What is Ginger
Ginger also known as the rhizome of Zingiber officinale Roscoe, is a tropical plant that has green-purple flowers and a fragrant underground stem called a rhizome. The rhizome (underground part of the stem) is the part commonly used as a spice. Ginger is often called ginger root, or simply ginger. Ginger belongs to the Zingiberaceae family and the Zingiberaceae family consists of 49 genera and 1,300 species, of which there are 80–90 species of Zingiber and 250 species of Alpinia 1. Ginger is widely used as a flavoring or fragrance in foods, beverages, soaps, and cosmetics 2. Ginger is a very popular spice and the world production is estimated at 100,000 tons annually, of which 80% is grown in China 3. Ancient Sanskrit, Chinese, Greek, Roman, and Arabic texts discussed the use of ginger for health-related purposes. Ginger has a long history of medicinal use dating back 2,500 years in China and India for conditions such as headaches, nausea, rheumatism, stomach ache, diarrhea, and colds 4, 2. Characterized in traditional Chinese medicine as spicy and hot, ginger is claimed to warm the body and treat cold extremities, improve a weak and tardy pulse, address a pale complexion, and strengthen the body after blood loss 5. Over the last few years, interest in ginger or its various components as valid preventive or therapeutic agents has increased markedly, and scientific studies focusing on verification of ginger’s pharmacological and physiological actions have likewise increased 6. Today, ginger is used as a dietary supplement for postsurgery nausea; nausea caused by motion, chemotherapy, or pregnancy (morning sickness); rheumatoid arthritis; and osteoarthritis. Ginger can have side effects such as abdominal discomfort, heartburn, diarrhea, and mouth and throat irritation, especially if taken in large doses. Some studies of the use of ginger during pregnancy suggest it is safe, but the evidence is not conclusive. If you’re considering using ginger while pregnant, consult your health care provider. Little is known about whether it’s safe to use ginger while breastfeeding.
Ginger benefits:
- Ginger has been studied for several types of nausea and vomiting. Almost all of these studies tested dietary ginger supplements rather than foods.
- Research shows that ginger may be helpful for mild nausea and vomiting associated with pregnancy 7, 8.
- Most studies of ginger for motion sickness haven’t shown it to be helpful.
- In general, ginger hasn’t been shown to relieve nausea and vomiting associated with cancer chemotherapy, although it might be helpful if taken in combination with certain types of chemotherapy or certain drugs used to prevent nausea and vomiting 9.
- It’s uncertain whether ginger is helpful for postoperative nausea and vomiting 10.
- A small amount of research suggests that ginger dietary supplements might be helpful for menstrual cramps 11, 12..
- There’s not enough evidence to show whether ginger supplements are helpful for symptoms of knee osteoarthritis 13.
Usage, preparation and processing of Ginger
Ginger is used in numerous forms, including fresh, dried, pickled, preserved, crystallized, candied, and powdered or ground. Common forms of ginger include the fresh or dried root, tablets, capsules, liquid extracts, and teas. The flavor is somewhat peppery and slightly sweet, with a strong and spicy aroma. The concentration of essential oils increases as ginger ages and, therefore, the intended use of the rhizome determines the time when it is harvested. If extracting the oil is the main purpose, then ginger can be harvested at 9 months or longer. Ginger is commonly pickled in sweet vinegar, which turns it a pink color; this form is popular with sushi. Ginger harvested at 8-9 months has a tough skin that must be removed before eating, and the root is more pungent and is used dried or pulverized into ground ginger. This is the form most commonly found in our spice racks and used in cookies, cakes, and curry mixes. Candied or crystallized ginger is cooked in sugar syrup and coated with granulated sugar. Ginger harvested at 5 months is not yet mature and has a very thin skin, and the rhizomes are tender with a mild flavor and are best used in fresh or preserved forms.

