What is fallopian tube
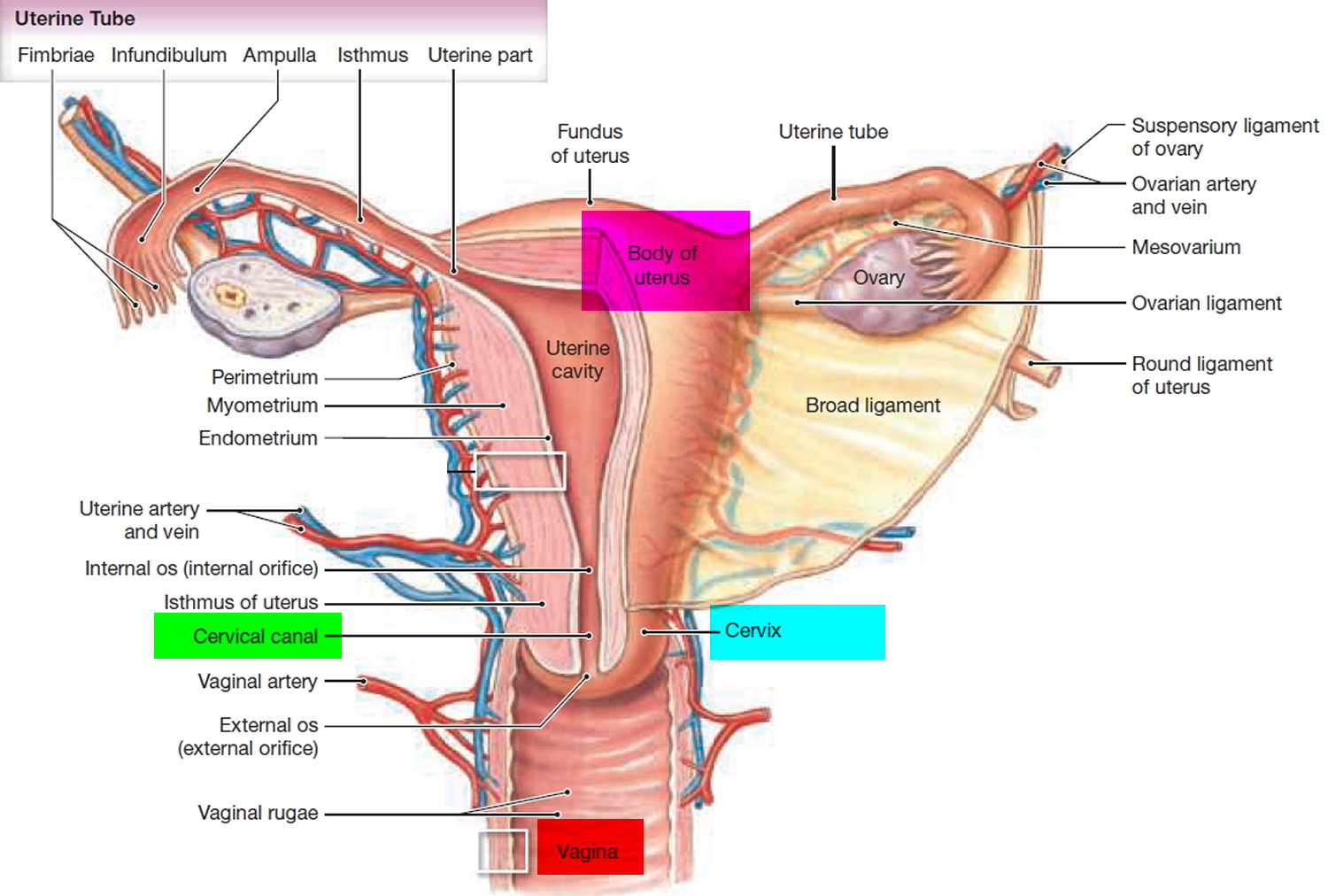
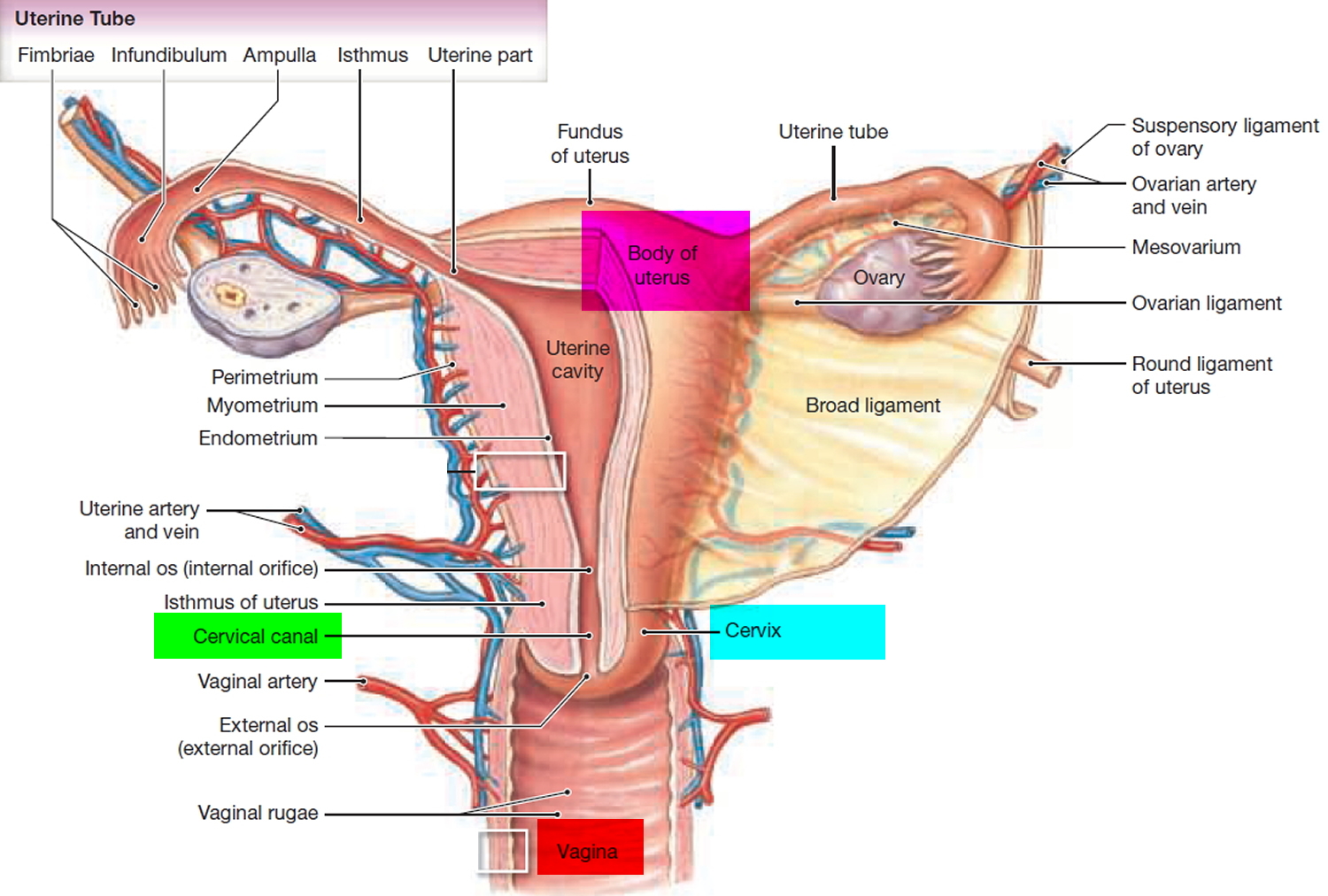
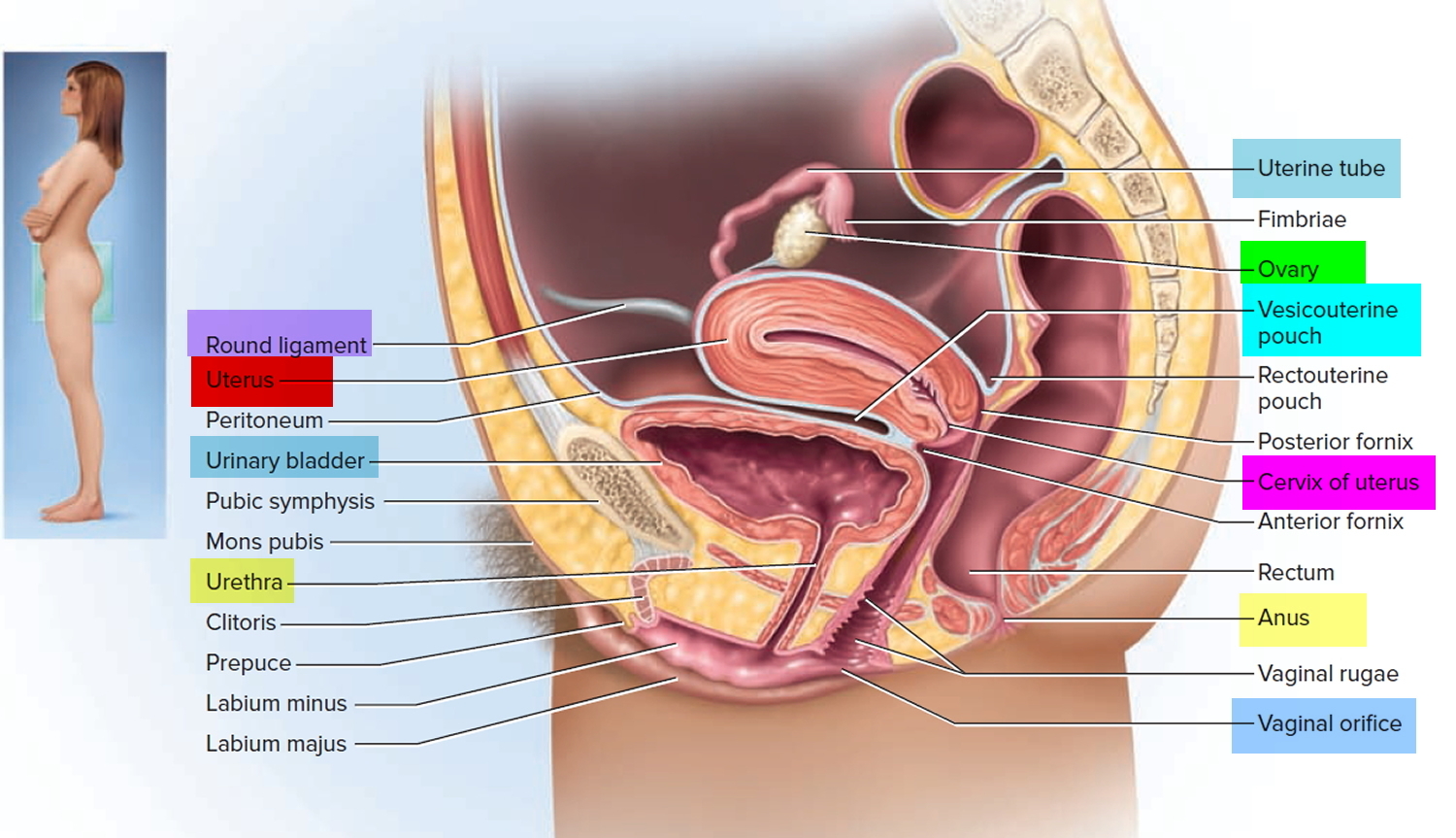
The fallopian tubes (uterine tubes or oviducts) open near the ovaries (Figure 1 and 2). Each fallopian tube is about 10 centimeters long and 0.7 centimeters in diameter, the middle and longest part of the fallopian tube is the ampulla and the segment near the uterus is a narrower isthmus – which passes medially to the uterus, penetrates its wall and opens into the uterine cavity. Near each ovary, a fallopian tube expands, forming a funnel-shaped infundibulum, which partially encircles the ovary. Fingerlike extensions called fimbriae fringe the infundibulum margin. Although the infundibulum generally does not touch the ovary, one of the larger fimbriae connects directly to the ovary.
