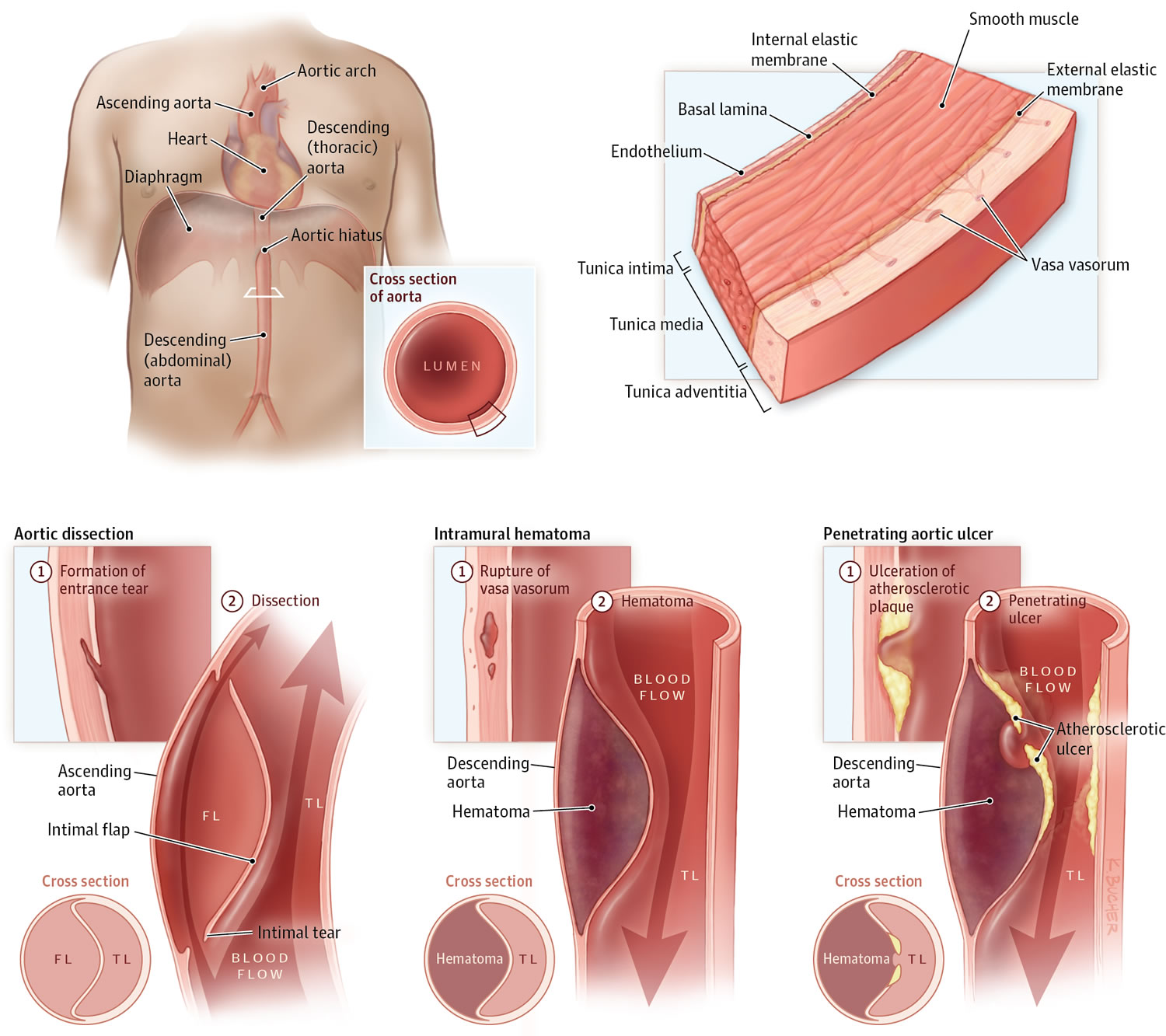
What is aortic dissection
Aortic dissection is a serious condition in which there is a tear in the layers within the aortic wall of the major artery carrying blood out of the heart (aorta) (see Figures 1, 2 and 3 below). As the tear extends along the wall of the aorta, blood can flow in between the layers of the blood vessel wall (dissection). This can lead to aortic rupture or decreased blood flow (ischemia) to organs.
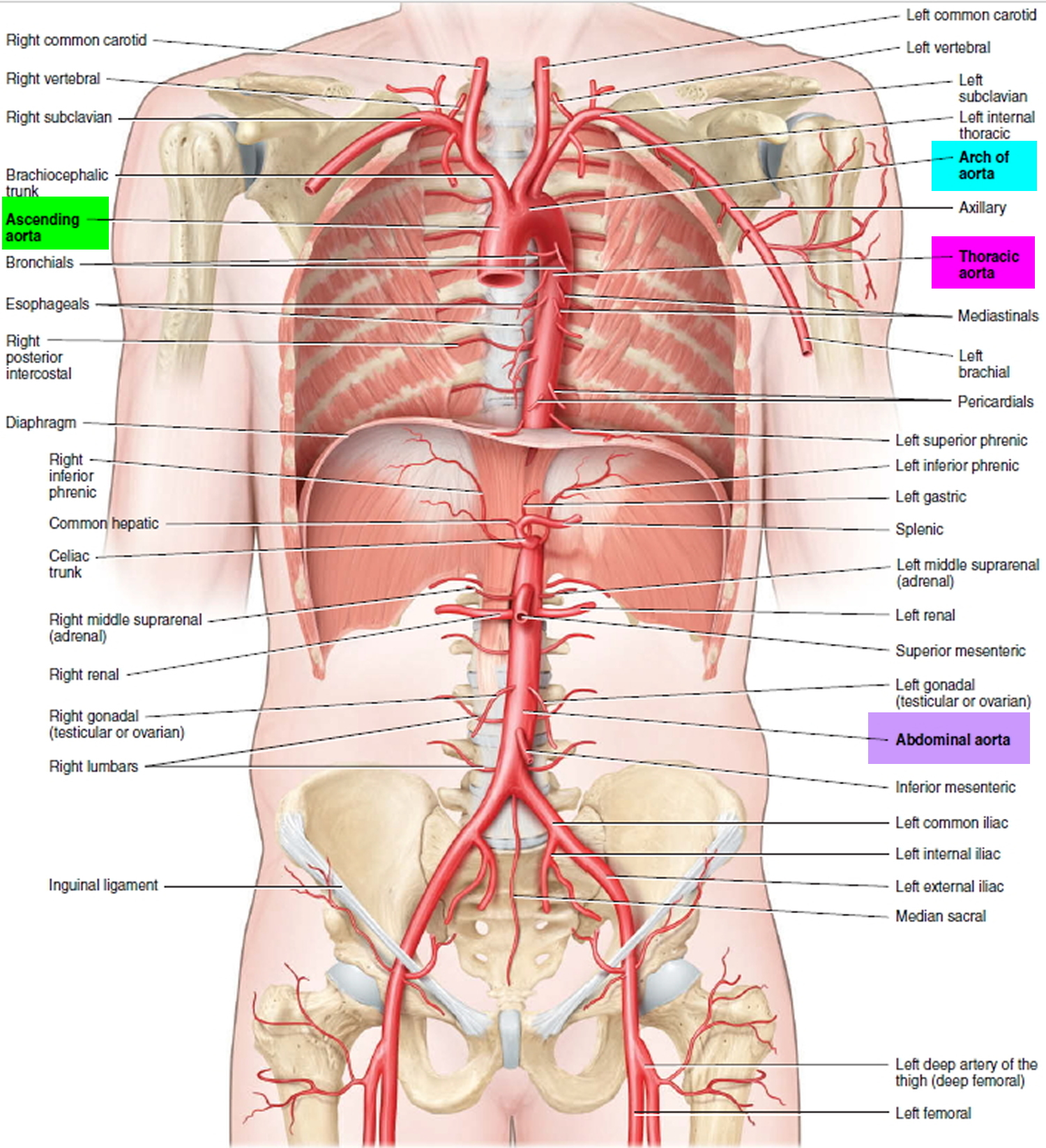
When aorta leaves the heart, the aorta first moves up through the chest towards the head (the ascending aorta). It then bends or arches, and finally moves down through the chest and abdomen (the descending aorta) (Figure 1).