Ginger Nutrition Content
Table 1. Ginger Root (Raw) Nutrition Content
| Nutrient | Unit | Value per 100 g | tsp 2 g | cup slices (1″ dia) 24 g | slices (1″ dia) 11 g | |
| Approximates | ||||||
| Water | g | 78.89 | 1.58 | 18.93 | 8.68 | |
| Energy | kcal | 80 | 2 | 19 | 9 | |
| Protein | g | 1.82 | 0.04 | 0.44 | 0.2 | |
| Total lipid (fat) | g | 0.75 | 0.01 | 0.18 | 0.08 | |
| Carbohydrate, by difference | g | 17.77 | 0.36 | 4.26 | 1.95 | |
| Fiber, total dietary | g | 2 | 0 | 0.5 | 0.2 | |
| Sugars, total | g | 1.7 | 0.03 | 0.41 | 0.19 | |
| Minerals | ||||||
| Calcium, Ca | mg | 16 | 0 | 4 | 2 | |
| Iron, Fe | mg | 0.6 | 0.01 | 0.14 | 0.07 | |
| Magnesium, Mg | mg | 43 | 1 | 10 | 5 | |
| Phosphorus, P | mg | 34 | 1 | 8 | 4 | |
| Potassium, K | mg | 415 | 8 | 100 | 46 | |
| Sodium, Na | mg | 13 | 0 | 3 | 1 | |
| Zinc, Zn | mg | 0.34 | 0.01 | 0.08 | 0.04 | |
| Vitamins | ||||||
| Vitamin C, total ascorbic acid | mg | 5 | 0.1 | 1.2 | 0.6 | |
| Thiamin | mg | 0.025 | 0 | 0.006 | 0.003 | |
| Riboflavin | mg | 0.034 | 0.001 | 0.008 | 0.004 | |
| Niacin | mg | 0.75 | 0.015 | 0.18 | 0.083 | |
| Vitamin B-6 | mg | 0.16 | 0.003 | 0.038 | 0.018 | |
| Folate, DFE | µg | 11 | 0 | 3 | 1 | |
| Vitamin B-12 | µg | 0 | 0 | 0 | 0 | |
| Vitamin A, RAE | µg | 0 | 0 | 0 | 0 | |
| Vitamin A, IU | IU | 0 | 0 | 0 | 0 | |
| Vitamin E (alpha-tocopherol) | mg | 0.26 | 0.01 | 0.06 | 0.03 | |
| Vitamin D (D2 + D3) | µg | 0 | 0 | 0 | 0 | |
| Vitamin D | IU | 0 | 0 | 0 | 0 | |
| Vitamin K (phylloquinone) | µg | 0.1 | 0 | 0 | 0 | |
| Lipids | ||||||
| Fatty acids, total saturated | g | 0.203 | 0.004 | 0.049 | 0.022 | |
| Fatty acids, total monounsaturated | g | 0.154 | 0.003 | 0.037 | 0.017 | |
| Fatty acids, total polyunsaturated | g | 0.154 | 0.003 | 0.037 | 0.017 | |
| Fatty acids, total trans | g | 0 | 0 | 0 | 0 | |
| Cholesterol | mg | 0 | 0 | 0 | 0 | |
| Other | ||||||
| Caffeine | mg | 0 | 0 | 0 | 0 | |
Bioactive Components of Ginger
Pharmacologically, ginger, similar to other plants, is a very complex mixture of compounds. More than approximately 115 compounds have reportedly been isolated from fresh and dried ginger 15. Specifically, the major classes of ginger compounds are gingerol, shogaols, zingiberene, and zingerone, as well as other less common compounds, including terpenes, vitamins, beta-carotene, capsaicin, caffeic acid, volatile oils and curcumin 16. Among them, gingerols (6-Gingerol) are considered as the primary components, providing ginger with its unique fragrance and flavor and is reported to possess several biological activities including anticancer, anti-inflammation, and anti-oxidation 17, 18. As a result, many related biological activities have been explored such as those of antioxidant, antimicrobial, and anti-neuroinflammation, just to name a few 19. 6-Gingerol has been found to possess anticancer activities via its effect on a variety of biological pathways involved in apoptosis, cell cycle regulation, cytotoxic activity, and inhibition of angiogenesis 18. Thus, due to its efficacy and regulation of multiple targets, as well as its safety for human use, 6-gingerol has received considerable interest as a potential therapeutic agent for the prevention and/or treatment of various diseases.