The fallopian tube is enclosed in the mesosalpinx, which is the superior margin of the broad ligament.
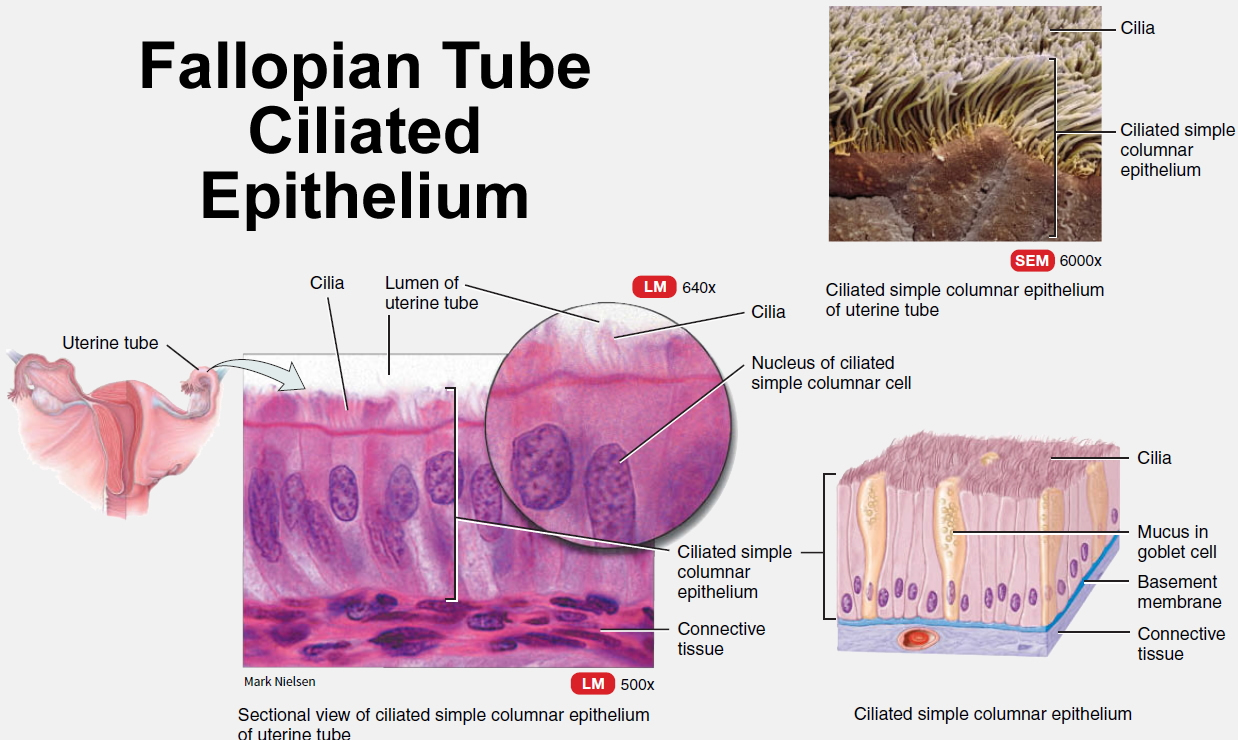
Simple columnar epithelial cells, some ciliated, line the fallopian tube. The epithelium secretes mucus, and the cilia beat toward the uterus. These actions help draw the secondary oocyte (an immature egg cell of the female ovary) and expelled follicular fluid into the infundibulum following ovulation. Ciliary action and peristaltic contractions of the fallopian tube’s smooth muscle layer aid transport of the oocyte down the uterine tube. Fertilization usually occurs in the fallopian tube.
Figure 1. Fallopian tube
Figure 2. Fallopian tube location
Fallopian tube function
The normal epithelium of the fallopian tube is comprised of two cell types: ciliated and secretory. The ciliated cells of the fallopian tube play a major role in the transport of the ovum, the sperm cells, and the zygote (a fertilized ovum). The human ovary usually releases one egg (oocyte) per month, around day 14 of a typical 28-day ovarian cycle. This egg is swept into the fallopian (uterine) tube by the beating of cilia on the fallopian tube’s epithelial cells and begins a 3-day trip down the fallopian tube toward the uterus. If the egg is not fertilized, it dies within 24 hours and gets no more than one-third of the way to the uterus. Therefore, if a sperm is to fertilize an egg, it must migrate up the fallopian tube to meet it. The vast majority of sperm never make it. Although a typical ejaculation may contain 200 million sperm, many of these are destroyed by vaginal acid or drain out of the vagina; others fail to get through the cervical canal into the uterus; still more are destroyed by leukocytes in the uterus; and half of the survivors of all these ordeals are likely to go up the wrong fallopian tube tube. Only about 200 (1 in a million) reach the general vicinity of the egg.
The fallopian tube secretory cells secrete mucus that slows the progression of the spermatozoa through the fallopian tube, preserves their viability and facilitates their appropriate capacitation (functional maturation of the spermatozoon and is required to render them competent to fertilize an oocyte) and activation 1.
Figure 3. Fallopian tube ciliated epithelium
Fallopian tube cyst
Fallopian tube cysts are the remnants of the congenital paramesonephric duct and are considered benign cysts 2. The prevalence of fallopian tube or paraovarian cysts in a healthy population is 7%-10% regardless of age 3. In another study involving pediatric and adolescent population, the incidence of fallopian tube cysts was 7.3% in this pediatric and adolescent population 4. In addition, this is the first study to confirm presence of fallopian tube cysts in prepubertal females 4. Surgeon should be aware of these benign cysts because they are frequently a source of abdominal pain in young girls. As nonphysiologic cysts, these will not resolve spontaneously and may increase in size and/or be at risk for adnexal torsion. Most fallopian tube and paraovarian cysts are asymptomatic and accidentally discovered, but they may occasionally give rise to clinical problems due to enlargement or torsion. Ultimately, surgical management is required for definitive resolution.
In a study 5 of 338 young patients with a clinical picture of acute appendicitis, 44 had acute appendicitis plus a coincidental paratubal cyst, and 2 additional cases of torsion of huge paratubal cysts (13.6%) were detected. Only 2.5% were associated with ectopic tubal pregnancy, which was seen just distal to the site of the fallopian tube cysts. The exact implication of the presence of fallopian tube cyst in the causation of ectopic pregnancy is not clear 6, but it may be explained by disturbed tubal motility, compression of the already narrow tubal lumen, or defective vascularization of the fallopian tubes.
In another study involving 19 women, the authors found most of the patients (84%) had adnexal (uterus appendage) torsion at the time of diagnosis of fallopian tube cyst. Irregular menses and hirsutism was found in 52.6% of the patients, among whom 36.8% were obese 3. When the patients were compared on the basis of their body mass index, the size of fallopian tube cysts was significantly larger in the overweight/obese group. The author concluded by suggesting a possible correlation exists between obesity, fallopian tube cyst size, and hyperandrogenism 3.
When a fallopian tube cyst undergoes torsion, however, it is very difficult to diagnose by ultrasonography 7. When ultrasonographic diagnosis of these cysts is not always feasible, and surgeons usually use laparoscopy to diagnose the cyst. Your doctor will make a small opening in your belly and insert a special telescope or laparoscope to look at your uterus and fallopian tubes. Endocysticendoscopic visualization is a simple, valuable step prior to cystectomy. Bipolar coagulation or extraction of these cysts diagnosed at laparoscopy is easy, not time-consuming, and should be routinely performed in all cases following basic laparoscopic microsurgical principles.
