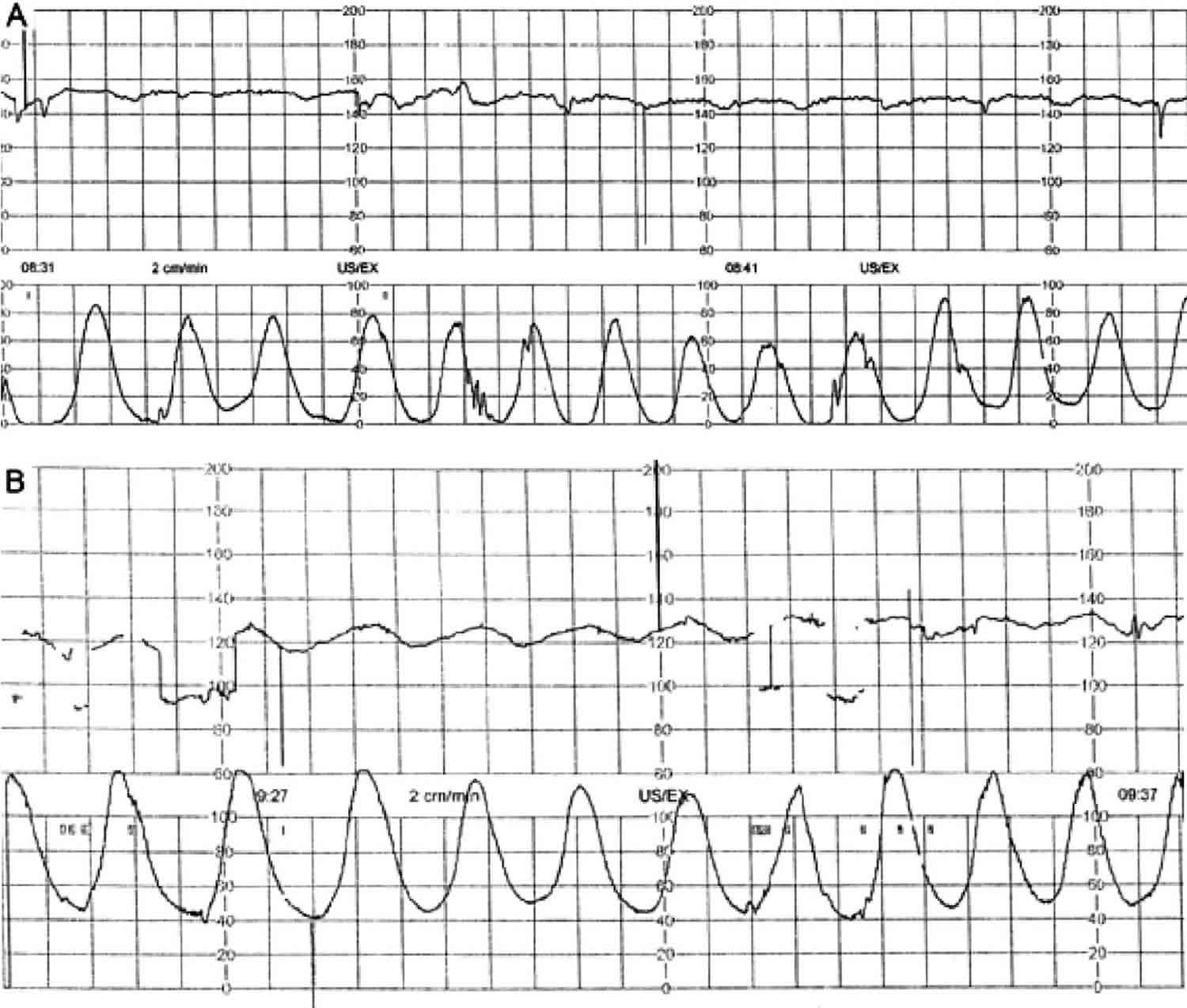
Uterine tachysystole
Uterine tachysystole is an excessive uterine activity defined as more than five uterine contractions per 10 minutes in at least two consecutive intervals 1, is common during labor 2, particularly with use of labor‐stimulating agents 3. Tachysystole may reduce fetal oxygenation by interrupting maternal blood flow to the placenta during contractions. Uterine tachysystole is associated with abnormal fetal heart rate patterns and may lead to adverse neonatal outcomes 4. Multiparity and maternal age of 30 years and older were associated with a decreased risk of tachysystole inmultivariate analysis 5.
