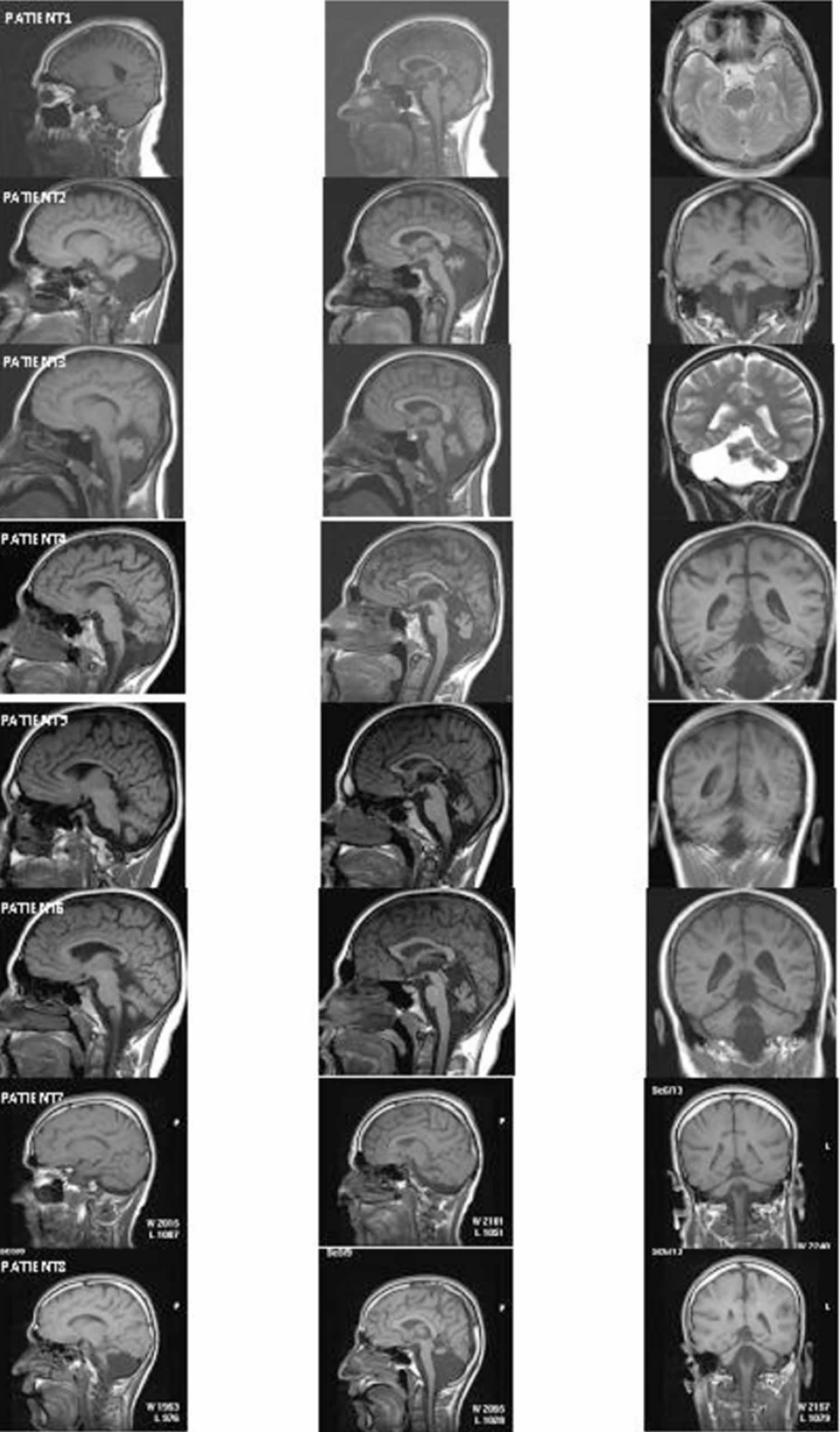
Figure 1 depicts the MRI sections of eight patients. All of the patients except Patient 1 showed cerebello-vermial hypoplasia; Patient 1 seemed to have a normal cerebellum with a mild vermial hypoplasia. Notice that in contrast to other patients, Patient 1 had only peripheral vestibular impairment, the central vestibular system being normal.
Figure 1. Uner Tan syndrome MRI scans
Footnote: MRI scans from eight patients. Left column: cerebellum in sagittal section; middle column: vermis in sagittal section; right column: cerebellum in coronal section.
[Source 1 ]Uner Tan syndrome treatment
From all the available literature, there are no cases of treatment being undertaken.