The treatment of complicated fallopian tube cysts is excision, either by laparotomy or by laparoscopy 8. Although excision of silent fallopian tube cysts has not been recommended by some authors 9, prophylactic fallopian tube cyst excision is a common practice in the published literature to prevent possible complications 10, 8. These complications include hemorrhage, perforation, tubal-ovarian torsion, recurrence of torsion and ectopic pregnancy 11. Furthermore, it has been
reported that paratubal cysts can transform into malignancies (2% to 3%) 12, 13, 14. The high tubal-ovarian torsion rate caused by fallopian tube cycts of up to 45% in the literature 12 suggests that prophylactic excision is beneficial before complications occur. Theefore due to the significant effect of fallopian tube cystectomy on tubal patency supports the concept of routine removal of any fallopian tube or paraovarian cyst discovered at laparoscopy. An additional value of removal of these cysts detected at laparoscopy is the exclusion of the rare possibility of malignancy (2% to 3%) and obtaining sufficient tissues for histopathologic evaluation.
Blocked fallopian tubes
Blocked fallopian tubes is usually caused by an old infection in the fallopian tubes, sometimes a sexually transmitted infection. Other causes include previous surgery (particularly surgeries on the tube), severe adhesions of your pelvis, endometriosis, or other sources of infection such as appendicitis 15.
Damage and blockage of the end portion of a fallopian tube can cause it to become filled with fluid; the swollen and fluid-filled tube is called a hydrosalpinx. A normal pregnancy in the uterus may not occur because the tube may be severely damaged and blocked and not work properly. A pregnancy may develop in the tube (ectopic pregnancy) which can be life-threatening. In addition, as the tube is blocked, secretions that collect in the tube may backflow into the uterus and prevent a pregnancy from implanting into the uterus.
What are the symptoms of blocked fallopian tubes ?
Most women do not have any symptoms other than fertility problems. In some women, an ectopic pregnancy may be the first sign of a problem with their tubes. Occasionally, some women may complain of regular or constant pain in their pelvis or lower belly, which may get worse during and after their period. A vaginal discharge can also be associated with this condition.
How is blocked fallopian tubes diagnosed ?
There are three ways to check if one or both of your tubes are blocked and if you have hydrosalpinx; one or more may be performed:
Hysterosalpingogram (HSG) (x-ray)
The doctor will inject a special liquid that shows up on an x-ray into your uterus through your cervix (neck of the womb) and then take an x-ray (called a hysterosalpingogram or HSG) to see where the liquid goes. If your fallopian tubes are open, the liquid will flow out of the ends of the tube into the pelvic cavity. If the tubes are blocked, the liquid will be trapped and your doctor will be able to tell that you have a hydrosalpinx. However, this test can sometimes falsely suggest that the tubes are blocked and sometimes results can be difficult to interpret.
Ultrasound
Your doctor may be able to detect the presence of a hydrosalpinx on ultrasound. If your tube appears enlarged on ultrasound, this usually indicates a more severe hydrosalpinx.
Surgery (laparoscopy)
Your doctor will make a small opening in your belly and insert a special telescope or laparoscope to look at your uterus and fallopian tubes. During this surgery, the doctor can look at your fallopian tubes to see if they are blocked. Usually the doctor inserts a dye through the cervix into the uterus and fallopian tubes to confirm that the dye passes through the ends of the tubes. For more information about laparoscopy, see the ASRM booklet titled Laparoscopy and Hysteroscopy.
If you have hydrosalpinx, can you still have a baby ?
If your fallopian tubes are completely blocked, an egg cannot travel through them to your womb. You will need to be treated by a fertility specialist to become pregnant. Your doctor may occasionally be able to open the tubes with surgery.
If there is too much damage to the tube(s), you will need treatment that does not involve the tubes to help you get pregnant, such as in vitro fertilization (IVF). In this procedure, your egg is joined (fertilized) with sperm in the laboratory. Then the doctor places the fertilized eggs (embryos) into your womb. Your doctor may recommend that your hydrosalpinx is removed or separated from the womb before you start IVF treatment, as the hydrosalpinx may lower your chance of pregnancy.
You have only one fallopian tube. Is it possible to get pregnant ?
You may still be able to get pregnant with only one fallopian tube if:
- You have at least one functioning ovary
- You have monthly menstrual cycles (ovulate)
- Your remaining fallopian tube is healthy
If you’re unable to get pregnant after a year of trying to conceive, see your gynecologist or a reproductive endocrinologist for evaluation.
Fallopian tube pregnancy
The uterus, or womb, is the place where a baby grows when a woman is pregnant. Pregnancy begins with a fertilized egg. Normally, the fertilized egg attaches itself to the lining of the uterus. If you have an ectopic pregnancy, the fertilized egg grows in the wrong place, outside the uterus, usually in the fallopian tubes –fallopian tube pregnancy. The result is usually a miscarriage 16.
An ectopic pregnancy occurs when a fertilized egg implants somewhere other than the main cavity of the uterus – in this case the fallopian tube.
An ectopic pregnancy most often occurs in one of the fallopian tubes that carry eggs from the ovaries to the uterus. This type of ectopic pregnancy is known as a tubal pregnancy. In some cases, however, an ectopic pregnancy occurs in the abdominal cavity, ovary or neck of the uterus (cervix).
To date, the incidence of ectopic pregnancy has increased from 0.5% in 1970 to an estimated 20 in every 1,000 pregnancies are ectopic 17. Approximately 98% of ectopic pregnancies occur in the fallopian tube 18.
An ectopic pregnancy can’t proceed normally. The fertilized egg can’t survive, and the growing tissue might destroy various maternal structures. Left untreated, life-threatening blood loss is possible 19.
Ectopic pregnancy can be a medical emergency if it ruptures, in fact, it is the leading cause of maternal death in early pregnancy 20.
Signs of ectopic pregnancy include:
- Abdominal pain
- Shoulder pain
- Vaginal bleeding
- Feeling dizzy or faint
Early treatment of an ectopic pregnancy can help preserve the chance for future healthy pregnancies.
Symptoms of ectopic pregnancy
At first, an ectopic pregnancy might not cause any signs or symptoms. In other cases, early signs and symptoms of an ectopic pregnancy might be the same as those of any pregnancy — a missed period, breast tenderness and nausea.
If you take a pregnancy test, the result will be positive. Still, an ectopic pregnancy can’t continue as normal.
Light vaginal bleeding with abdominal or pelvic pain is often the first warning sign of an ectopic pregnancy. If blood leaks from the fallopian tube, it’s also possible to feel shoulder pain or an urge to have a bowel movement — depending on where the blood pools or which nerves are irritated. If the fallopian tube ruptures, heavy bleeding inside the abdomen is likely — followed by lightheadedness, fainting and shock.
Seek emergency medical help if you experience any signs or symptoms of an ectopic pregnancy, including:
- Severe abdominal or pelvic pain accompanied by vaginal bleeding
- Extreme lightheadedness or fainting
- Shoulder pain.
Causes of ectopic pregnancy
A fallopian tube pregnancy — the most common type of ectopic pregnancy — happens when a fertilized egg gets stuck on its way to the uterus, often because the fallopian tube is damaged by inflammation or is misshapen. Hormonal imbalances or abnormal development of the fertilized egg also might play a role.
Various factors are associated with ectopic pregnancy, including:
- Previous ectopic pregnancy. If you’ve had one ectopic pregnancy, you’re more likely to have another.
- Inflammation or infection. Inflammation of the fallopian tube (salpingitis) or an infection of the uterus, fallopian tubes or ovaries (pelvic inflammatory disease) increases the risk of ectopic pregnancy. Often, these infections are caused by gonorrhea or chlamydia.
- Fertility issues. Some research suggests an association between difficulties with fertility — as well as use of fertility drugs — and ectopic pregnancy.
- Structural concerns. An ectopic pregnancy is more likely if you have an unusually shaped fallopian tube or the fallopian tube was damaged, possibly during surgery. Even surgery to reconstruct the fallopian tube can increase the risk of ectopic pregnancy.
- Contraceptive choice. Pregnancy when using an intrauterine device (IUD) is rare. If pregnancy occurs, however, it’s more likely to be ectopic. The same goes for pregnancy after tubal ligation — a permanent method of birth control commonly known as “having your tubes tied.” Although pregnancy after tubal ligation is rare, if it happens, it’s more likely to be ectopic.
- Smoking. Cigarette smoking just before you get pregnant can increase the risk of an ectopic pregnancy. And the more you smoke, the greater the risk.
Complications of ectopic pregnancy
When you have an ectopic pregnancy, the stakes are high. Without treatment, a ruptured fallopian tube could lead to life-threatening bleeding.
Seek emergency medical help if you develop any signs or symptoms of an ectopic pregnancy, including:
- Severe abdominal or pelvic pain accompanied by vaginal bleeding
- Extreme lightheadedness or fainting
If you have possible signs or symptoms of an ectopic pregnancy — such as light vaginal bleeding or abdominal pain — see your doctor. He or she might recommend an office visit or immediate medical care.
How is ectopic pregnancy diagnosed ?
If your doctor suspects an ectopic pregnancy, he or she might do a pelvic exam to check for pain, tenderness, or a mass in the fallopian tube or ovary. A physical exam alone usually isn’t enough to diagnose an ectopic pregnancy, however. The diagnosis is typically confirmed with blood tests and imaging studies, such as an ultrasound.
With a standard ultrasound, high-frequency sound waves are directed at the tissues in the abdominal area. During early pregnancy, however, the uterus and fallopian tubes are closer to the vagina than to the abdominal surface. The ultrasound will likely be done using a wandlike device placed in your vagina (transvaginal ultrasound).
Sometimes it’s too soon to detect a pregnancy through ultrasound. If the diagnosis is in question, your doctor might monitor your condition with blood tests until the ectopic pregnancy can be confirmed or ruled out through ultrasound — usually by four to five weeks after conception.
In an emergency situation — if you’re bleeding heavily, for example — an ectopic pregnancy might be diagnosed and treated surgically.
How is ectopic pregnancy treated
A fertilized egg can’t develop normally outside the uterus.