Aortic dissection most often happens because of a tear or damage to the inner wall of the aorta (see Figure 2). This very often occurs in the chest (thoracic) part of the aorta, but it may also occur in the abdominal aorta.
Tears in the intimal layer result in the propagation of dissection (proximally or distally) secondary to blood entering the intima-media space.
When a tear occurs, it creates 2 channels:
- One in which blood continues to travel
- Another where blood stays still
If the channel with non-traveling blood gets bigger, it can push on other branches of the aorta. This can narrow the other branches and reduce blood flow through them.
An aortic dissection may also cause abnormal widening or ballooning of the aorta (aneurysm).
An acute aortic dissection (<2 weeks) is associated with high morbidity and mortality rates. Mortality is highest in the first 7 days; indeed, many patients die before presentation to the emergency department or before diagnosis is made in the emergency department. Patients with chronic aortic dissection (>2 weeks) have a better prognosis. The mortality associated with aortic dissection is still high despite advancements in diagnostic and therapeutic modalities 1.
Although acute aortic dissection classically produces sudden onset of severe chest pain that often has a tearing or ripping quality, no one sign or symptom can positively identify acute aortic dissection. The clinical manifestations are diverse, making the diagnosis difficult and necessitating a high index of suspicion 1. An estimated 38% of acute aortic dissections are missed on initial evaluation 2.
Most classic aortic dissections begin at one of the following three distinct anatomic locations:
- Approximately 2.2 cm above the aortic root
- Distal to the left subclavian artery
- The aortic arch
The most common site of dissection is the first few centimeters of the ascending aorta, with 90% occurring within 10 cm of the aortic valve. The second most common site is just distal to the left subclavian artery. Between 5% and 10% of dissections do not have an obvious intimal tear. These often are attributed to rupture of the aortic vasa vasorum.
Ascending aortic involvement may result in death from wall rupture, hemopericardium and tamponade, occlusion of the coronary ostia with myocardial infarction, or severe aortic insufficiency. The nervi vascularis (ie, bundles of nerve fibers found in the aortic adventitia) are involved in the production of pain.
Keeping the descending aorta in mind is important. The descending aorta is the location of most late clinical events after any dissection of the aorta 3.
- In the United States, aortic dissection is an uncommon disease. The true frequency of aortic dissection is difficult to estimate, and most estimates are based on autopsy studies. Evidence of aortic dissection is found in 1-3% of all autopsies (1 in 350 cadavers) 4. The incidence of aortic dissection is estimated to be 5-30 cases per 1 million people per year. Aortic dissection occurs once per 10,000 patients admitted to the hospital; approximately 2000 new cases are reported each year in the United States.
- Aortic dissection is more common in blacks than in whites and is less common in Asians than in whites. Also, it is more common in males than females, with a male-to-female ratio of 2-3:1.
- Approximately 75% of dissections occur in those aged 40-70 years, with a peak in the range of 50-65 years 1. Patients with Marfan syndrome present earlier, usually in the third and fourth decades of life.
There are no validated clinical decision rules to help identify acute aortic dissection. The diagnosis is best made when there is high clinical suspicion. A good patient history and physical examination are essential, along with imaging studies, electrocardiography, and laboratory studies (see aortic dissection diagnosis below).
Acute aortic dissection can be treated surgically or medically. In surgical treatment, the area of the aorta with the intimal tear is usually resected and replaced with a Dacron graft.