Factors associated with an increased risk of uterine tachysystole 5
- Patient
- Younger maternal age
- Nulliparity
- Chronic hypertension
- Smoking/alcohol/drug history
- Pregnancy/delivery
- Preeclampsia
- Oligohydramnios
- Induction of labor (not elective)
- Use of oxytocin
- Use of misoprostol
- Longer time in labor
- Epidural
Reducing uterine contractions may improve placental blood flow, improving fetal oxygenation. Uterine tachysystole was documented in at least one‐fifth of women with spontaneous onset of labor who had not received any labor augmentation, and in 48.6% of women whose labor had been induced by prostaglandin 2. Similarly, non‐reassuring fetal heart rate patterns are commonly reported during active labor 6. Although uterine tachysystole is more commonly noted after the use of labor‐stimulating agents, it may also be a clinical sign of serious pregnancy complications, such as placental abruption or obstructed labor 3.
Uterine contraction compresses maternal spiral arteries and increases the intraplacental pressure, profoundly reducing the placental blood flow when the intraplacental pressure exceeds the pressure of maternal perfusion to the placenta 7. It was reported that a contraction at 30 mmHg or more was able to diminish or even interrupt maternal blood flow to the placenta and potentially reduces fetal oxygenation status 8. In one study, fetal oxygenation saturation, quantitatively measured by an intrauterine sensor, reached its nadir 92 seconds after the peak of a contraction, and took another 90 seconds to obtain complete recovery 7. Normal labor with sufficient physiological relaxation time allows the oxygenation level of an uncompromised baby to be restored between contractions.
The compensatory fetoplacental mechanism may no longer be effective in the presence of uterine tachysystole, which is usually associated with strong contractions of 80 mmHg or more 9 and significantly shortened relaxation time. Strong contractions contribute to further compression of maternal spiral arteries, while a shortened relaxation time presumably does not allow the blood supply to the placenta to return to baseline levels before the next contraction 10. Uterine tachysystole was found to be associated with a progressive decrease of fetal oxygen saturation (FSpO2) 11, and with a fall in fetal intracerebral oxygen saturation 10. In one study, five and six or more contractions per 10 minutes for 30 minutes resulted in 20% and 29% fetal oxygen desaturation, respectively 11. Importantly, tachysystole over a one‐hour period in active labor can be correlated with a significantly increased risk of fetal acidosis. A study of 1433 women with singleton pregnancies in the Netherlands concluded that increased uterine activity in first and second stage is correlated with a higher incidence of umbilical artery pH of 7.11 or less 9. This can in turn lead to an increase in adverse neonatal complications 12.