- To prevent life-threatening complications, the ectopic tissue needs to be removed.
If the ectopic pregnancy is detected early, an injection of the drug methotrexate is sometimes used to stop cell growth and dissolve existing cells. It’s imperative that the diagnosis of ectopic pregnancy is certain before this treatment is undertaken.
After the injection, your doctor will monitor your blood for the pregnancy hormone human chorionic gonadotropin (HCG). If the HCG level remains high, you might need another injection of methotrexate.
In other cases, ectopic pregnancy is usually treated with laparoscopic surgery. In this procedure, a small incision is made in the abdomen, near or in the navel. Then your doctor uses a thin tube equipped with a camera lens and light (laparoscope) to view the area.
Other instruments can be inserted into the tube or through other small incisions to remove the ectopic tissue and repair the fallopian tube. If the fallopian tube is significantly damaged, it might need to be removed.
If the ectopic pregnancy is causing heavy bleeding or the fallopian tube has ruptured, you might need emergency surgery through an abdominal incision (laparotomy). In some cases, the fallopian tube can be repaired. Typically, however, a ruptured tube must be removed.
Your doctor will monitor your HCG levels after surgery to be sure all of the ectopic tissue was removed. If HCG levels don’t come down quickly, an injection of methotrexate may be needed.
Prevention of ectopic pregnancy
You can’t prevent an ectopic pregnancy, but you can decrease certain risk factors. For example, limit your number of sexual partners and use a condom when you have sex to help prevent sexually transmitted infections and reduce the risk of pelvic inflammatory disease. Quitting smoking before you attempt to get pregnant may also reduce your risk.
Fallopian tube removal
The surgical removal of the fallopian tube, also called salpingectomy is largely used in case of fallopian tube pregnancy (ectopic pregnancy) and hydrosalpinx in infertile women scheduled for assisted reproductive technologies. Salpingectomy was the standard procedure for ectopic pregnancy 21 until 1978, when laparoscopic salpingotomy was first reported by Bruhat et al. 22.
In clinical practice for ectopic pregnancy, the choice of salpingotomy versus salpingectomy depends on many factors, including patient age, tube condition, serum human chorionic gonadotropin (hCG) levels, and patient’s future fertility desire. The decision of whether to preserve or remove the tube when treating women with tubal pregnancy has been debated for many years, and controversy remains. Previous trials have resulted in different conclusions 23, 24, 25, 26. Whether salpingotomy improves postoperative fertility outcomes compared with salpingectomy remains unclear.
The results of the randomized clinical trials showed that preservation of the tube via salpingotomy did not provide improved fertility 27. This lack of an effect is largely because the transport function of the tube is damaged by the mechanical damage, and the tube is burned by bipolar electric coagulation during the course of the operation. Although the anatomical structure of the tube is preserved, the preserved tube might not be available. In addition, as a result of the operation-induced wound, secretion of cytokines, prostaglandin and leukocyte chemotactic factors by the tubal tissues would exert a negative effect on the reflux in the capillaries and lymphatic system, leading to postoperative tubal adhesion and hydrosalpinx. Consequently, future pregnancies would be affected.
Salpingectomy might decease the ovarian function 28. Most recent studies on this subject have focused only on fertility outcomes after surgery. Furthermore, the effects of salpingectomy on ovarian function have not been taken into account. The blood supply of the ovary originates from both the ovarian artery and the ovarian branch of the uterine artery. These branches of arteries anastomose into nets in the mesosalpinx. Blood circulation is easily damaged during salpingectomy, and the destruction of the ovarian blood supply can lead to ovarian dysfunction 29; however, no information was provided regarding the effect that the two procedures have on ovarian function. The effects of salpingectomy on ovarian function should also be considered when determining the surgical regimen.
One disadvantage of salpingotomy is the increased risk of persistent ectopic pregnancies. A previous study indicated that persistent ectopic pregnancies after salpingotomy occurred in 5% to 20% of cases via laparoscopic surgery and 3% to 5% of cases via laparotomy 30.
According to the recent meta analysis and systematic review, the authors believe that for patients with a healthy contralateral tube operated for tubal pregnancy, the subsequent fertility after salpingectomy and salpingotomy are similar in the long term 27. However, additional multi-center, high quality randomzied clinical trials with large samples are required for further verification. As for the case where both fallopian tubes are blocked with or without hydrosalpinx, further trials aimed at confirming both the positive effects of tubal surgery before assisted reproductive technologies (IVF) and the safety of bilateral salpingectomy are necessary to definitively state when and why unilateral rather than bilateral salpingectomy are recommended 31. This 2010 Cochrane review now provides evidence that laparoscopic tubal occlusion is an alternative to laparoscopic salpingectomy in improving IVF pregnancy rates in women with hydrosalpinges 32.
What is fallopian tube pain ?
Pain in the fallopian tube area (fallopian tube pain) is commonly called pelvic pain – which is pain in the lowest part of your abdomen and pelvis 33.
Fallopian tube pain is a general term used to describe pain that occurs mostly or only in the region below a woman’s belly button. This region includes the lower stomach, lower back, buttocks, and genital area 34.
Fallopian tube pain is chronic if it lasts for more than 6 months and affects a woman’s quality of life 35. This condition is a common reason why women seek medical care 36.
There are many possible causes of pelvic pain, and it may be difficult to figure out the specific cause or causes 34. A woman’s pelvic pain may result from multiple causes occurring all at the same time. In many cases, pelvic pain indicates a problem with one or more of the organs in the pelvic area, such as the uterus, vagina, intestine, bladder or from musculoskeletal sources. Problems may include infection, inflammation, or conditions such as endometriosis. And a woman with one chronic pain condition is at increased risk for other types of chronic pain.
The intensity of a woman’s pelvic pain may not relate to the severity of the problem or condition causing the pain 37. For example, a woman with only small areas of endometriosis may experience intense pain.
How many women have pelvic pain ?
Researchers are not sure exactly how many women in the United States have chronic pelvic pain.
Because it is often linked to other disorders, such as endometriosis or vulvodynia, chronic pelvic pain may be misdiagnosed as another condition, making it difficult to estimate reliable prevalence rates for pelvic pain 38. According to one study, about 15% of women of childbearing age in the United States reported having pelvic pain that lasted at least 6 months 39. Worldwide, the rates of chronic pelvic pain for women of childbearing age range from 14% to 32% 39. Between 13% and 32% of these women have pain that is severe enough to cause them to miss work 40.
How Do Doctors Narrow the Diagnosis to Focus on Important Common Conditions of Fallopian Tube Pain ?
Doctors usually categorize female patients presenting with uterus pain (acute pelvic pain) according to age (e.g., is she in her reproductive years or postmenopausal ?). If the patient is of reproductive age, possible diagnoses are next categorized by whether she is pregnant or attempting pregnancy.
Non-pregnant Reproductive Aged-Women
The typical diagnoses made in nonpregnant reproductive-aged women who present with acute pelvic pain include the following (from most to least common) 41, 42:
- Idiopathic (unknown cause or origin) pelvic pain,
- Pelvic inflammatory disease. Pelvic inflammatory disease is an infection and inflammation of the uterus, ovaries, and other female reproductive organs. It is usually caused by sexually transmitted infections e.g. gonorrhea and chlamydia
- Acute appendicitis,
- Conditions related to ovarian cysts, and
- Endometriosis.
Other less common causes of acute pelvic pain in this population are listed in Table 1 below.