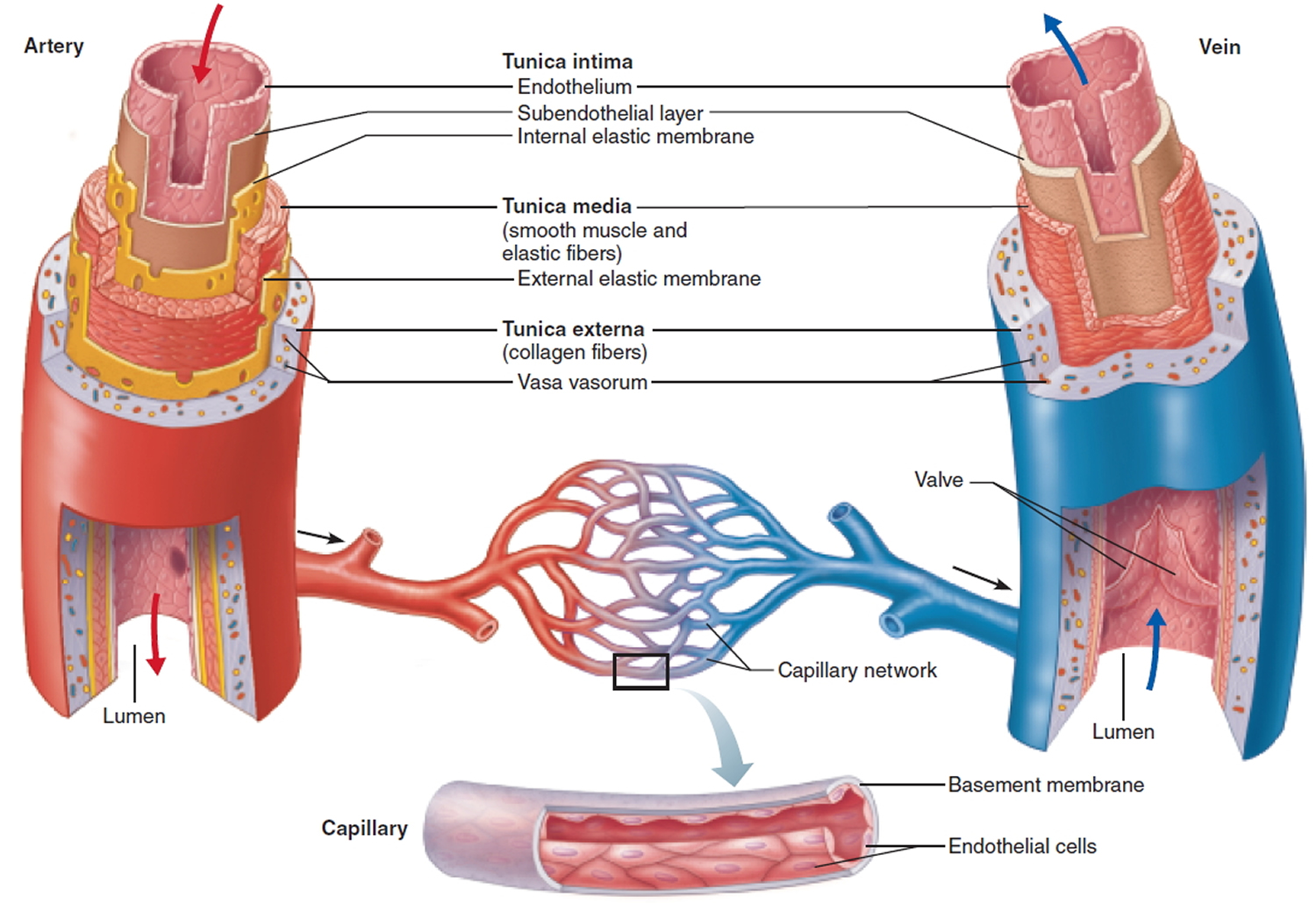
The aorta has three layers (see Figure 2):
- The inner layer (or intima)
- The middle layer (or media)
- The outer layer (or adventitia)
The aorta is composed of the intima, media, and adventitia. The intima, the innermost layer, is thin, delicate, lined by endothelium, and easily traumatized.
The media is responsible for imparting strength to the aorta and consists of laminated but intertwining sheets of elastic tissue. The arrangement of these sheets in a spiral provides the aorta with its maximum allowable tensile strength. The aortic media contains very little smooth muscle and collagen between the elastic layers and thus has increased distensibility, elasticity, and tensile strength. This contrasts with peripheral arteries, which, in comparison, have more smooth muscle and collagen between the elastic layers.
The outermost layer of the aorta is adventitia. This largely consists of collagen. The vasa vasorum, which supplies blood to the outer half of the aortic wall, lies within the adventitia. The nervi vascularis, bundles of nerve fibers found in the aortic adventitia, are involved in the production of pain, which occurs with acute stretching of the aortic wall from a dissection 5. The aorta does not have a serosal layer.
The aorta plays an integral role in the forward circulation of the blood in diastole. During left ventricular contraction, the aorta is distended by blood flowing from the left ventricle, and kinetic energy from the ventricle is transformed into potential energy stored in the aortic wall. During recoil of the aortic wall, this potential energy is converted to kinetic energy, propelling the blood within the aorta to the peripheral vasculature.
The volume of blood ejected into the aorta, the compliance of the aorta, and resistance to blood flow are responsible for the systolic pressures within the aorta. Resistance is mainly due to the tone of the peripheral vessels, although the inertia exerted by the column of blood during ventricular systole also plays a small part.
The aorta has thoracic and abdominal regions. The thoracic aorta is divided into the ascending, arch, and descending segments; the abdominal aorta is divided into suprarenal and infrarenal segments. The ascending aorta is the anterior tubular portion of the thoracic aorta from the aortic root proximally to the innominate artery distally. The ascending aorta is 5 cm long and is made up of the aortic root and an upper tubular segment.
The aortic root consists of the aortic valve, sinuses of Valsalva, and left and right coronary arteries. It extends from the aortic valve to the sinotubular junction and supports the base of the aortic leaflets. The aortic root allows the three sinuses of Valsalva to bulge outward, facilitating the full excursion of the leaflets in systole. The left and right coronary arteries arise from these sinuses.
The upper tubular segment of the ascending aorta starts at the sinotubular junction and ends at the beginning of the aortic arch. The ascending aorta lies slightly to the right of the midline, with its proximal portion in the pericardial cavity. Structures around the aorta include the pulmonary artery anteriorly; the left atrium, right pulmonary artery, and right mainstem bronchus posteriorly; and the right atrium and superior vena cava to the right.
When a problem occurs with the aorta, the heart and the entire body’s blood supply can be jeopardized (see Figure 1).