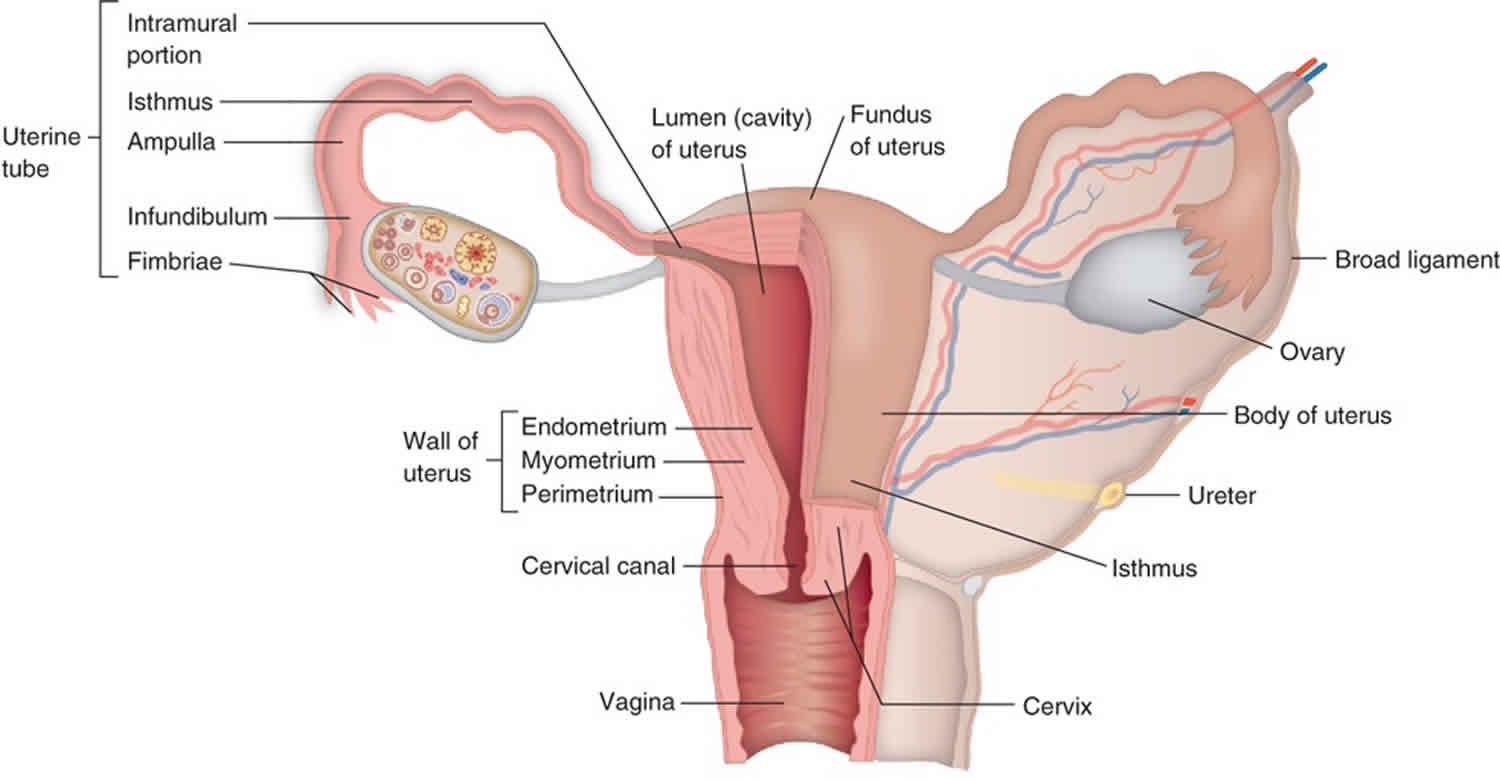
Figure 1. Uterus anatomy
Uterine tachysystole treatment
Intrauterine fetal resuscitation consists of interventions that aim to increase oxygen delivery to the placenta and enhance fetoplacental perfusion 1. National and international guidelines currently recommend intervention to correct non‐reassuring fetal heart rate patterns or suspected fetal distress during labour 1, fetal heart rate changes in the presence of excessive uterine activity 13, or excessive uterine activity in the absence of fetal heart rate changes 14. The available interventions include discontinuation of any labour‐stimulating agents, change of maternal position, administration of supplemental maternal oxygen, and if the abnormal fetal heart rate patterns persist, the use of tocolytic agents 1.
There is a wide range of tocolytic agents used acutely during labor for correcting uterine tachysystole, with or without the presence of suspected fetal distress. While studies have predominantly evaluated beta2 (ß2)‐adrenergic receptor agonists 15, some have also used magnesium sulphate 16, nitric oxide donors 17, calcium channel blockers 18, or oxytocin antagonists 15.