The oleoresin (i.e., oily resin) from the rhizomes (i.e., ginger roots) of ginger contains many bioactive components, such as [6]-gingerol (1-[4′-hydroxy-3′- methoxyphenyl]-5-hydroxy-3-decanone), which is the primary pungent ingredient that is believed to exert a variety of remarkable pharmacological and physiological activities 20. Gingerols possess the labile β-hydroxy keto functional group, which makes it susceptible to transformation to less-pungent compounds such as shogaols and zingerone by elevated temperature 15. Gingerols and shogaols have been reported to exhibit many interesting pharmacological and physiological functions, for example, immuno-modulatory, anti-tumorigenic, anti-inflammatory, anti-apoptotic, anti-hyperglycemic, anti-lipidemic, anti-pyretic, cardiotonic, chemopreventive, anti-inflammatory, cancer preventive properties and antioxidant properties 21, 22, 23, 24. It has been reported that ginger extract inhibits the production of nitric oxide (NO) and proinflammatory cytokines in lipopolysaccharide (LPS)-stimulated BV-2 microglial cells via the NF-κB (nuclear factor kappa-light-chain-enhancer of activated B cells) pathway 25. An inhibitory effect by 6-gingerol was shown on the production of proinflammatory cytokines in murine peritoneal macrophages 26. Likewise, 6-shogaol has been shown to inhibit LPS-induced inducible nitric oxide synthase (iNOS) and cyclooxygenase (COX) gene expression in macrophages.14 Moreover, 6-shogaol showed significant neuroprotective effects in vivo in transient global ischemia via the inhibition of microglia. It suppressed the microglial activation induced by LPS both in primary cortical neuron-glia culture and in an in vivo neuroinflammatory model 27. In vitro data have shown that ginger’s active principles protect nerve cells and may have potential in the treatment of Alzheimer’s disease 28. This study 15 shows that ginger phytochemicals targets such as AChE, BuChE, COX-1, COX-2, JNK, and NOS, have already been verified by experiments for their inhibition by ginger extracts; it is being proposed that 1,3-diacetoxy derivative (compound 7) binds to the AChE active site with certain orientation and conformation so that it may act as an inhibitor of that enzyme. This study provides important information for optimizing lead compounds of ginger for the treatment of Alzheimer’s disease 15.
Ginger and Gingerols
Gingerols are the major constituents of fresh ginger and are found slightly reduced in dry ginger, whereas the concentrations of shogaols, which are the major gingerol dehydration products, are more abundant 29 in dry ginger than in fresh ginger. At least 31 gingerol-related compounds have been identified from the methanolic crude extracts of fresh ginger rhizome 30. Ginger has been fractionated into at least 14 bioactive compounds, including [4]-gingerol, [6]-gingerol, [8]-gingerol, [10]-gingerol, [6]-paradol, [14]-shogaol, [6]-shogaol, 1-dehydro-[10]-gingerdione, [10]-gingerdione, hexahydrocurcumin, tetrahydrocurcumin, gingerenone A, 1,7-bis-(4′ hydroxyl-3′ methoxyphenyl)-5-methoxyhepthan-3-one, and methoxy-[10]-gingerol 31. The proportion of each individual component in a sample of ginger depends on country of origin, commercial processor, and whether the ginger is fresh, dried, or processed 32. Of the bioactive pungent components of Jamaican ginger, including [6]-, [8]-, and [10]-gingerols and [6]-shogaol, [6]-gingerol appears to be the most abundant pungent bioactive compound in most of the oleoresin samples studied 33. Although phylogenetic analysis has showed that all ginger samples from widely different geographical origins are genetically indistinguishable, metabolic profiling showed some quantitative differences in the contents of [6]-, [8]-, and [10]-gingerols 34. An examination of the concentrations of [6]-, [8]-, and [10]-gingerols and [6]-shogaol in 10 different ginger-root dietary supplements purchased randomly from a variety of pharmacies and health food stores yielded some disconcerting results 32. Perhaps not surprisingly, the content of these active components was found to vary extensively from none or very minute amounts to several milligrams per gram. In addition, the suggested serving size ranged from about 250 mg to 4.8 g/day 32. The basis for the wide range of dosing is not clear. These studies suggest that ginger contains a variety of bioactive compounds and standardization of contents is critically lacking.
Benefits of Ginger
There’s some information from scientific studies suggest that ginger (Zingiber officinale) might have beneficial effects on nausea and vomiting associated with motion sickness, surgery, and pregnancy. Moreover, in recent years, the role of ginger has been extended to anticancer, chemotherapy-induced nausea and vomiting and fatigue, as well as improvements in the quality of life in daily human work 35, 36.