Table 1. Conditions Causing Acute Pelvic Pain in Different Populations
| Patient category | Common diagnoses | Less common diagnoses | Rare diagnoses |
|---|---|---|---|
Reproductive age (not pregnant) | Endometriosis (ruptured endometrioma) | Adenomyosis | Endosalpingiosis |
Idiopathic (no cause identified) | Dysmenorrhea | Round ligament mass (lipoma, teratoma) | |
Ovarian cyst, ruptured | Endometritis (postprocedure) | Transverse vaginal septum | |
Ovarian torsion | Imperforate hymen | ||
PID, tubo-ovarian abscess | Intrauterine device perforation | ||
Leiomyoma (degenerating) | |||
Mittelschmerz | |||
Reproductive age (pregnancy related) | Corpus luteum cyst | Leiomyoma (degenerating) | Incarcerated gravid uterus |
Ectopic pregnancy | Pubic symphysis separation | Ovarian vein thrombosis | |
Endometritis (postpartum) | Subchorionic hemorrhage | PID (rare after first trimester) | |
Normal labor | Uterine rupture | ||
Ovarian torsion | |||
PID (first trimester) | |||
Placental abruption | |||
Preterm labor | |||
Spontaneous abortion | |||
Reproductive age (undergoing fertility treatment) | Ectopic pregnancy | — | Heterotopic pregnancy |
Ovarian follicular cyst | |||
Ovarian hyperstimulation syndrome | |||
Ovarian torsion | |||
Postmenopausal | Malignancy | Ischemic colitis | Endometriosis |
PID, tubo-ovarian abscess | |||
Retained intrauterine device | |||
All groups | Appendicitis | Bowel obstruction | Mesenteric adenitis |
Diverticulitis | Inguinal hernia | ||
Inflammatory bowel disease | Interstitial cystitis | ||
Irritable bowel syndrome | Pelvic adhesive disease (postoperative scarring) | ||
Musculoskeletal (abdominal wall) pain | Perirectal abscess | ||
Urinary tract infection | Urethral diverticulum | ||
Urolithiasis | Urinary retention |
Figure 4. An algorithm for using physical examination findings to evaluate selected common causes of pelvic pain in non-pregnant women
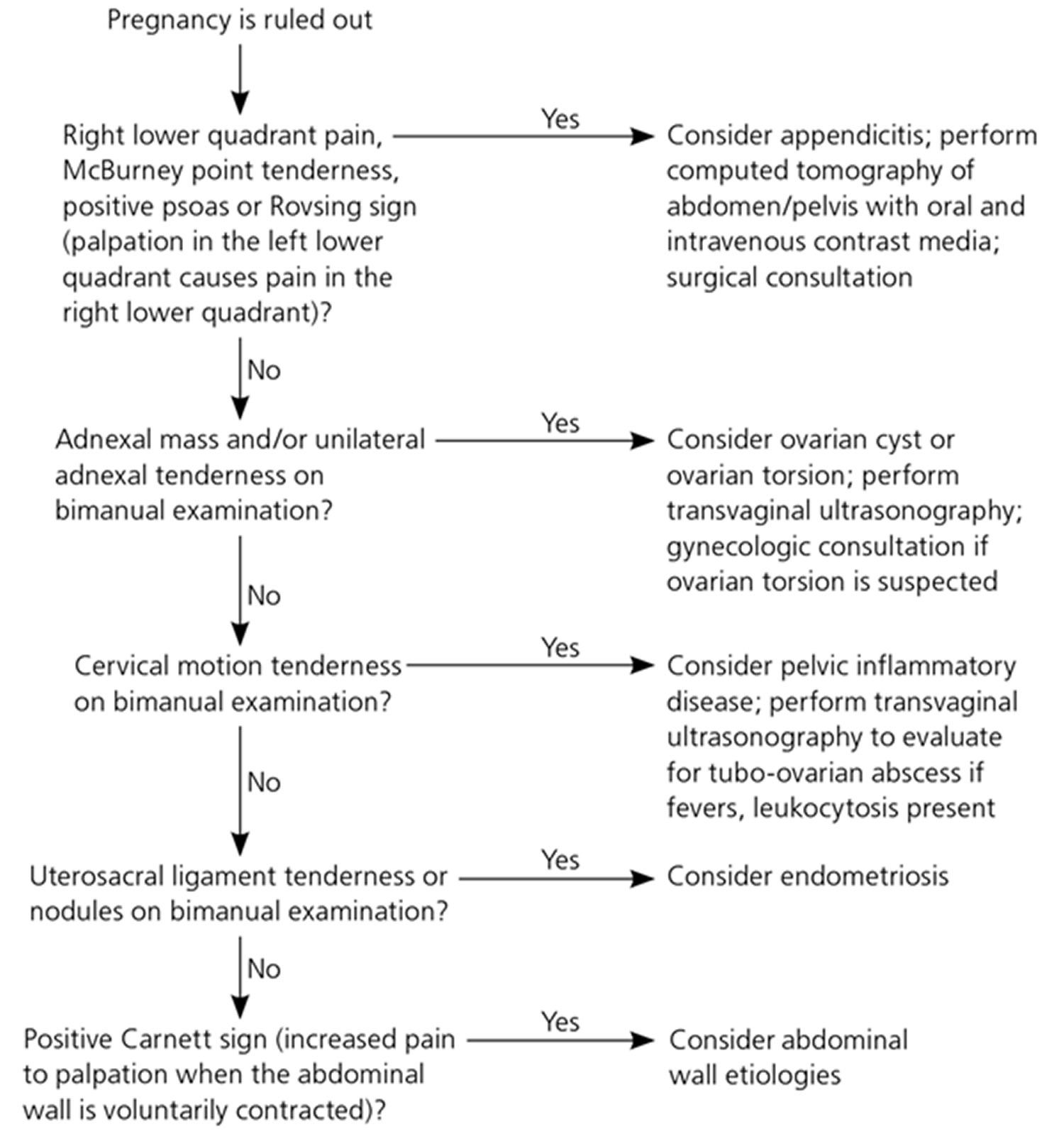
Note: McBurney’s point = is the name given to the point over the right side of the abdomen that is one-third of the distance from the anterior superior iliac spine to the umbilicus (navel), this point roughly corresponds to the most common location of the base of the appendix where it is attached to the cecum, deep tenderness at McBurney’s point, known as McBurney’s sign, is a sign of acute appendicitis; Rovsing sign = is a sign of appendicitis, if palpation of the left lower quadrant of a person’s abdomen increases the pain felt in the right lower quadrant, the patient is said to have a positive Rovsing’s sign and may have appendicitis; Positive Carnett’s sign = is a finding on clinical examination in which (acute) abdominal pain remains unchanged or increases when the muscles of the abdominal wall are tensed. A positive test increases the likelihood that the abdominal wall and not the abdominal cavity is the source of the pain (for example, due to rectus sheath hematoma instead of appendicitis).
[Source 43]Reproductive-aged women who are pregnant or attempting pregnancy
Women who are pregnant or attempting pregnancy through fertility treatments have other common causes of acute pelvic pain (Table 1). For pregnant women, the clinical scenario narrows the list of possible causes, but it is critical to detect serious or life-threatening conditions, such as ectopic pregnancy in the early weeks of pregnancy and placental abruption in the later stages of pregnancy. Nongynecologic conditions, such as appendicitis, can also occur during pregnancy.
Women undergoing infertility treatments through ovarian stimulation or in vitro fertilization have unique risks, such as ovarian hyperstimulation syndrome (i.e., ovarian enlargement with multiple ovarian cysts and leakage of fluid from the ovary into the abdominal/pelvic space), ovarian torsion, and heterotopic pregnancy (i.e., simultaneous ectopic and intrauterine pregnancy) 44.
Postmenopausal women
Cancer must be primarily considered in a postmenopausal woman with acute pelvic pain. Other rare causes are postmenopausal endometriosis 45 and, on occasion, a retained intrauterine device that the patient had forgotten to remove 46.
Which History and Physical Examination Findings Are Most Helpful in Evaluating Acute Pelvic Pain ?
Several findings in the patient’s history can help guide laboratory testing and imaging to confirm a suspected diagnosis (Table 2). Relevant historical features include whether the patient is sexually active (e.g., pain related to a complication of pregnancy), specific symptoms (e.g., description of the pain; changes in menstrual pattern; the presence of vaginal discharge, dysuria, hematuria, or nausea), and whether she has had intra-uterine instrumentation or pelvic surgery. A recent study reported rare occurrences of pelvic inflammatory disease in women who denied ever having sexual intercourse, illustrating the need for a high index of suspicion for this condition 47.
Table 2. History and Physical Examination Findings Are Most Helpful in Evaluating Acute Pelvic Pain
| Finding | Suggested diagnoses | Further diagnostic considerations | |
|---|---|---|---|
History of intrauterine instrumentation, multiple cesarean deliveries, or other uterine surgeries | Adenomyosis (endometrial tissue grown into the uterine wall) | Magnetic resonance imaging | |
Pelvic adhesions | Consider nonurgent referral to gynecologist or general surgeon in absence of other findings | ||
Menstrual abnormalities | |||
Amenorrhea (abnormal absence of menstruation) | Imperforate hymen | Pelvic examination | |
Transverse vaginal septum | Pelvic ultrasonography | ||
Dysmenorrhea (painful menstruation) | Endometriosis, ovarian cyst | Pelvic ultrasonography (to assess for ovarian cyst) | |
Nausea and vomiting | Appendicitis, ovarian torsion | If appendicitis is more likely: proceed with contrast CT | |
If ovarian torsion is more likely: proceed with pelvic ultrasonography with Doppler flow study | |||
Early urgent referral for surgical evaluation and treatment is recommended | |||
Pain symptoms | |||
Bilateral pain, particularly if associated with mucopurulent vaginal discharge | Pelvic inflammatory disease | Testing for sexually transmitted infections | |
Complete blood count to test for leukocytosis or left shift | |||
Dull, unilateral adnexal pain that is constant or intermittent | Ovarian torsion | Presence of risk factors (nausea, vomiting, pregnancy) | |
Pelvic ultrasonography with Doppler flow study | |||
Consider urgent referral for surgical evaluation and treatment | |||
Right lower quadrant pain | Acute appendicitis | Complete blood count demonstrating leukocytosis | |
Contrast CT of the abdomen and pelvis | |||
Ectopic pregnancy | Qualitative urine β-hCG can detect a pregnancy at four weeks’ gestation | ||
Quantitative serum β-hCG can determine if pregnancy is above the discriminatory level such that an intrauterine pregnancy should be visible on pelvic ultrasonography to rule out ectopic pregnancy | |||
Blood type to determine Rh status; if bleeding and pregnant, will need Rho(D) immune globulin (RhoGam) | |||
Pelvic ultrasonography | |||
Ovarian torsion | Pelvic ultrasonography with Doppler flow study | ||
Sexually active; pregnancy possible | Ectopic pregnancy, spontaneous abortion | Qualitative urine β-hCG can detect a pregnancy at four weeks’ gestation | |
Quantitative serum β-hCG can determine if pregnancy is above the discriminatory level such that an intrauterine pregnancy should be visible on pelvic ultrasonography to rule out ectopic pregnancy | |||
Blood type to determine Rh status; if bleeding and pregnant, will need Rho(D) immune globulin | |||
Pelvic ultrasonography | |||
Urinary symptoms | |||
Dysuria (painful or difficult urination) | Urinary tract infection | Urinalysis demonstrating white blood cells, bacteria, leukocyte esterase, or nitrites | |
Gross hematuria (blood in urine) | Urolithiasis (urinary bladder or urinary tract stone) | Abdominal ultrasonography | |
β-hCG = beta human chorionic gonadotropin; CT = computed tomography.
Fallopian tube cancer
Fallopian tube cancer is a disease in which malignant (cancer) cells form in the tissue covering the lining the fallopian tube 48.
The fallopian tubes are a pair of long, slender tubes, one on each side of the uterus. Eggs pass from the ovaries, through the fallopian tubes, to the uterus. Cancer sometimes begins at the end of the fallopian tube near the ovary and spreads to the ovary.