Acute tocolysis inhibits uterine smooth muscle activity 19, and has been used on the assumption that such relaxation improves placental perfusion and therefore may enhance fetal oxygenation 20. The intervention is employed to either improve fetal status and facilitate vaginal delivery, or as an interim method to improve fetal status whilst preparations for operative delivery are made 20. It is also possible that an improvement in the fetal heart rate trace may reduce caesarean section rates, given that a non‐reassuring fetal heart tracing is one of the leading indications for emergency caesarean section 21.
Overall, the use of a tocolytic agent appeared to improve some measures of fetal well‐being and reduce uterine tachysystole, though no studies demonstrated any improvement in neonatal condition, or in the primary outcomes 3. However, the limited data for most outcomes means specific conclusions cannot be drawn.
- Women who received a selective ß2‐adrenergic agonist whilst awaiting emergency delivery for fetal distress were more likely to experience an improvement in the fetal heart rate tracing than those who received no treatment. There was no difference detected between the groups in the rate of Apgar scores less than seven at five minutes, and the intervention may make little or no difference to this outcome.
- Atosiban for the treatment of fetal distress was associated with fewer cases of maternal palpitations or tachycardia than hexoprenaline, with no differences detected between the two across other outcomes. The small sample size means that rare events, such as fetal or neonatal mortality or severe morbidity, were unlikely to be detected.
- Women who received fenoterol for fetal distress were less likely to have infants that required admission to a neonatal intensive care unit (NICU) than women who were delivered by emergency delivery, however they were more likely to deliver by caesarean section. This was hypothesized to be due to reduced uterine contractility in the fenoterol group, reducing the likelihood of a vaginal delivery. We did not detect any differences across any other outcomes.
- Women who received hexoprenaline whilst awaiting emergency delivery for fetal distress, compared to those who did not, were less likely to have meconium‐stained liquor and were more likely to show an improvement in the fetal heart rate tracing. The Cochrane Review authors did not detect any differences between groups in other outcomes, including mortality and morbidity, Apgar scores, umbilical artery pH or NICU admission 3.
- Women who received terbutaline whilst awaiting emergency delivery, compared to those who did not, were more likely to show an improvement in the fetal heart rate tracing. The Cochrane Review authors did not detect any differences in any other outcomes including Apgar score, umbilical artery pH or maternal side effects 3.
- The Cochrane Review authors did not detect any differences in the umbilical artery pH, resolution of fetal distress or maternal blood loss between women treated with terbutaline for fetal distress and those treated with magnesium sulphate 3.
- Women treated with terbutaline for fetal distress were less likely to have uterine tachysystole following treatment than those treated with nitroglycerin, however the Cochrane Review authors did not detect any differences between groups in caesarean section rates, successful intrauterine fetal resuscitation or measures of neonatal well‐being postpartum.
- In women receiving oxytocin for labour augmentation who developed uterine hyperstimulation, hyperstimulation resolved faster in women who received terbutaline and continued the oxytocin compared to those who had the oxytocin infusion ceased. The Cochrane Review authors did not detect any differences between the groups in terms of caesarean section rates, Apgar scores or umbilical artery pH. The rate of recurrence of uterine hyperstimulation was also not different between groups 3.