Figure 1. Aorta and its principal branches
Figure 2. Layers of blood vessels
Figure 3. Aortic dissection
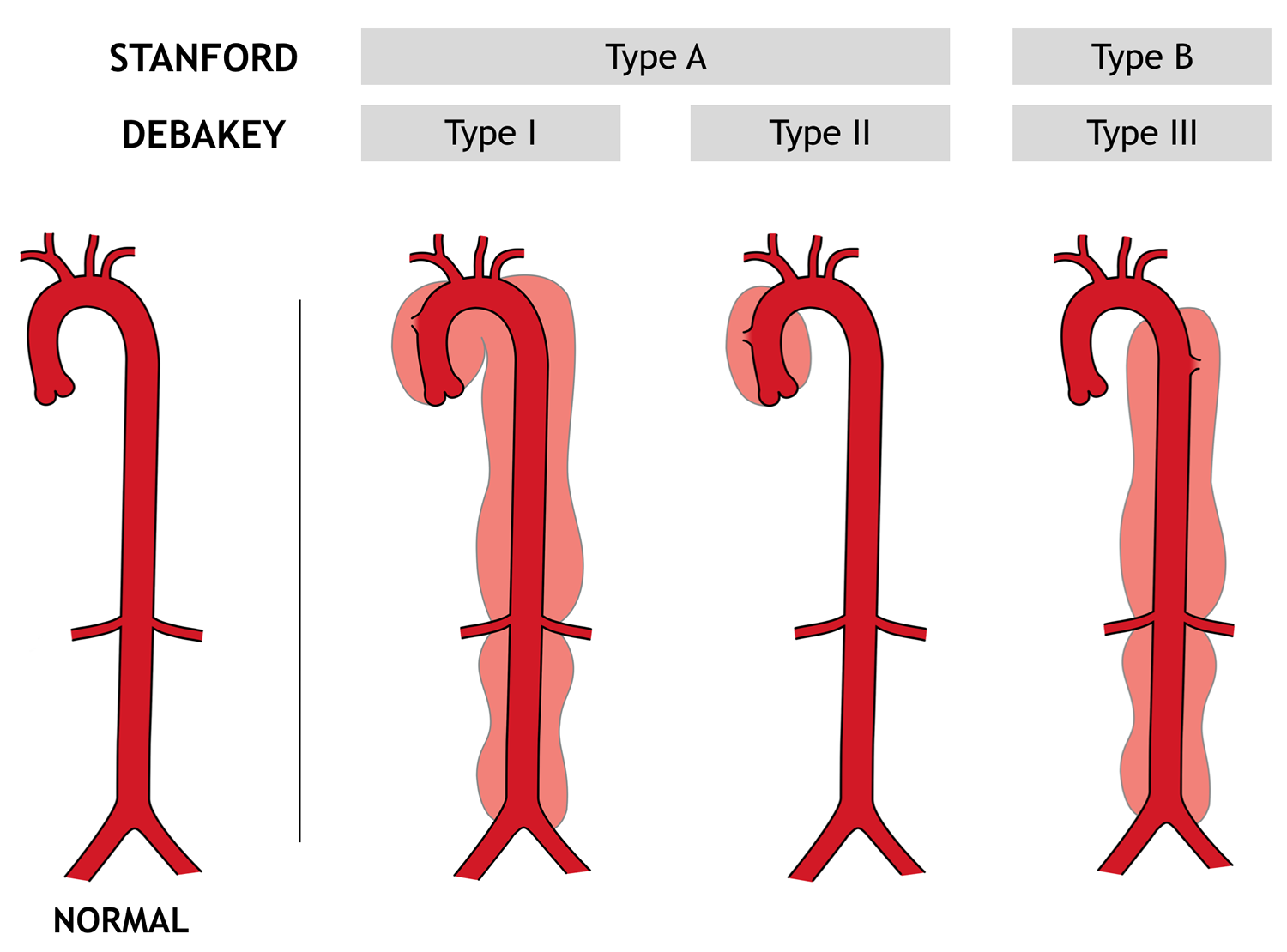
 Classification of aortic dissection
Classification of aortic dissection
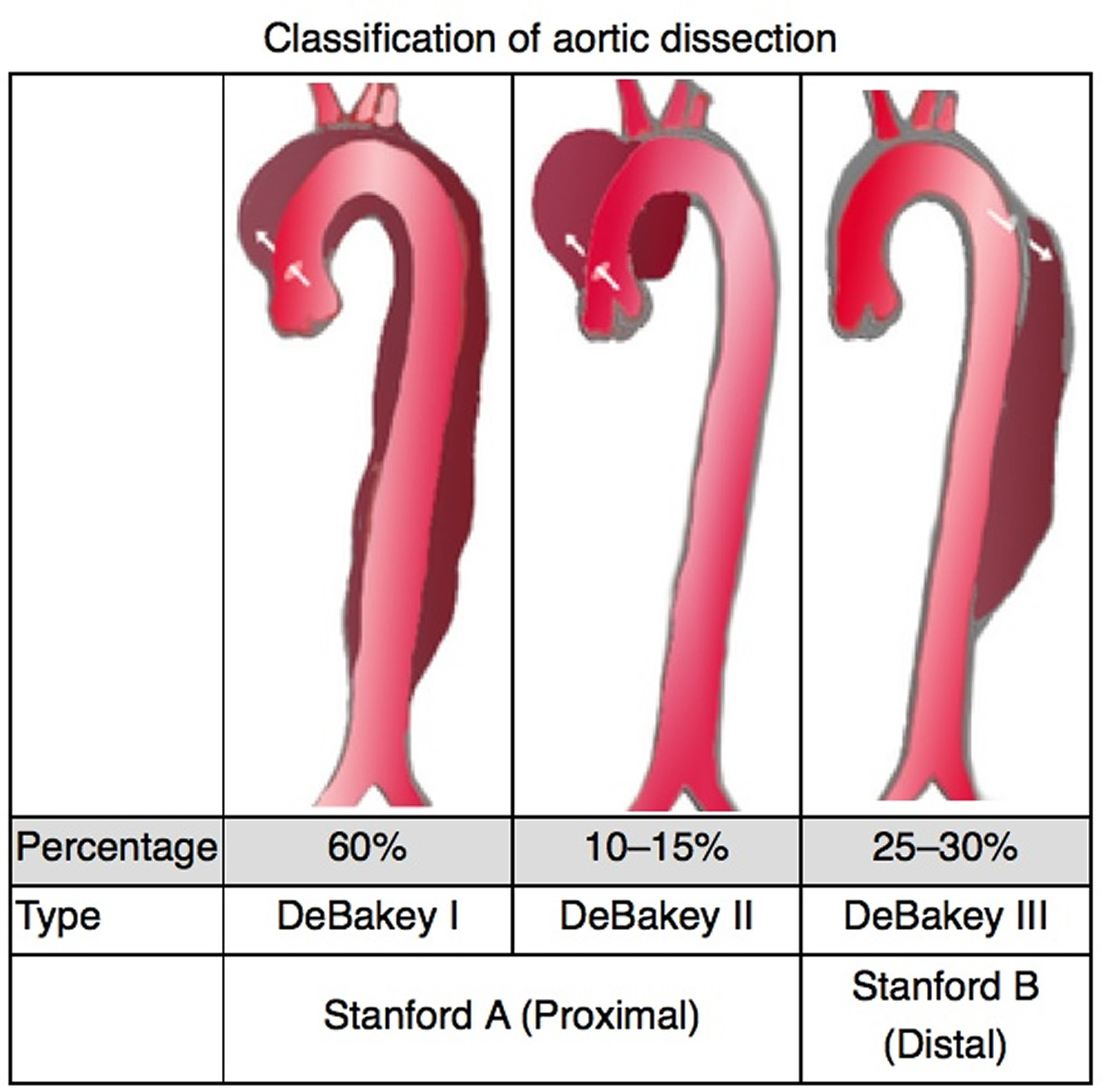
Two major anatomic classification schemes for aortic dissection are the DeBakey and the Stanford systems (see Figure 4 below) 6.
DeBakey et al classified aortic dissection into three types, as follows:
- Type I – The intimal tear occurs in the ascending aorta, but the descending aorta is also involved
- Type II – Only the ascending aorta is involved
- Type III – Only the descending aorta is involved; type IIIA originates distal to the left subclavian artery and extends as far as the diaphragm, whereas type IIIB involves the descending aorta below the diaphragm
The Stanford classification specified two types, as follows:
- Type A aortic dissection – The ascending aorta is involved (DeBakey types I and II)
- Type B aortic dissection – The descending aorta is involved (DeBakey type III)
This system also helps delineate treatment. Type A aortic dissections usually must be treated surgically, whereas type B aortic dissections are managed medically under most conditions 7.