Fallopian tube cancer, ovarian epithelial cancer and primary peritoneal cancer form in the same type of tissue and are treated the same way 48. The hypothesis that many high-grade serous ovarian cancers (the most common histologic subtype) may arise from precursor lesions that originate in the fimbriae of the fallopian tubes has been supported by findings from risk-reducing surgeries in healthy women with BRCA1 or BRCA2 mutations 49. In addition, histologically similar cancers diagnosed as primary peritoneal carcinomas share molecular findings, such as loss or inactivation of the tumor-suppressor p53 and BRCA1 or BRCA2 proteins 50. Therefore, high-grade serous adenocarcinomas arising from the fallopian tube and elsewhere in the peritoneal cavity, together with most ovarian epithelial cancers, represent extrauterine adenocarcinomas of Müllerian epithelial origin and are staged and treated similarly to ovarian cancer 48. Regardless of the site of origin, the hallmark of these cancers is their early peritoneal spread of metastases. Since 2000, fallopian tube cancer and primary peritoneal cancer have usually been included in ovarian cancer clinical trials 51.
Clear cell and endometrioid ovarian cancers that are linked to endometriosis have different gene-expression signatures, as do mucinous subtypes 50.
Stromal and germ cell tumors are relatively uncommon and comprise fewer than 10% of cases.
Incidence and Mortality
Epithelial carcinoma of the ovary is one of the most common gynecologic malignancies, with 50% of all cases occurring in women older than 65 years. It is the fifth most frequent cause of cancer death in women 52.
Estimated new cases and deaths from ovarian cancer in the United States in 2017 53:
- New cases: 22,440.
- Deaths: 14,080.
Risk Factors
Family history and genetic alterations
Women who have a family history of ovarian cancer are at an increased risk of ovarian cancer.
Anything that increases your risk of getting a disease is called a risk factor. Having a risk factor does not mean that you will get cancer; not having risk factors doesn’t mean that you will not get cancer. Talk with your doctor if you think you may be at risk.
The most important risk factor for ovarian cancer is a history of ovarian cancer in a first-degree relative (mother, daughter, or sister). This risk is higher in women who have one first-degree relative and one second-degree relative (grandmother or aunt) with a history of ovarian cancer. This risk is even higher in women who have two or more first-degree relatives with a history of ovarian cancer. Approximately 20% of ovarian cancers are familial, and although most of these are linked to mutations in either the BRCA1 or BRCA2 gene, several other genes have been implicated 54. The risk is highest in women who have two or more first-degree relatives with ovarian cancer 55. The risk is somewhat less for women who have one first-degree relative and one second-degree relative (grandmother or aunt) with ovarian cancer.
Some ovarian, fallopian tube, and primary peritoneal cancers are caused by inherited gene mutations (changes).
The genes in cells carry the hereditary information that is received from a person’s parents. Hereditary ovarian cancer makes up about 20% of all cases of ovarian cancer. There are three hereditary patterns: ovarian cancer alone, ovarian and breast cancers, and ovarian and colon cancers.
In most families affected with breast and ovarian cancer syndrome or site-specific ovarian cancer, genetic linkage to the BRCA1 locus on chromosome 17q21 has been identified 56. BRCA2, also responsible for some instances of inherited ovarian and breast cancer, has been mapped by genetic linkage to chromosome 13q12 57.
The lifetime risk for developing ovarian cancer in patients harboring germline mutations in BRCA1 is substantially increased over that of the general population 58. Two retrospective studies of patients with germline mutations in BRCA1 suggest that the women in these studies have improved survival compared with BRCA1 mutation–negative women 59. Most women with a BRCA1 mutation probably have family members with a history of ovarian and/or breast cancer; therefore, the women in these studies may have been more vigilant and inclined to participate in cancer screening programs that may have led to earlier detection.
For women at increased risk, prophylactic oophorectomy may be considered after age 35 years if childbearing is complete. In a family-based study among 551 women with BRCA1 or BRCA2 mutations, of the 259 women who had undergone bilateral prophylactic oophorectomy, 2 (0.8%) developed subsequent papillary serous peritoneal carcinoma, and 6 (2.8%) had stage I ovarian cancer at the time of surgery. Of the 292 matched controls, 20% who did not have prophylactic surgery developed ovarian cancer. Prophylactic surgery was associated with a reduction in the risk of ovarian cancer that exceeded 90% (relative risk, 0.04; 95% confidence interval, 0.01–0.16), with an average follow-up of 9 years 60; however, family-based studies may be associated with biases resulting from case selection and other factors that influence the estimate of benefit. After a prophylactic oophorectomy, a small percentage of women may develop a primary peritoneal carcinoma that is similar in appearance to ovarian cancer 61.
There are tests that can detect gene mutations. These genetic tests are sometimes done for members of families with a high risk of cancer. Women with an increased risk of ovarian cancer may consider surgery to lessen the risk.
Some women who have an increased risk of ovarian cancer may choose to have a risk-reducing oophorectomy (the removal of healthy ovaries so that cancer cannot grow in them). In high-risk women, this procedure has been shown to greatly decrease the risk of ovarian cancer.
Signs and symptoms of ovarian, fallopian tube, or peritoneal cancer
Signs and symptoms of ovarian, fallopian tube, or peritoneal cancer include pain or swelling in the abdomen.
Ovarian, fallopian tube, or peritoneal cancer may not cause early signs or symptoms. When signs or symptoms do appear, the cancer is often advanced. Signs and symptoms may include the following:
- Pain, swelling, or a feeling of pressure in the abdomen or pelvis.
- Vaginal bleeding that is heavy or irregular, especially after menopause.
- Vaginal discharge that is clear, white, or colored with blood.
- A lump in the pelvic area.
- Gastrointestinal problems, such as gas, bloating, or constipation.
These signs and symptoms also may be caused by other conditions and not by ovarian, fallopian tube, or peritoneal cancer. If the signs or symptoms get worse or do not go away on their own, check with your doctor so that any problem can be diagnosed and treated as early as possible.
How is fallopian tube, ovarian and peritoneal cancer diagnosed
Tests that examine the ovaries and pelvic area are used to detect (find) and diagnose ovarian, fallopian tube, and peritoneal cancer.
The following tests and procedures may be used:
- Physical exam and history : An exam of the body to check general signs of health, including checking for signs of disease, such as lumps or anything else that seems unusual. A history of the patient’s health habits and past illnesses and treatments will also be taken.
- Pelvic exam : An exam of the vagina, cervix, uterus, fallopian tubes, ovaries, and rectum. A speculum is inserted into the vagina and the doctor or nurse looks at the vagina and cervix for signs of disease. A Pap test of the cervix is usually done. The doctor or nurse also inserts one or two lubricated, gloved fingers of one hand into the vagina and places the other hand over the lower abdomen to feel the size, shape, and position of the uterus and ovaries. The doctor or nurse also inserts a lubricated, gloved finger into the rectum to feel for lumps or abnormal areas.
- CA-125 assay.
- Ultrasound exam (pelvic or transvaginal).
- Computed tomography (CT) scan.
- Positron emission tomography (PET) scan.
- Magnetic resonance imaging (MRI).
- Chest x-ray.
- Biopsy.
CA-125 levels can be elevated in other malignancies and benign gynecologic problems such as endometriosis. CA-125 levels and histology are used to diagnose epithelial ovarian cancer 62.
Prognostic Factors
Prognosis for patients with ovarian cancer is influenced by multiple factors. Multivariate analyses suggest that the most important favorable prognostic factors include the following 63:
- Younger age.
- Good performance status.
- Cell type other than mucinous or clear cell.
- Well-differentiated tumor.
- Early-stage disease.
- Absence of ascites.
- Lower disease volume before surgical debulking.
- Smaller residual tumor after primary cytoreductive surgery.
- BRCA1 or BRCA2 mutation carrier.
For patients with stage I disease, the most important prognostic factor associated with relapse is grade, followed by dense adherence and large-volume ascites 64. Stage I tumors have a high proportion of low-grade serous cancers. These cancers have a derivation distinctly different from that of high-grade serous cancers, which usually present in stages III and IV. Many high-grade serous cancers originate in the fallopian tube and other areas of extrauterine Müllerian epithelial origin.
If the tumor is grade III, densely adherent, or stage IC, the chance of relapse and death from ovarian cancer is as much as 30% 65.
The use of DNA flow cytometric analysis of tumors from stage I and stage IIA patients may identify a group of high-risk patients 66. Patients with clear cell histology appear to have a worse prognosis 67. Patients with a significant component of transitional cell carcinoma appear to have a better prognosis 68.
Case-control studies suggest that BRCA1 and BRCA2 mutation carriers have improved responses to chemotherapy when compared with patients with sporadic epithelial ovarian cancer. This may be the result of a deficient homologous DNA repair mechanism in these tumors, which leads to increased sensitivity to chemotherapy agents 69.
Figure 4. Fallopian tube cancer
Fallopian tube cancer prevention
Should prophylactic surgery be limited to the fallopian tubes or can one or both ovaries be spared in BRCA mutations carriers ?
Several germ-line mutations and copy number variations that harbor increased risk for high-grade serous ovarian carcinomas have been reported, BRCA1 and BRCA2 being the most prevalent ones, accounting for 5% to 10% of the cases with up to 54% lifetime risk 70. Women recognized to have BRCA mutations are currently treated with risk-reducing (prophylactic) excision of the adnexa (bilateral salpingo-oophorectomy). This practice, which targets healthy women, has provided most of the emerging data in the study of early serous carcinomas. Currently there is no data to support or reject such a practice 71. Removing the fallopian tubes with surgery carries an obvious decrease in early menopause–related morbidity and an improvement in quality of life, including preservation of fertility, but evidence supporting this approach is still limited in terms of the number of prospectively accrued cases.
Is there hope for early detection of intraepithelial carcinomas as one strategy to reduce serous cancer mortality ?