Asthma is a chronic disease characterized by inflammation and hypersensitivity of airway smooth muscle cells to different substances that induce spasms, and ginger has been used for centuries in treating respiratory illnesses. Components of ginger rhizomes are reported to contain potent compounds capable of suppressing allergic reactions and might be useful for the treatment and prevention of allergic diseases 37. Ghayur et al. 38 reported that a ginger extract inhibits airway contraction and associated calcium signaling, possibly by blocking plasma membrane calcium channels. In a mouse model of Th2-mediated pulmonary inflammation, an intraperitoneal injection of a ginger extract mainly comprised of gingerols markedly decreased the recruitment of eosinophils to the lungs in ovalbumin-sensitized mice and also suppressed the Th2 cell-driven response to allergen 39.
Ginger has also been suggested to have antidiabetic effects. In the streptozotocin-induced diabetic rat model, rats that were fed ginger exhibited better glucose tolerance and higher serum insulin levels than untreated rats, suggesting that it can help control blood sugar levels 40. Treatment with a ginger extract produced a significant reduction in fructose-induced elevation in lipid levels, body weight, hyperglycemia, and hyperinsulinemia associated with insulin resistance 41. An aqueous extract of raw ginger (administered daily, 500 mg/kg intraperitoneally) to streptozotocin-induced diabetic rats lowered serum glucose, cholesterol, and triacylglycerol levels; decreased urine protein levels, water intake, and urine output; and prevented the weight loss associated with diabetes in this model 42.
Dried ginger may have beneficial effects in treating dementia, including Alzheimer’s disease 43. Ulcerative colitis is a chronically recurrent inflammatory bowel disease of unknown origin, and in rats, ginger extract alleviated the symptoms of acetic acid-induced ulcerative colitis 44.
Ginger for Pregnancy-induced nausea and vomiting
A review of four randomized clinical trials 45, all trials found orally administered ginger to be significantly more effective than placebo in reducing the frequency of vomiting and intensity of nausea in pregnancy also called hyperemesis gravidarum in severe cases. The authors concluded that the best available evidence suggests that ginger is a safe and effective treatment for pregnancy related nausea and vomiting. Simultaneously, ginger showed a similar effect compared to other medication groups, such as vitamin B6 (pyridoxine), antihistamine, or metoclopramide. In addition, a study by Ensiyeh et al. 46 concluded that ginger is more effective in relieving the severity of nausea compared to vitamin B6. However, there remains uncertainty regarding the maximum safe dosage of ginger, appropriate duration of treatment, consequences of over-dosage, and potential drug-herb interactions; all of which are important areas for future research 45.
Another study on ginger and its safety for use during pregnancy 47 involving 1,020 Norwegian women who reported using ginger during pregnancy. The study found no evidence that using ginger during pregnancy was not associated with any increased risk of congenital malformations. No increased risk for stillbirth/perinatal death, preterm birth, low birth weight, or low Apgar score was detected for the women exposed to ginger during pregnancy compared to women who had not been exposed. This finding is clinically important for health care professionals giving advice to pregnant women with pregnancy-induced nausea and vomiting 47.
Ginger and Osteoarthritis
Osteoarthritis (OA) is a degenerative joint disease, which is associated with increased pain and disability, and a simultaneous decline in the quality of life of sufferers. While there is no cure for osteoarthritis, there are numerous treatments that aim to reduce sufferers’ symptoms and disability, and improve their quality of life. Medications, which have long been integral interventions for the management of osteoarthritis, have recently been found to cause harm in some patients. Simultaneously, the increasing recognition of complementary and alternative therapies as part of mainstream health care, has seen many sufferers of osteoarthritis use these therapies. Ginger has been commonly prescribed by herbalists for sufferers of osteoarthritis due to its anti-inflammatory and circulatory stimulant effects 48.