- American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No.106: Intrapartum fetal heart rate monitoring: nomenclature, interpretation, and general management principles. Obstetrics & Gynecology 2009;114(1):192‐202.[↩][↩][↩][↩]
- Crane JM, Young DC, Butt KD, Bennett KA, Hutchens D. Excessive uterine activity accompanying induced labor. Obstetrics & Gynecology 2001;97(6):926‐31.[↩][↩]
- Leathersich SJ, Vogel JP, Tran TS, Hofmeyr GJ. Acute tocolysis for uterine tachysystole or suspected fetal distress. Cochrane Database Syst Rev. 2018;7(7):CD009770. Published 2018 Jul 4. doi:10.1002/14651858.CD009770.pub2 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6513259[↩][↩][↩][↩][↩][↩][↩]
- Pierce, Stephanie & Bakker, Ronan & Myers, Dean & Edwards, Rodney. (2018). Clinical Insights for Cervical Ripening and Labor Induction Using Prostaglandins. American Journal of Perinatology Reports. 08. e307-e314. 10.1055/s-0038-1675351.[↩]
- Heuser CC, Knight S, Esplin MS, et al. Tachysystole in term labor: incidence, risk factors, outcomes, and effect on fetal heart tracings. Am J Obstet Gynecol 2013;209(01):32.e1–32.e6[↩][↩]
- Bloom SL, Spong CY, Thom E, Varner MW, Rouse DJ, Weininger S, et al. Fetal pulse oximetry and cesarean delivery. New England Journal of Medicine 2006;355(21):2195‐202.[↩]
- McNamara H, Johnson N. The effect of uterine contractions on fetal oxygen saturation. BJOG: an international journal of obstetrics and gynaecology 1995;102(8):644‐7.[↩][↩]
- Brar HS, Platt LD, DeVore GR, Horenstein J, Medearis AL. Qualitative assessment of maternal uterine and fetal umbilical artery blood flow and resistance in laboring patients by Doppler velocimetry. American Journal of Obstetrics and Gynecology 1988;158(4):952‐6.[↩]
- Bakker PC, Kurver PH, Kuik DJ, Geijn HP. Elevated uterine activity increases the risk of fetal acidosis at birth. American Journal of Obstetrics and Gynecology 2007;196(4):313.[↩][↩]
- Peebles DM, Spencer JA, Edwards AD, Wyatt JS, Reynolds EO, Cope M, et al. Relation between frequency of uterine contractions and human fetal cerebral oxygen saturation studied during labour by near infrared spectroscopy. BJOG: an international journal of obstetrics and gynaecology 1994;101(1):44‐8.[↩][↩]
- Simpson KR, James DC. Effects of oxytocin‐induced uterine hyperstimulation during labor on fetal oxygen status and fetal heart rate patterns. American Journal of Obstetrics and Gynecology 2008;199(1):34.e1‐.e5.[↩][↩]
- Hankins GD, Speer M. Defining the pathogenesis and pathophysiology of neonatal encephalopathy and cerebral palsy. Obstetrics & Gynecology 2003;102(3):628‐36.[↩]
- National Collaborating Centre for Women’s and Children’s Health (UK). Induction of Labour. London: RCOG Press, 2008.[↩]
- World Health Organization. WHO Recommendations for Induction of Labour. Geneva: World Health Organization, 2011.[↩]
- Heus R, Mulder EJ, Derks JB, Kurver PH, Wolfswinkel L, Visser GH. A prospective randomized trial of acute tocolysis in term labour with atosiban or ritodrine. European Journal of Obstetrics & Gynecology and Reproductive Biology 2008;139(2):139‐45.[↩][↩]
- Vigil‐De Gracia P, Simiti E, Lora Y. Intrapartum fetal distress and magnesium sulfate. International Journal of Gynecology and Obstetrics 2000;68(1):3‐6.[↩]
- Buhimschi CS, Buhimschi IA, Malinow AM, Weiner CP. Effects of sublingual nitroglycerin on human uterine contractility during the active phase of labor. American Journal of Obstetrics and Gynecology 2002;187(1):235‐8.[↩]
- Chao A, El‐Sayed YY, Sapugay AM. Does nifedipine prevent the tachysystole associated with misoprostol induction?. Journal of Maternal‐Fetal & Neonatal Medicine 2002;11(4):266‐9.[↩]
- Hubinont C, Debieve F. Prevention of preterm labour: 2011 update on tocolysis. Journal of Pregnancy 2011;2011:941057[↩]
- Simpson K. Intrauterine resuscitation during labor: review of current methods and supportive evidence. Journal of Midwifery & Women’s Health 2007;52(3):229‐37[↩][↩]
- Barber E, Lundsberg L, Belanger K, Pettker C, Funai E, Illuzzi J. Contributing indications to the rising cesarean delivery rate. Obstetrics & Gynecology 2011;118(1):29‐38[↩]