One might expect that the practice of more complete fallopian tube examination will uncover more early serous carcinomas and provide more information as to their prognosis. Whether such tumors can be identified by noninvasive (serologic) assays or imaging will not be known until such techniques are developed. Of the few cases of stage 0 (intraepithelial) fallopian tube serous cancers diagnosed prospectively in BRCA-positive women, no recurrences have been documented. However, higher stage fallopian tube cancer is associated with relatively poor prognosis despite adjuvant therapy.107,108 Larger studies devoted to detailed examination of the distal fallopian tube will expand the database of stage 0 fallopian tube serous cancer cases and provide greater insight into the potential value of early detection, the prognosis, and the appropriateness of postoperative therapy in these cases.
Is fimbriectomy a viable alternative to tubal ligation that will significantly reduce serous cancer risk ?
The current data indicates that a significant percentage of serous carcinomas arise from a precursor condition in the distal fallopian tube. It is reasonable to expect that sterilization practices that targeted the fimbria would maximize the protective effect of tubal sterilization on serous cancer prevention. However, the value of such a practice must be evaluated in the context of the safety and feasibility of fimbriectomy and the fact that such a strategy precludes so-called tubal reversal procedures in the future for the patient. Nevertheless, barring the emergence of a successful chemopreventive or early detection algorithm, removing fimbrial tissue is the most obvious, albeit yet unproven, surgical approach to pelvic serous cancer prevention.
- Capacitation of mammalian spermatozoa in vivo, with a specific focus on events in the Fallopian tubes. Hunter RH, Rodriguez-Martinez H. Mol Reprod Dev. 2004 Feb; 67(2):243-50. https://www.ncbi.nlm.nih.gov/pubmed/14694441/[↩]
- Dietrich JE, Heard MJ, Edwards C. Uteroovarian ligament torsion of the due to a paratubal cyst. J Pediatr Adolesc Gynecol 2005; 18: 125-127.[↩]
- Paratubal Cyst Size Correlates With Obesity and Dysregulation of the Wnt Signaling Pathway. J Pediatr Adolesc Gynecol. 2017 Oct;30(5):571-577. doi: 10.1016/j.jpag.2017.04.002. Epub 2017 Apr 27. https://www.ncbi.nlm.nih.gov/pubmed/28456695[↩][↩][↩]
- The incidence and surgical management of paratubal cysts in a pediatric and adolescent population. Muolokwu, Enyioma et al. Journal of Pediatric Surgery , Volume 46 , Issue 11 , 2161 – 2163. http://www.jpedsurg.org/article/S0022-3468(11)00385-X/fulltext[↩][↩]
- Ovarioscopy, a technique to determine the nature of cystic ovarian tumors. Darwish AM, Amin AF, El-Feky MA. J Am Assoc Gynecol Laparosc. 2000 Nov; 7(4):539-44. https://www.ncbi.nlm.nih.gov/pubmed/11044509/[↩]
- Paratubal cysts in young females as an incidental finding in laparotomies performed for right lower quadrant abdominal pain. Vlahakis-Miliaras E, Miliaras D, Koutsoumis G, Miliaras S, Spyridakis I, Papadopoulos MS. Pediatr Surg Int. 1998 Mar; 13(2-3):141-2. https://www.ncbi.nlm.nih.gov/pubmed/9563027/[↩]
- Said MR, Bamigboye V. Twisted paraovarian cyst in a young girl. J Obstet Gynaecol 2008; 28: 549-550.[↩]
- Thakore SS, Chun MJ, Fitzpatrick K. Recurrent ovarian torsion due to paratubal cysts in an adolescent female. J Pediatr Adolesc Gynecol 2012; 25: e85-e87.[↩][↩]
- Muthucumaru M, Yahya Z, Ferguson P, Cheng W. Torsion of hydatids of Morgagni in premenarchal adolescent girls–a case report and review of literature. J Pediatr Surg 2011; 46: e13-e5.[↩]
- Okada T, Yoshida H, Matsunaga T, et al. Paraovarian cyst with torsion in children. J Pediatr Surg 2002; 37: 937-940.[↩]
- Seshadri S, Morris A, Uchil D, Joloaso A. Bilateral paratubal cysts with co-existent fallopian tube torsion in an adolescent. J Obstet Gynaecol 2009; 29: 564-565.[↩]
- Kiseli M, Caglar GS, Cengiz SD, Karadag D, Yılmaz MB. Clinical diagnosis and complications of paratubal cysts: review of the literature and report of uncommon presentations. Arch Gynecol Obstet 2012; 285: 1563-1569.[↩][↩]
- Muolokwu E, Sanchez J, Bercaw JL, et al. The incidence and surgical management of paratubal cysts in a pediatric and adolescent population. J Pediatr Surg 2011;46: 2161-2163.[↩]
- Lee S, Ahn KH, Park HT, et al. Paratubal Borderline Malignancy: ACase of a 17-Year-Old Adolescent Female Treated with Laparo-Endoscopic Single-Site Surgery and a Review of the Literature. J Pediatr Adolesc Gynecol 2016; 29: 74-76.[↩]
- Hydrosalpinx. American Society for Reproductive Medicine. http://www.reproductivefacts.org/news-and-publications/patient-fact-sheets-and-booklets/fact-sheets-and-info-booklets/hydrosalpinx/[↩]
- Ectopic Pregnancy. Medline Plus. https://medlineplus.gov/ectopicpregnancy.html[↩]
- Clinical diagnosis and treatment of ectopic pregnancy. Alkatout I, Honemeyer U, Strauss A, Tinelli A, Malvasi A, Jonat W, Mettler L, Schollmeyer T. Obstet Gynecol Surv. 2013 Aug; 68(8):571-81. https://www.ncbi.nlm.nih.gov/pubmed/23921671/[↩]
- Sites of ectopic pregnancy: a 10 year population-based study of 1800 cases. Bouyer J, Coste J, Fernandez H, Pouly JL, Job-Spira N. Hum Reprod. 2002 Dec; 17(12):3224-30. https://www.ncbi.nlm.nih.gov/pubmed/12456628/[↩]
- Ectopic pregnancy. Mayo Clinic. http://www.mayoclinic.org/diseases-conditions/ectopic-pregnancy/basics/definition/con-20024262[↩]
- Surgical treatment of ectopic pregnancy. Agdi M, Tulandi T. Best Pract Res Clin Obstet Gynaecol. 2009 Aug; 23(4):519-27. https://www.ncbi.nlm.nih.gov/pubmed/19231293/[↩]
- Hajenius PJ, Mol F, Mol BWJ, Bossuyt PMM, Ankum WM, Van der Veen F. Interventions for tubal ectopic pregnancy. Cochrane Database of Systematic Reviews 2007, Issue 1. Art. No.: CD000324. DOI: 10.1002/14651858.CD000324.pub2.[↩]
- Endoscopic treatment of ectopic pregnancies. Bruhat MA, Pouly JL. Curr Opin Obstet Gynecol. 1993 Apr; 5(2):260-6. https://www.ncbi.nlm.nih.gov/pubmed/8490098/[↩]
- Mol F, van Mello NM, Strandell A, Strandell K, Jurkovic D, Ross J, et al. Salpingotomy versus salpingectomy in women with tubal pregnancy (European surgery in ectopic pregnancy study): An open-label, multicenter, randomized controlled trial. Lancet. 2014; 383:1483–1489. http://www.thelancet.com/journals/lancet/article/PIIS0140-6736(14)60123-9/fulltext[↩]
- Clausen I. Conservative versus radical surgery for tubal pregnancy. A review. Acta Obstet Gynecol Scand.1996; 75(1):8–12. https://www.ncbi.nlm.nih.gov/pubmed/8561006[↩]
- Clausen I. Conservative versus radical surgery for tubal pregnancy. A review. Acta Obstet Gynecol Scand.1996; 75(1):8–12. http://www.fertstert.org/article/S0015-0282(97)80064-7/pdf[↩]
- Bangsgaard N, Lund CO, Ottesen B, Nilas L. Improved fertility following conservative surgical treatment of ectopic pregnancy. BJOG. 2003; 110:765–770. https://www.ncbi.nlm.nih.gov/pubmed/12892689[↩]
- Cheng X, Tian X, Yan Z, et al. Comparison of the Fertility Outcome of Salpingotomy and Salpingectomy in Women with Tubal Pregnancy: A Systematic Review and Meta-Analysis. Li D, ed. PLoS ONE. 2016;11(3):e0152343. doi:10.1371/journal.pone.0152343. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4807767/[↩][↩]
- Anti-Müllerian hormone levels in salpingectomized compared with nonsalpingectomized women with tubal factor infertility and women with unexplained infertility. Grynnerup AG, Lindhard A, Sørensen S. Acta Obstet Gynecol Scand. 2013 Nov; 92(11):1297-303. https://www.ncbi.nlm.nih.gov/pubmed/24032634/[↩]
- Total salpingectomy during abdominal hysterectomy: effects on ovarian reserve and ovarian stromal blood flow. Sezik M, Ozkaya O, Demir F, Sezik HT, Kaya H. J Obstet Gynaecol Res. 2007 Dec; 33(6):863-9. https://www.ncbi.nlm.nih.gov/pubmed/18001455/[↩]
- Ben Arie A, Goldchmit R, Dgani R, Hazan Y, Ben-Hur H, Open M. Trophoblastic peritoneal implants after laparoscopic treatment of ectopic pregnancy. Eur J Obstet Gynecol Reprod Biol. 2001; 96(1):113–115. https://www.ncbi.nlm.nih.gov/pubmed/11311773[↩]
- Noventa M, Gizzo S, Saccardi C, et al. Salpingectomy before assisted reproductive technologies: a systematic literature review. Journal of Ovarian Research. 2016;9:74. doi:10.1186/s13048-016-0284-1. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5094031/[↩]