A systematic review was to evaluate the safety and effectiveness of ginger in adults with osteoarthritis 48. For changes in pain severity, studies comparing ginger extract (n = 110) to placebo (n = 111) reported mixed findings in support of the use of Ginger. Studies comparing ginger to an active control found participants who received Ibuprofen (n = 96) had a greater change in median pain intensity compared with participants who received Ginger (n = 110), and while findings were statistically significant for only one of the two studies, the results had limited clinical significance. Similarly, while two placebo-controlled studies reported differences between ginger (n = 70) and placebo (n = 71) for changes in disability and functional capacity, the difference was statistically and clinically significant for only one study. In one study comparing ginger to an active control, participants receiving Ibuprofen (n = 56) reported a statistically significant improvement in disability and functional capacity over time when compared with participants receiving Ginger (n = 56). The three randomized control trials that considered the efficacy of oral ginger (powder supplementation of 1 g/day) on pain in knee osteoarthritis demonstrated that efficacy of ginger on knee pain seemed not to have a unique position. Two studies suggested that the use of ginger in subjects with knee pain could decrease pain 49, while other did not find significant differences 50, 51. The two randomized control trial that assessed the efficacy of the topical use of ginger considered an aromatic essential oil (1% Zingiber officinale and 0.5% Citrus sinesis) 52 and 4% ginger gel 51 and agree in stating that the topical use of ginger seems to have potential as an alternative method for short‐term knee pain relief. Finally, all the four studies that considered the ginger supplementation in combination with other botanicals agree in demonstrating that these combinations are effective in order to decrease knee pain 53. Only one randomized control trial, that considered the efficacy of oral ginger (powder supplementation of 1 g/day) on pain in knee osteoarthritis, did not find significant changes 51. In terms of safety, Ginger was well tolerated when compared with Ibuprofen, with infrequent reports of mild, and predominantly gastrointestinal, adverse effects.
Conclusion of the systematic review: Current evidence is weak for the use of ginger in adults with osteoarthritis of the knee and/or hip 48. Much of this can be attributed to significant heterogeneity between studies. Improvements in research design, instrumentation and ginger dosage, which more closely reflect current clinical practice, may help to demonstrate the safe and effective use of ginger in osteoarthritis sufferers.
Pain relieve function
Four randomized control trials administered ginger to a group of participants with muscular pain, with varied result observed. Two studies reported a lack of evidence regarding the effect of ginger, and the other two reported that ginger partially attenuated muscular pain compared to the placebo group. Migraines and headaches were examined to assess the pain-relieving attributes of ginger in three randomized control trials. Maghbooli et al. 54 and Martins et al. 55 compared the effects of sumatriptan and placebo, respectively, both showing there was a significant difference in symptom attenuation. Patients with low back pain and chest pain caused by percutaneous transluminal coronary angioplasty were also selected to assess the analgesic effects of ginger, and both studies concluded that ginger was a useful option for pain relief.
Ginger for treating primary Dysmenorrhea
A systematic review evaluated the use of ginger in alleviating symptoms of primary dysmenorrhea (painful menstruation, typically involving abdominal cramps) 56, the review authors conclusion based on four randomized clinical trials, there is suggestive evidence for the effectiveness of 750–2000 mg ginger powder during the first 3–4 days of menstrual cycle for primary dysmenorrhea 56. All randomized control trials agree in demonstrating that ginger is more effective for pain relief than placebo, and no significant difference was found between ginger and nonsteroidal anti-inflammatory drugs (NSAIDs) 57. These findings, however, need to be interpreted with caution due to the small number of studies, poor methodological quality, and high heterogeneity across the trials. Moreover, all of the included trials were conducted in Asia. For example, Rahnama et al. 58 reported that ginger significantly improved primary dysmenorrhea in ginger-treated patients for five days, beginning two days before the onset of menstruation. Only one study concluded that ginger was an insufficient pain reliever upon comparison with stretching and exercising for alleviation 59.
Ginger Helps Reduce Nausea from Chemotherapy
According to the National Cancer Institute 60 ginger helped prevent or reduce chemotherapy-induced nausea when taken with traditional anti-nausea drugs by patients with cancer, researchers have found. The results are from a randomized, double-blind, placebo-controlled clinical trial, the largest study to examine the potential effects of ginger on chemotherapy-related nausea 61.
Dr. Ryan and colleagues conducted a multisite randomized trial to assess the efficacy of ginger for chemotherapy-related nausea in cancer patients at member sites of the University of Rochester–affiliated Community Clinical Oncology Program 61. The participants consisted of 644 primarily female patients (90%) who were receiving chemotherapy for breast, digestive, lung, or other cancers. These patients were randomly assigned to receive a placebo or one of three doses of ginger (0.5 g, 1.0 g, or 1.5 g) in capsule form which was divided into 2 doses given each day for 6 days, including 3 days leading up to the first day of chemotherapy and 2 days after chemotherapy began. Breast cancer was the most common type of malignancy among the participants (66%), followed by alimentary (6.5%) and lung (6.1%) cancers.