Figure 4. Aortic dissection type (classification)
Aortic dissection prognosis
Hospital-based mortality for aortic dissection is approximately 30%.
Without treatment, about 80% of people who have an aortic dissection die within the first 2 weeks. With treatment, about 70% who have dissection of the first part of the aorta and about 90% of those who have dissection of the aorta farther from the heart survive to leave the hospital. About 60% of people who survive the first 2 weeks are still alive 5 years after treatment, and 40% live at least 10 years. Of people who die after the first 2 weeks, about one third die of complications of the dissection, and the other two thirds die of other disorders.
Patients with type A aortic dissection who undergo surgical treatment have a 30% mortality; patients who receive medical treatment have a 60% mortality. Comorbidities and advanced aged can pose a contraindication to surgery in selected patients. Medically treated patients with type B dissection have a 10% mortality; surgically treated patients with type B dissection have a 30% mortality.
An acute aortic dissection (<2 weeks) is associated with high morbidity and mortality rates (highest mortality in the first 7 days). From 1% to 2% of patients with aortic dissection die per hour for the first 24-48 hours. Patients with chronic aortic dissection (>2 weeks) have a better prognosis.
Complications of Aortic dissection
Aortic dissection may decrease or stop the blood flow to many different parts of the body. This may result in short-term or long-term problems, or damage to the:
- Brain
- Heart
- Intestines or bowels
- Kidneys
- Legs
Complications include:
- Stroke (if the cerebral arteries, which supply the brain, are blocked),
- Heart attack (if the coronary arteries, which supply the heart muscle, are blocked),
- Kidney failure (if the renal arteries, which supply the kidneys, are blocked)
- Nerve and/or spinal cord damage that causes tingling or an inability to move a limb (if the spinal arteries are blocked).
Blood may leak from the dissection and accumulate in the chest. Blood leaking from a dissection near the heart may enter the pericardial space (between the two layers of membranes that surround the heart), preventing the heart from filling properly and causing cardiac tamponade—a life-threatening disorder.
A dissection that involves the first few inches of the aorta (ascending aorta) closest to the heart may affect the attachments of the aortic valve, the heart valve that keeps blood from flowing back into the heart. If the aortic valve attachments are weakened, the valve can leak, causing heart failure.
Aortic dissection causes
Congenital and acquired factors, alone or in combination, can lead to aortic dissection. Aortic dissection is more common in patients with hypertension (high blood pressure), connective tissue disorders, congenital aortic stenosis, or a bicuspid aortic valve 8, as well as in those with first-degree relatives with a history of thoracic dissection. These diseases affect the media of the aorta and predispose it to dissection.
Aortic dissection occurs in about 2 out of every 10,000 people. It can affect anyone, but is most often seen in men ages 40 to 70.
The exact cause is unknown, but most common cause of aortic dissection is:
- Deterioration of the artery’s wall because of longstanding high blood pressure
More than two thirds of people who have an aortic dissection have high blood pressure.
The more common risks include:
- Aging.
- Atherosclerosis
- Blunt trauma to the chest, such as hitting the steering wheel of a car during an accident
Less common causes of aortic dissection include:
- Hereditary connective tissue disorders, especially Marfan syndrome and Ehlers-Danlos syndrome
- Birth defects of the heart and blood vessels, such as coarctation (narrowing) of the aorta, patent ductus arteriosus (a connection between the aorta and the pulmonary artery), and defects of the aortic valve
- Arteriosclerosis
- Injury such as a car crash or fall causing a strong blow to the chest
Rarely, a dissection occurs accidentally when doctors are inserting a catheter into an artery (for example, during aortography or angiography) or doing surgery on the heart or blood vessels.
Other risk factors and conditions linked to aortic dissection include:
- Bicuspid aortic valve
- Coarctation (narrowing) of the aorta
- Connective tissue disorders (such as Marfan syndrome and Ehlers-Danlos syndrome) and rare genetic disorders
- Heart surgery or procedures
- Pregnancy
- Swelling of the blood vessels due to conditions such as arteritis and syphilis
Congenital causes
Aortopathy may be due to the following heritable diseases:
- Marfan syndrome
- Ehlers-Danlos syndrome
- Annuloaortic ectasia
- Familial aortic dissections
- Adult polycystic kidney disease
- Turner syndrome
- Noonan syndrome
- Osteogenesis imperfecta
- Bicuspid aortic valve
- Coarctation of the aorta
- Connective tissue disorders
- Metabolic disorders (eg, homocystinuria, familial hypercholesterolemia)
Acquired conditions
Arterial hypertension is an important predisposing factor for aortic dissection 1. Of patients with aortic dissection, 70% have elevated blood pressure. Hypertension or pulsatile blood flow can propagate the dissection.
Pregnancy can be a risk factor for aortic dissection, particularly in patients with an underlying anomaly such as Marfan syndrome. An estimated 50% of all cases of aortic dissection that occur in women younger than 40 years are associated with pregnancy. Most cases occur in the third trimester or early postpartum period.
Other acquired causes of aortic dissection include the following:
- Syphilitic aortitis
- Deceleration injury possibly with related chest trauma
- Cocaine use
Cystic medial necrosis
The normal aorta contains collagen, elastin, and smooth muscle cells that contribute the intima, media, and adventitia, which are the layers of the aorta. With aging, degenerative changes lead to breakdown of the collagen, elastin, and smooth muscle and an increase in basophilic ground substance. This condition is termed cystic medial necrosis. Atherosclerosis that causes occlusion of the vasa vasorum also produces this disorder. Cystic medial necrosis is the hallmark histologic change associated with dissection in those with Marfan syndrome.
Sources disagree over the accuracy of this term in elderly patients because the true histopathologic changes are neither cystic nor necrotic. Researchers have used the term cystic medial degeneration.
Early on, cystic medial necrosis described an accumulation of basophilic ground substance in the media with the formation of cystlike pools. The media in these focal areas may show loss of cells (ie, necrosis). This term still is used commonly to describe the histopathologic changes that occur.
Iatrogenic causes
Iatrogenic aortic dissection can result from cardiologic procedures such as the following:
- Aortic and mitral valve replacements
- Coronary artery bypass graft surgery
- Percutaneous catheter placement (eg, cardiac catheterization, percutaneous transluminal coronary angioplasty)
Aortic dissection occurs when the layers are split in the process of cannulation or aortotomy.
Prevention of aortic dissection
Many cases of aortic dissection cannot be prevented.
Things you can do to reduce your risk include:
- Treating and controlling hardening of the arteries (atherosclerosis)
- Keeping high blood pressure under control, especially if you are at risk for dissection
- Taking safety precautions to prevent injuries that can cause dissections
- If you have been diagnosed with Marfan or Ehlers-Danlos syndrome, making sure you regularly follow-up with your provider.