- Tan U. Uner tan syndrome: history, clinical evaluations, genetics, and the dynamics of human quadrupedalism. Open Neurol J. 2010;4:78-89. Published 2010 Jul 16. doi:10.2174/1874205X01004010078 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3024602[↩][↩][↩][↩][↩][↩][↩]
- Tan U., Tamam Y., Karaca S., Tan M., Tan U. (2012). Uner Tan syndrome: review and emergence of human quadrupedalism in self-organization, attractors and evolutionary perspectives, in Latest Findings in Intellectual and Developmental Disabilities Research, ed Tan U. (Croatia: InTech Publications; ), 1–44.[↩]
- Guertin P. A. (2013). Central pattern generator for locomotion: anatomical, physiological, and pathophysiological considerations. Front. Neurol. 3:183 10.3389/fneur.2012.00183[↩]
- Gulsuner S., Tekinay A. B., Doerschner K., Boyaci H., Bilguvar K., Unal H., et al. (2011). Homozygocity mapping and targeted genomic sequencing reveal the gene responsible for cerebellar hypoplasia and quadrupedal locomotion in consanguineous kindred. Genome Res. 21, 1995–2003 10.1101/gr.126110.111[↩]
- Tan U. Evidence for “Unertan Syndrome” as a human model for reverse evolution. Int J Neurosci. 2006;116:1539–47.[↩]
- Tan U, Pence S, Yilmaz M, et al. “Unertan Syndrome” in two Turkish families in relation to devolution and emergence of homo erectus: neurological examination, MRI, and PET scans. Int J Neurosci. 2008;118:313–36.[↩][↩][↩][↩][↩]
- Tan U. Unertan Syndrome: review and report of four new cases. Int J Neurosci. 2008;118:211–25.[↩][↩]
- Tan U, Tan M. A new variant of Unertan syndrome: running on all fours in two upright-walking children. Int J Neurosci. 2009;119:909–18.[↩][↩][↩]
- Tan M, Karacas S, Tan U. A new case of uner tan syndrome—with late childhood quadrupedalism. Mov Dis. 2010;25:652.[↩][↩][↩][↩][↩]
- Onat OE, Gulsuner S, Bilguvar K, Nazli Basak A, Topaloglu H, et al. (2013) Missense mutation in the ATPase, aminophospholipid transporter protein ATP8A2 is associated with cerebellar atrophy and quadrupedal locomotion. Eur J Hum Genet 21: 281–285 10.1038/ejhg.2012.170[↩]
- Ozcelik T, Akarsu N, Uz E, Caglayan S, Gulsuner S, et al. (2008) Mutations in the very low-density lipoprotein receptor VLDLR cause cerebellar hypoplasia and quadrupedal locomotion in humans. Proc Natl Acad Sci 105: 4232–4236 10.1073/pnas.0710010105[↩]
- Breuss MW, Nguyen T, Srivatsan A, et al. Uner Tan syndrome caused by a homozygous TUBB2B mutation affecting microtubule stability. Hum Mol Genet. 2017;26(2):258-269. doi:10.1093/hmg/ddw383 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6075555[↩]
- Ivanenko YP, Wright WG, St George RJ, Gurfinkel VS. Trunk orientation, stability, and quadrupedalism. Front Neurol. 2013;4:20. Published 2013 Mar 14. doi:10.3389/fneur.2013.00020 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3596858[↩]
- Karaca S, Tan M, Tan U (2012) Human quadrupedalism is not an epiphenomenon caused by neurodevelopmental malformation and ataxia. Front Neurol 3: 1–3 10.3389/fneur.2012.00154[↩]
- Ozcelik T, Akarsu N, Uz E, Caglayan S, Gulsuner S, et al. (2008) Reply to Herz et al. and Humphrey et al.: Genetic heterogeneity of cerebellar hypoplasia with quadrupedal locomotion. Proc Natl Acad Sci 105: E32–E33 10.1073/pnas.0804078105[↩]
- Karaca S, Tan M, Tan U (2013) Humans Walking on all Four Extremities with Mental Retardation and Dysarthric or no Speech: A Dynamical Systems Perspective. In: Salehi A, editor. Developmental Disabilities – Molecules Involved, Diagnosis, and Clinical Care. InTech. DOI: 10.5772/55685 https://www.intechopen.com/books/developmental-disabilities-molecules-involved-diagnosis-and-clinical-care/humans-walking-on-all-four-extremities-with-mental-retardation-and-dysarthric-or-no-speech-a-dynamic[↩]