Patients who had experienced nausea following any chemotherapy cycle and who were scheduled to receive at least 3 additional treatment cycles were eligible to participate in the study. All of the patients also received traditional anti-nausea medications during the study, 5-HT3 receptor antagonist antiemetics — ondansetron (Zofran) or granisetron (Kytril) — starting on day 1 of all cycles and began supplementation with either ginger or placebo 3 days before the first day of a chemotherapy cycle.
Patients rated their nausea four times per day on a scale of 1 to 7. The degree of nausea experienced was rated at various times during the first 4 days of each chemotherapy cycle.
Their results showed that all doses of ginger significantly reduced nausea more than the placebo, with the 0.5 g and 1.0 g doses having the greatest effect. That dose is the equivalent of 1/4 to 1/2 a teaspoon of ground ginger, Dr. Ryan added 61.
Patients using ginger rated their nausea 1, which is none at all, or 2, which is slight nausea, said Dr. Ryan, while the placebo group rated their nausea 4, which is extremely nauseated.
Dr. Ryan speculates that the highest dose (1.5 gram) might not have worked as well as the lower doses because they may have reached the saturation point with the lower doses. She also could not speculate whether food products that contain ginger, such as cookies and tea, would have the same effect. “If they contain the same amount of ginger, then it’s possible,” she said. “But my guess is that a lot of them just use ginger flavoring, not real ginger. And they may also contain ingredients like sugar, which could affect the efficacy.”
The researchers concluded each dose of ginger was more effective than the placebo at mitigating nausea. The most effective doses were either 0.5 g or 1.0 g, taken during the first day of chemotherapy. Effectiveness decreased linearly over a 24-hour period. The highest dose (1.5 gram) may not be as effective, the researchers speculated, because it is more than the maximum absorption dose for biological activity 61. Reduced nausea will improve cancer patient quality of life during chemotherapy.
In another study using ginger root powder as an anti-emetic and an add-on therapy in children and young adults receiving high emetogenic chemotherapy 62. Acute chemotherapy-induced nausea and vomiting was defined as nausea and vomiting occurring within 24 hr of start of chemotherapy (days 1-4) and delayed chemotherapy-induced nausea and vomiting as that occurring after 24 hr of completion of chemotherapy (days 5-10). Sixty chemotherapy cycles of cisplatin/doxorubicin in bone sarcoma patients were randomized to ginger root powder capsules or placebo capsules as an additional antiemetic to ondensetron and dexamethasone in a double-blind design. The results were acute moderate to severe nausea was observed in 28/30 (93.3%) cycles in control group as compared to 15/27 (55.6%) cycles in experimental group (those receiving ginger root powder). Acute moderate to severe vomiting was significantly more in the control group compared to the experimental group [23/30 (76.7%) vs. 9/27 (33.33%) respectively. Delayed moderate to severe nausea was observed in 22/30 (73.3%) cycles in the control group as compared to 7/27 (25.9%) in the experimental group. Delayed moderate to severe vomiting was significantly more in the control group compared to the experimental group [14/30 (46.67%) vs. 4/27 (14.81%). The study concluded that ginger root powder was effective in reducing severity of acute and delayed chemotherapy-induced nausea and vomiting as additional therapy to ondensetron and dexamethasone in patients receiving high emetogenic chemotherapy 62.
Metabolic syndrome
Studies evaluating the efficiency of ginger in metabolic syndromes have also been widely conducted. Most of the included studies assessed the association of type 2 diabetes mellitus and obesity with ginger supplementation. In detail, five studies explored the effect of ginger on the diabetes-related indices such as glycemic markers, lipid level, and blood pressure, while four studies focused on various conditions related to obesity such as cardiovascular disease, serum adipocytokines, and breast cancer. For example, three studies evaluated the influence of ginger on biochemical parameters related to type 2 diabetes and demonstrated the significant lowering of fasting blood sugar, hemoglobin A1c (HbA1c), insulin sensitivity, and insulin resistance. Furthermore, lipid profile, inflammatory markers, and antioxidants were also affected by ginger intake, which was demonstrated by the reduction of the C-reactive protein, triglycerides, low-density lipoprotein cholesterol (LDL-C), and malondialdehyde. Another study assessed the relationship between ginger and blood pressure in type 2 diabetes but showed no significant differences compared to the control group. Regarding obesity, the included study targeted the effect of ginger on obesity. In obese women, Attari et al. 63 reported that ginger supplements had a minor benefit on weight loss, the reduction of insulin and homeostasis model assessment of insulin resistance (HOMA-IR), and the increasing of quantitative insulin sensitivity check index (QUICKI). In terms of obesity-related cardiovascular risk factors, ginger was reported to be beneficial in lowering the risk factors, such as body fat mass, body fat percentage, total cholesterol, waist circumference, waist-to-hip ratio, and insulin resistance. In addition, ginger was suggested to have antioxidant and anti-dysmetabolic effects in obese women with breast cancer. Finally, other studies aimed to explore the effect of ginger on lipid metabolism including fat utilization and triglyceride-lowering efficacy. In general, ginger was believed to provide potential benefits by reducing the risk factors of metabolic syndromes. Moreover, no serious adverse effects were observed in all included studies 64.