Aortic dissection symptoms
Aortic dissection can be rapidly fatal, with many patients dying before presentation to the emergency department or before diagnosis is made in the emergency department.
No one sign or symptom can positively identify acute aortic dissection.
In most cases, the symptoms begin suddenly, and include severe chest pain. The pain may feel like a heart attack.
- Sudden onset of severe chest pain that often has a tearing or ripping quality (classic symptom).
- Chest pain may be mild.
- No pain in about 10% of patients.
- Pain can be described as sharp, stabbing, tearing, or ripping.
- It is felt below the chest bone, and then moves under the shoulder blades or to the back.
- Pain can move to the shoulder, neck, arm, jaw, abdomen, or hips.
- The pain changes position, often moving to the arms and legs as the aortic dissection gets worse.
- Anterior chest pain: Usually associated with anterior arch or aortic root dissection.
- Neck or jaw pain: With aortic arch involvement and extension into the great vessels.
- Tearing or ripping intrascapular pain: May indicate dissection involving the descending aorta.
- Syncope
- Altered mental status
- Numbness and tingling, pain, or weakness in the extremities
- Horner syndrome (ie, ptosis, miosis, anhidrosis)
- Dyspnea
- Hemoptysis
- Dysphagia
- Flank pain (with renal artery involvement)
- Fever
Symptoms are caused by a decrease of blood flowing to the rest of the body, and can include:
- Anxiety and a feeling of doom or death
- Fainting or dizziness
- Heavy sweating (clammy skin)
- Nausea and vomiting
- Pale skin (pallor)
- Rapid, weak pulse
- Shortness of breath and trouble breathing when lying flat (orthopnea)
Other symptoms may include:
- Pain in the abdomen (with abdominal aorta involvement)
- Stroke (cerebrovascular accident) symptoms (eg, hemianesthesia, and hemiparesis, hemiplegia) 9
- Swallowing difficulties from pressure on the esophagus
Patients with acute aortic dissection typically present with the sudden onset of severe chest pain, although this description is not universal. Some patients present with only mild pain, often mistaken for a symptom of musculoskeletal conditions in the thorax, groin, or back. Consider thoracic aortic dissection in the differential diagnosis of all patients presenting with chest pain.
The location of the pain may indicate where the dissection arises. Anterior chest pain and chest pain that mimics acute myocardial infarction usually are associated with anterior arch or aortic root dissection. This is caused by the dissection interrupting flow to the coronary arteries, resulting in myocardial ischemia. Pain in the neck or jaw indicates that the dissection involves the aortic arch and extends into the great vessels.
Tearing or ripping pain in the intrascapular area may indicate that the dissection involves the descending aorta. The pain typically changes as the dissection evolves.
The pain of aortic dissection is typically distinguished from the pain of acute myocardial infarction by its abrupt onset and maximal severity at onset, though the presentations of the two conditions overlap to some degree and are easily confused. Aortic dissection can be presumed in patients with symptoms and signs suggestive of myocardial infarction but without classic electrocardiographic (ECG) findings.
Aortic dissection is painless in about 10% of patients 9. Painless dissection is more common in those with neurologic complications from the dissection and those with Marfan syndrome.
Neurologic deficits are a presenting sign in as many as 20% of cases. Syncope is part of the early course of aortic dissection in approximately 5% of patients and may be the result of increased vagal tone, hypovolemia, or dysrhythmia 9. Cerebrovascular accident (stroke) symptoms include hemianesthesia and hemiparesis or hemiplegia 9. Altered mental status is also reported. Patients with peripheral nerve ischemia can present with numbness and tingling, pain, or weakness in the extremities.
Horner syndrome is caused by interruption in the cervical sympathetic ganglia and manifests as ptosis, miosis, and anhidrosis. Hoarseness from recurrent laryngeal nerve compression has also been described.
Cardiovascular manifestations involve symptoms suggestive of congestive heart failure 9 secondary to acute severe aortic regurgitation. These include dyspnea and orthopnea.
Respiratory symptoms can include dyspnea and hemoptysis if dissection ruptures into the pleura or if tracheal or bronchial obstruction has occurred. Physical findings of a hemothorax may be found if the dissection ruptures into the pleura.
Other manifestations include the following 10:
- Dysphagia from compression of the esophagus
- Flank pain if the renal artery is involved
- Abdominal pain if the dissection involves the abdominal aorta
- Fever
- Anxiety and premonitions of death
A retrospective chart review of 83 patients with a thoracic aortic dissection revealed that only 40% of alert patients were asked the basic questions about their pain. Remember to cover the P, Q, R, S, and T (position, quality, radiation, severity, and timing) of pain in all able patients. Timing includes the rate of onset, duration, and frequency of episodes. Also ask about migration of pain, aggravating or alleviating factors, and associated symptoms.
Aortic dissection diagnosis
The diagnosis of acute aortic dissection requires a high index of suspicion and involves the following:
- History and physical examination
- Imaging studies
- Electrocardiography
- Complete blood count, serum chemistry studies, cardiac marker assays
The health care provider will take your family history and listen to your heart, lungs, and abdomen with a stethoscope. The exam may find:
- A “blowing” murmur over the aorta, heart murmur, or other abnormal sound
- A difference in blood pressure between the right and left arms, or between the arms and legs
- Low blood pressure
- Signs resembling a heart attack
- Signs of shock, but with normal blood pressure
The distinctive symptoms of an aortic dissection usually make the diagnosis obvious to doctors, although the disorder causes a variety of symptoms that sometimes resemble those of other disorders. In about two thirds of people with aortic dissection, pulses in the arms and legs are diminished or absent. Depending on the location of the dissection along the aorta, there may be a difference in blood pressure between the right and left arm. A dissection that is moving backward toward the heart may cause a murmur that can be heard through a stethoscope.
Possible physical examination findings include the following:
- Hypertension (high blood pressure)
- Hypotension (low blood pressure)
- Interarm blood pressure differential greater than 20 mm Hg
- Signs of aortic regurgitation (eg, bounding pulses, wide pulse pressure, diastolic murmurs)
- Findings suggestive of cardiac tamponade (eg, muffled heart sounds, hypotension, pulsus paradoxus, jugular venous distention, Kussmaul sign)
- Neurologic deficits (eg, syncope, altered mental status)
- Peripheral paresthesias
- Horner syndrome
- New diastolic murmur
- Asymmetrical pulses (eg, carotid, brachial, femoral)
- Progression or development of bruits
Possible laboratory study findings include the following:
- Leukocytosis: Stress state
- Decreases in hemoglobin and hematocrit values: Leaking or rupture of the dissection
- Elevation of the blood urea nitrogen and creatinine levels: Renal artery involvement or prerenal azotemia
- Elevation of the myocardial muscle creatine kinase isoenzyme, myoglobin, and troponin I and T levels: Myocardial ischemia from coronary artery involvement
- Lactate dehydrogenase elevation: Hemolysis in the false lumen
- Smooth muscle myosin heavy-chain assay: Increased levels in the first 24 hours are 90% sensitive and 97% specific for aortic dissection
- Fibrin degradation product (FDP) elevation: In symptomatic patients, a plasma FDP of 12.6 μg/mL or higher suggests possible aortic dissection with a patent false lumen; an FDP level of 5.6 μg/mL or higher suggests the possibility of dissection with complete thrombosis of the false lumen 11
- Blood work to rule out a heart attack is needed.