- Johnson N, van Voorst S, Sowter MC, Strandell A, Mol BWJ. Surgical treatment for tubal disease in women due to undergo in vitro fertilisation. Cochrane Database of Systematic Reviews 2010, Issue 1. Art. No.: CD002125. DOI: 10.1002/14651858.CD002125.pub3.[↩]
- Pelvic pain. Mayo Clinic. https://www.mayoclinic.org/symptoms/pelvic-pain/basics/definition/sym-20050898[↩]
- American College of Obstetricians and Gynecologists. (2004). ACOG practice bulletin no. 51. Chronic pelvic pain. Obstetrics & Gynecology, 103, 589–605.[↩][↩]
- UCSF Medical Center. (n.d.). Pelvic pain. https://www.ucsfhealth.org/conditions/pelvic_pain/[↩]
- Reiter, R. C. (1996). A profile of women with chronic pelvic pain. Clinical Obstetrics and Gynecology, 33, 130–136.[↩]
- What causes pelvic pain ? National Institute of Child Health and Human Development. https://www.nichd.nih.gov/health/topics/pelvicpain/conditioninfo/Pages/causes.aspx[↩]
- Andrews J, Yunker A, Reynolds WS, et al. Noncyclic Chronic Pelvic Pain Therapies for Women: Comparative Effectiveness [Internet]. Rockville (MD): Agency for Healthcare Research and Quality (US); 2012 Jan. (Comparative Effectiveness Reviews, No. 41.) Available from: https://www.ncbi.nlm.nih.gov/books/NBK84586/[↩]
- Mathias, S. D., Kuppermann, M., Liberman, R. F., Lipschutz, R. C., & Steege, J. F. (1996). Chronic pelvic pain: Prevalence, health-related quality of life, and economic correlates. Obstetrics & Gynecology, 87, 321–327.[↩][↩]
- Ahangari, A. (2014). Prevalence of chronic pelvic pain among women: An updated review. Pain Physician, 17, E141–E147. http://www.painphysicianjournal.com/current/pdf?article=MjA2NQ%3D%3D&journal=81[↩]
- Yeh JM, Hook EW III, Goldie SJ. A refined estimate of the average lifetime cost of pelvic inflammatory disease. Sex Transm Dis. 2003;30(5):369–378.[↩]
- Morino M, Pellegrino L, Castagna E, Farinella E, Mao P. Acute nonspecific abdominal pain: a randomized, controlled trial comparing early lap-aroscopy versus clinical observation. Ann Surg. 2006;244(6):881–888.[↩]
- Common Questions About the Evaluation of Acute Pelvic Pain. Am Fam Physician. 2016 Jan 1;93(1):41-48A. http://www.aafp.org/afp/2016/0101/p41.html[↩][↩][↩]
- Svare J, Norup P, Grove Thomsen S, et al. Heterotopic pregnancies after in-vitro fertilization and embryo transfer—a Danish survey. Hum Reprod. 1993;8(1):116–118.[↩]
- Oxholm D, Knudsen UB, Kryger-Baggesen N, Ravn P. Postmenopausal endometriosis. Acta Obstet Gynecol Scand. 2007;86(10):1158–1164.[↩]
- Wagner CA, Gimpelson RJ. Postmenopausal bleeding due to a Cu-7 intrauterine device retained for thirty years. JSLS. 2012;16(2):329–332.[↩]
- Cho HW, Koo YJ, Min KJ, Hong JH, Lee JK. Pelvic inflammatory disease in virgin women with tubo-ovarian abscess: a single-center experience and literature review [published online ahead of print August 7, 2015]. J Pediatr Adolesc Gynecol. http://www.sciencedirect.com/science/article/pii/S1083318815002922[↩]
- Ovarian Epithelial, Fallopian Tube, and Primary Peritoneal Cancer Treatment. National Cancer Institute. https://www.cancer.gov/types/ovarian/hp/ovarian-epithelial-treatment-pdq[↩][↩][↩]
- Levanon K, Crum C, Drapkin R: New insights into the pathogenesis of serous ovarian cancer and its clinical impact. J Clin Oncol 26 (32): 5284-93, 2008. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2652087/[↩]
- Birrer MJ: The origin of ovarian cancer—is it getting clearer? N Engl J Med 363 (16): 1574-5, 2010. http://www.nejm.org/doi/full/10.1056/NEJMe1009527[↩][↩]
- Dubeau L, Drapkin R: Coming into focus: the nonovarian origins of ovarian cancer. Ann Oncol 24 (Suppl 8): viii28-viii35, 2013. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3805308/[↩]
- Yancik R: Ovarian cancer. Age contrasts in incidence, histology, disease stage at diagnosis, and mortality. Cancer 71 (2 Suppl): 517-23, 1993. https://www.ncbi.nlm.nih.gov/pubmed/8420671[↩]
- American Cancer Society: Cancer Facts and Figures 2017. Atlanta, Ga: American Cancer Society, 2017. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2017/cancer-facts-and-figures-2017.pdf[↩]
- Pennington KP, Swisher EM: Hereditary ovarian cancer: beyond the usual suspects. Gynecol Oncol 124 (2): 347-53, 2012. https://www.ncbi.nlm.nih.gov/pubmed/22264603[↩]
- Piver MS, Goldberg JM, Tsukada Y, et al.: Characteristics of familial ovarian cancer: a report of the first 1,000 families in the Gilda Radner Familial Ovarian Cancer Registry. Eur J Gynaecol Oncol 17 (3): 169-76, 1996. https://www.ncbi.nlm.nih.gov/pubmed/8780913[↩]
- Steichen-Gersdorf E, Gallion HH, Ford D, et al.: Familial site-specific ovarian cancer is linked to BRCA1 on 17q12-21. Am J Hum Genet 55 (5): 870-5, 1994. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1918337/[↩]
- Wooster R, Neuhausen SL, Mangion J, et al.: Localization of a breast cancer susceptibility gene, BRCA2, to chromosome 13q12-13. Science 265 (5181): 2088-90, 1994. https://www.ncbi.nlm.nih.gov/pubmed/8091231[↩]
- Struewing JP, Hartge P, Wacholder S, et al.: The risk of cancer associated with specific mutations of BRCA1 and BRCA2 among Ashkenazi Jews. N Engl J Med 336 (20): 1401-8, 1997. http://www.nejm.org/doi/full/10.1056/NEJM199705153362001[↩]
- Aida H, Takakuwa K, Nagata H, et al.: Clinical features of ovarian cancer in Japanese women with germ-line mutations of BRCA1. Clin Cancer Res 4 (1): 235-40, 1998. http://clincancerres.aacrjournals.org/content/4/1/235.long[↩]
- Rebbeck TR, Lynch HT, Neuhausen SL, et al.: Prophylactic oophorectomy in carriers of BRCA1 or BRCA2 mutations. N Engl J Med 346 (21): 1616-22, 2002. http://www.nejm.org/doi/full/10.1056/NEJMoa012158[↩]
- Piver MS, Jishi MF, Tsukada Y, et al.: Primary peritoneal carcinoma after prophylactic oophorectomy in women with a family history of ovarian cancer. A report of the Gilda Radner Familial Ovarian Cancer Registry. Cancer 71 (9): 2751-5, 1993. https://www.ncbi.nlm.nih.gov/pubmed/8467455[↩]
- Atack DB, Nisker JA, Allen HH, et al.: CA 125 surveillance and second-look laparotomy in ovarian carcinoma. Am J Obstet Gynecol 154 (2): 287-9, 1986. https://www.ncbi.nlm.nih.gov/pubmed/3456202[↩]
- Thigpen T, Brady MF, Omura GA, et al.: Age as a prognostic factor in ovarian carcinoma. The Gynecologic Oncology Group experience. Cancer 71 (2 Suppl): 606-14, 1993. https://www.ncbi.nlm.nih.gov/pubmed/8420683[↩]
- Dembo AJ, Davy M, Stenwig AE, et al.: Prognostic factors in patients with stage I epithelial ovarian cancer. Obstet Gynecol 75 (2): 263-73, 1990. https://www.ncbi.nlm.nih.gov/pubmed/2300355[↩]
- Kolomainen DF, A’Hern R, Coxon FY, et al.: Can patients with relapsed, previously untreated, stage I epithelial ovarian cancer be successfully treated with salvage therapy? J Clin Oncol 21 (16): 3113-8, 2003. https://www.ncbi.nlm.nih.gov/pubmed/12915602[↩]
- Schueler JA, Cornelisse CJ, Hermans J, et al.: Prognostic factors in well-differentiated early-stage epithelial ovarian cancer. Cancer 71 (3): 787-95, 1993. https://www.ncbi.nlm.nih.gov/pubmed/8431861[↩]
- Young RC, Walton LA, Ellenberg SS, et al.: Adjuvant therapy in stage I and stage II epithelial ovarian cancer. Results of two prospective randomized trials. N Engl J Med 322 (15): 1021-7, 1990. http://www.nejm.org/doi/full/10.1056/NEJM199004123221501[↩]
- Gershenson DM, Silva EG, Mitchell MF, et al.: Transitional cell carcinoma of the ovary: a matched control study of advanced-stage patients treated with cisplatin-based chemotherapy. Am J Obstet Gynecol 168 (4): 1178-85; discussion 1185-7, 1993. https://www.ncbi.nlm.nih.gov/pubmed/8475964[↩]
- Safra T, Borgato L, Nicoletto MO, et al.: BRCA mutation status and determinant of outcome in women with recurrent epithelial ovarian cancer treated with pegylated liposomal doxorubicin. Mol Cancer Ther 10 (10): 2000-7, 2011. http://mct.aacrjournals.org/content/10/10/2000.long[↩]
- Struewing JP, Hartge P, Wacholder S, et al: The risk of cancer associated with specific mutations of BRCA1 and BRCA2 among Ashkenazi Jews. N Engl J Med 336:1401-1408, 1997. http://www.nejm.org/doi/full/10.1056/NEJM199705153362001[↩]
- Levanon K, Crum C, Drapkin R. New Insights Into the Pathogenesis of Serous Ovarian Cancer and Its Clinical Impact. Journal of Clinical Oncology. 2008;26(32):5284-5293. doi:10.1200/JCO.2008.18.1107. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2652087/[↩]