General Antioxidant Properties of Ginger
The presence of oxidative stress is associated with numerous diseases and a common mechanism often put forth to explain the actions and health benefits of ginger is associated with its antioxidant properties 65, 66. Ginger was reported to decrease age-related oxidative stress markers 67 and was suggested to guard against ethanol-induced hepatotoxicity by suppressing oxidative consequences in rats treated with ethanol 68. Ginger root contains a very high level (3.85 mmol/100 g) of total antioxidants, surpassed only by pomegranate and some types of berries 69. The phorbol ester, 12-O-tetradecanoylphorbol-13-acetate (TPA), promotes oxidative stress by activating the nicotinamide adenine dinucleotide phosphate (NADPH) oxidase system or the xanthine oxidase system or both. Ginger was reported to suppress TPA-induced oxidative stress in human promyelocytic leukemia (HL)-60 cells and Chinese hamster ovary AS52 cells 70. Others have shown that ginger compounds effectively inhibit superoxide production 71. Several animal studies indicate that ginger suppresses lipid peroxidation and protects the levels of reduced glutathione 72, 73, 74, 75, 76, 77.
Reactive nitrogen species, such as nitric oxide (NO), influence signal transduction and cause DNA damage, which contributes to disease processes. Nitric oxide is produced by inducible nitric oxide synthase (iNOS), which is stimulated in response to various stresses. [6]-gingerol was reported to dose-dependently inhibit NO production and reduce iNOS in lipopolysaccharide (LPS)-stimulated mouse macrophages 78. [6]-gingerol also effectively suppressed peroxynitritemediated oxidative damage. Ippoushi et al. 78 later proposed that [6]-gingerol and peroxynitrite form a symmetric dimer with [6]-gingerol covalently linked at the aromatic ring of peroxynitrite, attenuating peroxynitrite-induced oxidation and nitration reactions 79. [6]-shogaol, 1-dehydro-[10]-gingerdione, and [10]-gingerdione also decreased LPS-induced NO production, and [6]-shogaol and 1-dehydro-[10]-gingerdione were reported to effectively reduce iNOS expression 31. In the bromobenzene (BB)-induced hepatotoxicity model, orally given ginger extract (100 mg/kg body weight) normalized NO levels and total and reduced glutathione levels, and also decreased the level of lipid peroxidation 77. Ginger consumption has also been reported to decrease lipid peroxidation and normalize the activities of superoxide dismutase and catalase, as well as GSH and glutathione peroxidase, glutathione reductase, and glutathione-S-transferase, in rats 76. Ginger supplementation before ischemia/reperfusion resulted in a higher total antioxidant capacity (i.e., normalized glutathione peroxidase and superoxide dismutase activities) and lower total oxidant (lower tissue malondialdehyde, NO, and protein carbonyl contents) status levels compared to an untreated group of Wistar albino rats 80. Overall, the rats fed ginger (5%) experienced less kidney damage due to oxidative stress induced by ischemia/reperfusion (Uz E, Karatas O. F, Mete E, Bayrak R, Bayrak O, Atmaca A. F, Atis O, Yildirim M. E, Akcay A. The effect of dietary ginger (Zingiber officinals Rosc.) on renal ischemia/reperfusion injury in rat kidneys. Ren Fail. 2009;31(4):251–60. https://www.ncbi.nlm.nih.gov/pubmed/19462272)).