Aortic dissection or aortic aneurysm may be seen on:
- Aortic angiography
- Chest x-ray
- Chest MRI
- CT scan of chest with dye
- Doppler ultrasonography (occasionally performed)
- Echocardiogram
- Transesophageal echocardiogram (TEE)
Chest radiography
Chest x-rays are the first step in detecting aortic dissection. X-rays show a widened aorta in 90% of people with symptoms. However, this finding may be due to other disorders. Hemothorax may be evident if the dissection has ruptured.
Computed tomography (CT) done after injecting a contrast agent (dye) can quickly and reliably detect aortic dissection and thus is useful in an emergency.
Computed tomography (CT) with contrast:
- The definitive test in most patients with suspicion of aortic dissection 12
- Useful only in hemodynamically stable patients
- Findings help determine whether hypothermic circulatory arrest is necessary for surgery
Standard or transesophageal echocardiography or magnetic resonance angiography can also reliably detect aortic dissections, even very small ones.
Echocardiography
- Transesophageal echocardiography (TEE) is more accurate than transthoracic echocardiography (TTE) 13
- Transesophageal echocardiography is most useful in ascending aortic dissections
- Transesophageal echocardiography is as sensitive and specific as CT and magnetic resonance imaging (MRI)
- Transesophageal echocardiography is strongly dependent on operator experience
MRI
- The most sensitive method for diagnosing aortic dissection
- Specificity is similar to that of CT scan
Aortography
- Has been the diagnostic criterion standard study for aortic dissection
- Is being replaced by newer, safer imaging modalities
Aortic dissection treatment
Aortic dissection is a life-threatening condition and needs to be treated right away. Acute aortic dissection can be treated surgically or medically.
- Dissections that occur in the part of the aorta that is leaving the heart (ascending) are treated with surgery.
- Dissections that occur in other parts of the aorta (descending) may be managed with surgery or medicines.
Drugs that lower blood pressure may be prescribed. These drugs may be given through a vein (intravenously). Beta-blockers are the first drugs of choice. Strong pain relievers are very often needed.
Type A aortic dissection
Treatment for type A aortic dissection may include:
- Surgery. Surgeons remove as much of the dissected aorta as possible, block the entry of blood into the aortic wall and reconstruct the aorta with a synthetic tube called a graft. If the aortic valve leaks as a result of the damaged aorta, it may be replaced at the same time. The new valve is placed within the graft used to reconstruct the aorta.
- Medications. Some medications, such as beta blockers and nitroprusside (Nitropress), reduce heart rate and lower blood pressure, which can prevent the aortic dissection from worsening. They may be given to people with type A aortic dissection to stabilize blood pressure before surgery.
Type B aortic dissection
Treatment of type B aortic dissection may include:
- Medications. The same medications that are used to treat type A aortic dissection may be used without surgery to treat type B aortic dissections.
- Surgery. The procedure is similar to that used to correct a type A aortic dissection. Sometimes stents — small wire mesh tubes that act as a sort of scaffolding — may be placed in the aorta to repair complicated type B aortic dissections.
After treatment, you may need to take blood pressure lowering medication for life. In addition, you may need follow-up CTs or MRIs periodically to monitor your condition.
Medical management
Initial therapeutic goals include the elimination of pain and the reduction of systolic blood pressure to 100-120 mm Hg or to the lowest level commensurate with adequate vital organ (ie, cardiac, cerebral, renal) perfusion. Whether systolic hypertension or pain is present, beta-blockers are used to reduce arterial delta pressure/delta time (dP/dt).
To prevent exacerbations of tachycardia and hypertension, treat patients with intravenous morphine sulfate. This reduces the force of cardiac contraction and the rate of rise of the aortic pressure. It then retards the propagation of the dissection and delays rupture.
Medical management includes the following:
- Decreasing the blood pressure and the shearing forces of myocardial contractility
- Antihypertensive therapy, including beta blockers, is the treatment of choice for all stable chronic aortic dissections
- Pain management: Narcotics and opiates are the preferred agents.
People with an aortic dissection are admitted to an intensive care unit, where their vital signs (pulse, blood pressure, and rate of breathing) are closely monitored. Death can occur a few hours after an aortic dissection begins. Therefore, as soon as possible, drugs are given intravenously to reduce the heart rate and blood pressure to the lowest level that can maintain a sufficient blood supply to the brain, heart, and kidneys. The lower heart rate and blood pressure help limit the spread of the dissection. Soon after drug therapy begins, doctors must decide whether to recommend surgery or to continue drug therapy without surgery.
Doctors almost always recommend surgery for dissections that involve the first few inches of the aorta (ascending aorta) closest to the heart, unless complications of the dissection make the risk of surgery too high. During surgery, surgeons remove the largest possible area of dissected aorta, close the channel that was torn between the middle and outer layers of the aorta’s wall, and rebuild the aorta with a synthetic graft. If the aortic valve is leaking, surgeons repair or replace it. Removal and repair of a dissected aorta usually takes 3 to 6 hours, and the hospital stay is usually 7 to 10 days.
For dissections in the aorta farther from the heart (descending aorta), doctors usually continue drug therapy without surgery or consider placement of an endovascular stent-graft, To do an endovascular stent-graft, doctors thread a long, thin wire through the large artery in the groin (femoral artery) and up to the dissecting area. Then they slide the stent-graft, which is a hollow tube like a collapsible straw, over the wire and inside the damaged area of the aorta. Then the stent-graft is opened, forming a stable channel for blood flow . This procedure takes 2 to 4 hours, and the hospital stay is usually 1 to 3 days. Stent-grafts, which are less invasive than open surgery, have improved the survival rate and lowered the risk of complications for people with dissections of the descending aorta.
Surgery or a stent-graft repair is always necessary if the dissection causes the artery to leak blood, blocks the blood supply to the legs or to vital organs in the abdomen, causes symptoms, is enlarging, or occurs in a person with Marfan syndrome.