- Shapiro LJ, Cole WG, Young JW, Raichlen DA, Robinson SR, Adolph KE. Human quadrupeds, primate quadrupedalism, and Uner Tan Syndrome. PLoS One. 2014;9(7):e101758. Published 2014 Jul 16. doi:10.1371/journal.pone.0101758 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4100729[↩]
- Palau F, Espinos C. Autosomal recessive cerebellar ataxias. Orphanet J Rare Dis. 2006;1:47.[↩][↩]
- Maschke M, Oehlert G, Xie TD, et al. Clinical feature profile of spinocerebellar ataxia type 1-8 predicts genetically defined sub-types. Mov Dis. 2005;20:1405–12.[↩]
- Ozcelik T, Akarsu N, Uz E, et al. Mutations in the very low density lipoprotein receptor VLDLR cause cerebellar hypoplasia and quadrupedal locomotion in humans. Proc Natl Acad Sci USA. 2008;105:4232–6.[↩][↩][↩][↩][↩][↩]
- Boycott KM, Bonnemann C, Herz J, et al. Mutations in VLDLR as a cause for autosomal recessive cerebellar ataxia with mental retardation (disequilibrium syndrome) J Child Neurol. 2009;24:1310–5.[↩]
- Ozcelik T, Akarsu N, Uz E, et al. Genetic heterogeneity of cerebellar hypoplasia with quadrupedal locomotion. Proc Natl Acad Sci USA. 2008;105:E32–E33.[↩]
- Kato M, Dobyns WB. Lissencephaly and the molecular basis of neuronal migration. Hum Mol Genet. 2003;12:R89–R96.[↩]
- Prasun P, Pradhan M, Agarwal S. One gene, many phenotypes. J Postgrad Med. 2007;53:257–61.[↩]
- Garber KB. Editor’s corner: This month in genetics; VLDLR receptor stands tall. Am J Hum Genet. 2008;82:807.[↩]
- Ewing B, Green P. Analysis of expressed sequence tags indicates 35,000 genes. Nat Genet. 2000;25:232–4.[↩]
- Bornholdt S, Rohl T. Self-organized critical neural networks. Phys Rev E Stat Nonlin Soft Matter Phys. 2003;67(6Pt 2):066118[↩]
- Skoyles JR. The paleoanthropological implications of neural plasticity. CogPrints. 2009[↩]
- Humphrey N, Mundlos S, Turkmen S. Genes and quadrupedal locomotion in humans. Proc Natl Acad Sci USA. 2008;105:E26.[↩]
- Kelly RE. Tripedal knuckle-walking: a proposal for the evolution of human locomotion and handedness. J Theor Biol. 2001;213:333–58.[↩]
- Parchman AJ, Reilly SM, Biknevicius AR. Whole-body mechanics and gaits in the gray short-tailed opossum monodelphis domestica: integrating patterns of locomotion in a semi-erect mammal. J Exp Biol. 2003;206:1379–88.[↩]
- Shapiro LJ, Raichien DA. Lateral sequence walking in infant papio cynocephalus: implications for the evolution of diagonal sequence walking in primates. Am J Phys Anthropol. 2005;126:205–13.[↩]
- Sasaki O, Asawa S, Katsuno S, Usami S, Taguchi K. Gait initiation in bilateral vestibular loss. Auris Nasus Larynx. 2001;28:295–99.[↩]
- Gramsbergen A. Postural control in man: the phylogenetic perspective. Neural Plast. 2005;12:77–88.[↩][↩]
- Tan U. Psychomotor theory: Mind-brain-body triad in health and disease. In: Sarbadhikari SH, editor. Depression and dementia: Progress in Brain Research, clinical applications. Hauppauge, New York: Nova Science Publishers, Inc; 2005. pp. 21–53.[↩]
- Tan U. A new syndrome with quadrupedal gait, primitive speech, and severe mental retardation as a live model for human evolution. Int J Neurosci. 2006;116:361–9.[↩]
- Tan U. Evidence for “Unertan Syndrome” and the evolution of the human mind. Int J Neurosci. 2006;116:763–74.[↩]
- Tan U, Karaca S, Tan M, Bagci NK, Ozkur A, Pence S. Unertan Syndrome: A case series demonstrating human devolution. Int J Neurosci. 2008;118:1–25.[↩]
- Turkmen S, Demirhan O, Hoffmann K, et al. Cerebellar hypoplasia and quadrupedal locomotion in humans as a recessive trait mapping to chromosome 17p. J. Med Genet. 2006;43:461–64.[↩]