All people who have an aortic dissection, including those treated surgically, have to take drug therapy to keep their blood pressure down, usually for the rest of their lives. Such therapy helps reduce stress on the aorta. Drug therapy to lower blood pressure usually consists of a beta-blocker or calcium channel blocker plus another antihypertensive drug such as an angiotensin-converting enzyme (ACE) inhibitor. Cholesterol-lowering drugs and diet modification are used if the person has atherosclerosis.
Doctors watch closely for complications that can occur in people who have had an aortic dissection. The most important are another dissection, development of aneurysms in the weakened aorta, and increasing leakage backward through the aortic valve. Any of these complications may require surgical repair.
Aortic dissection surgery
In surgical treatment, the area of the aorta with the intimal tear is usually resected and replaced with a Dacron graft. Endovascular repair is emerging as the preferred treatment for descending aortic dissection.
Two techniques may be used for surgery:
- Standard, open surgery. This requires a surgical incision that is made in the chest or abdomen.
- Endovascular aortic repair. This surgery is done without any major surgical incisions.
Emergency surgical correction is the preferred treatment for the following:
- Stanford type A (DeBakey type I and II) ascending aortic dissection
- Complicated Stanford type B (DeBakey type III) aortic dissections with specific clinical or radiologic evidence
If the aortic valve is damaged, valve replacement is needed. If the heart arteries are involved, a coronary bypass is also performed.
Surgical Overview
The major objectives of surgery for aortic dissection are to alleviate the symptoms, decrease the frequency of complications, and prevent aortic rupture and death. The affected layers of the aorta are sutured together, and the aorta is reinforced with a Dacron graft.
Improved cardiopulmonary bypass circuits have decreased the prevalence of injury to blood elements. Morbidity and mortality associated with this highly invasive surgery have decreased with the introduction of profound hypothermic circulatory arrest and retrograde cerebral perfusion 14.
A number of advances have resulted in a decreased frequency of complications associated with surgery on the aorta. Dacron grafts with impregnated collagen or gelatin have been developed that are impervious to blood. The development of more impermeable grafts has greatly enhanced the surgical repair of thoracic aortic dissections. Such grafts include the following:
- Woven Dacron
- Collagen-impregnated Hemashield (Meadox Medicals) aortic grafts
- Gel-coated Carbo-Seal Ascending Aortic Prosthesis (Sulzer CarboMedics)
The operative mortality with ascending aortic dissection is usually less than 10%. Serious complications are rare.
Dissections involving the arch are more complicated than those involving only the ascending aorta because the innominate, carotid, and subclavian vessels branch from the arch. Deep hypothermic arrest is usually required. If the arrest time is less than 45 minutes, the rate of central nervous system (CNS) complications is lower than 10%. Retrograde cerebral perfusion may improve the protection of the CNS during the arrest period.
The mortality associated with aortic arch dissections is approximately 10-15%. Significant neurologic complications occur in an additional 10% of patients.
Postoperative complications for extensive disease involving the thoracoabdominal aorta include myocardial infarction, respiratory failure, renal failure, stroke, and paraparesis or paraplegia. The use of adjunct procedures has decreased the frequency of procedure-related spinal cord injury during descending aorta and thoracoabdominal surgeries. These include the following:
- Distal aortic perfusion
- Induction of profound hypothermia
- Cerebrospinal fluid (CSF) drainage
- Monitoring of somatosensory and motor evoked potentials in the brain and spinal cord
Endovascular therapy is rapidly emerging as the preferred treatment for descending aortic dissection, provided vascular access is available. This methodology still remains controversial for ascending dissection 15. A 2011 study that included 28 complicated acute aortic dissection patients treated with endovascular repair suggested that this technique has improved mortality as compared with traditional surgical interventions 16.
References- Patel PD, Arora RR. Pathophysiology, diagnosis, and management of aortic dissection. Ther Adv Cardiovasc Dis. 2008 Dec. 2(6):439-68.
- Sutherland A, Escano J, Coon TP. D-dimer as the sole screening test for acute aortic dissection: a review of the literature. Ann Emerg Med. 2008 Oct. 52(4):339-43.
- Mimoun L, Detaint D, Hamroun D, et al. Dissection in Marfan syndrome: the importance of the descending aorta. Eur Heart J. 2011 Feb. 32(4):443-9.
- Clouse WD, Hallett JW Jr, Schaff HV, et al. Acute aortic dissection: population-based incidence compared with degenerative aortic aneurysm rupture. Mayo Clin Proc. 2004 Feb. 79(2):176-80.
- Spiegel EA, Wassreman S. Experimentalstudien uber die Entsehungdes Aortenschmerzes und seine Leitung zum zentralnervensystem. Ztschr F. d. ges. Exper Med. 1926. 52:180-196.
- Aortic Dissection. https://emedicine.medscape.com/article/2062452-overview
- Niino T, Hata M, Sezai A, et al. Optimal clinical pathway for the patient with type B acute aortic dissection. Circ J. 2009 Feb. 73(2):264-8.
- Friedman WF, Silverman N. Congenital Heart Disease in Infancy and Childhood. Braunwald E, Zipes DP, Libby P, eds. Heart Disease: A Textbook of Cardiovascular Medicine. 6th ed. Philadelphia: WB Saunders; 2001. 1546.
- Spittell PC, Spittell JA Jr, Joyce JW, et al. Clinical features and differential diagnosis of aortic dissection: experience with 236 cases (1980 through 1990). Mayo Clin Proc. 1993 Jul. 68(7):642-51.
- Roberts WC. Aortic dissection: anatomy, consequences, and causes. Am Heart J. 1981 Feb. 101(2):195-214.
- Hagiwara A, Shimbo T, Kimira A, Sasaki R, Kobayashi K, Sato T. Using fibrin degradation products level to facilitate diagnostic evaluation of potential acute aortic dissection. J Thromb Thrombolysis. 2013 Jan. 35(1):15-22.
- [Guideline] Jacobs JE, Latson LA Jr, Abbara S, et al, Expert Panel on Cardiac Imaging. ACR Appropriateness Criteria® acute chest pain — suspected aortic dissection. American College of Radiology. https://acsearch.acr.org/docs/69402/Narrative/
- Meredith EL, Masani ND. Echocardiography in the emergency assessment of acute aortic syndromes. Eur J Echocardiogr. 2009 Jan. 10(1):i31-9.
- Fann JI, Smith JA, Miller DC, et al. Surgical management of aortic dissection during a 30-year period. Circulation. 1995 Nov 1. 92(9 Suppl):II113-21.
- Kasirajan K, Kwolek CJ, Gupta N, Fairman RM. Incidence of and outcomes after misaligned deployment of the talent thoracic stent graft system. J Vasc Surg. 2010 May. 51(5):1096-101.
- O’Donnell S, Geotchues A, Beavers F, Akbari C, Lowery R, Elmassry S, et al. Endovascular management of acute aortic dissections. J Vasc Surg. 2011 Nov. 54(5):1283-9.