Ginger extract has been reported to exert radioprotective effects in mice exposed to gamma radiation 81, and the effect was associated with decreased lipid peroxidation and protection of GSH levels 82. [6]-gingerol pretreatment also decreased oxidative stress induced by ultraviolet B (UVB) and activated caspase-3, -8, -9, and Fas expression 83. Evidence does seem to suggest that ginger and some of its components are effective antioxidants in vitro. However, whether the physiological activity occurs in humans in vivo is not clear, and the specific mechanism and cellular targets are still to be determined.
Anti-Inflammatory Effects of Ginger
One of the many health claims attributed to ginger is its purported ability to decrease inflammation, swelling, and pain. Gingerol 84, a dried ginger extract, and a dried gingerol-enriched extract 85 were each reported to exhibit analgesic and potent anti-inflammatory effects. Ginger has been suggested to be effective against inflammation, osteoarthritis, and rheumatism 86. However, inconsistencies in clinical studies have led to debate regarding the effectiveness and safety of ginger for treatment of arthritis 87.
Earlier animal studies suggest that rat hind limbs perfused with [6]-gingerol showed increased heat production that was associated with increased oxygen consumption and lactate efflux 88. The thermogenesis was at least partly associated with vasoconstriction independent of adrenergic receptors or secondary catecholamine release. In contrast, larger doses of ginger components inhibited oxygen consumption, which was attributed to disruption of mitochondrial function 88. These results were supported in a later study in which rats that were given a single intraperitoneal injection of [6]-gingerol (2.5 or 25 mg/kg) exhibited a rapid, marked drop in body temperature and a significant decrease in metabolic rate 89.
In addition, salicylate has been found in ginger in amounts of 4.5 mg/100 gm fresh root 90. This would correspond to <1 mg salicylate in 1 capsule of the ginger extract. The actions and especially the interactions of these ingredients have not been (and probably can not be easily) evaluated. Various powders, formulations, and extracts have, however, been commercially used and tested, both in vitro and in vivo, in animal models. In these models, ginger has been shown to act as a dual inhibitor of both cyclooxygenase (COX) and lipooxygenase 91, to inhibit leukotriene synthesis 92 and to reduce caregeenan-induced rat-paw edema and yeast-induced fever 93, 94, an animal model of inflammation.
Researchers have hypothesized that the anti-inflammatory effects of ginger might be related to its ability to inhibit prostaglandin and leukotriene biosynthesis 95. Some others have showed that gingerols actively inhibit arachidonate 5-lipoxygenase, an enzyme of leukotriene biosynthesis 96. [8]-gingerol, but not [6]-gingerol, was shown to inhibit cyclooxygenase-2 (COX-2) expression, which is induced during inflammation to increase formation of prostaglandins 97. Others have also reported that ginger extract suppresses the activation of tumor necrosis factor α (TNF-α) and expression of COX-2 in human synoviocytes 98. Proinflammatory cytokines such as TNF-α, interleukin (IL)-1β, and IL-12, which are produced primarily by macrophages, play an important role in sepsis, ischemia/reperfusion injury, and transplant rejection. [6]-gingerol was reported to inhibit the production of proinflammatory cytokines from LPS-stimulated peritoneal macrophages, but to have no effect on the function of antigen presenting cells (APC) or the LPS-induced expression of proinflammatory chemokines. However, this same group later reported that a ginger extract attenuated the production of IL-12, TNF-α, and IL-1β proinflammatory cytokines and RANTES (regulated upon activation, normal T cell expressed and secreted) and monocyte chemoattractant protein 1 (MCP-1) proinflammatory chemokines in LPS-stimulated murine peritoneal macrophages 99. In general, ginger extract inhibited macrophage activation and APC function, and indirectly suppressed T-cell activation 99. Other stable [6]-gingerol metabolites or analogs were reported to suppress LPS-induced NO production in murine macrophages mainly by reducing inos gene and iNOS protein production (Aktan et al. 2006). Some of ginger’s anti-inflammatory effects appear to be associated with decreased IκBα degradation and impaired nuclear factor κB (NF-κB) nuclear translocation of p65 100. The majority of scientific evidence does seem to suggest that ginger and its various components have anti-inflammatory effects both in vitro and ex vivo. However, the data supporting ginger as an effective anti-inflammatory agent in humans in vivo are still contradictory and incomplete.
Overall, eight randomized control trials reported the anti-inflammatory effect of ginger supplementation. Among them, arthritis-related diseases were the most conducted studies, particularly osteoarthritis (OA). Regarding osteoarthritis, six studies investigated the efficiency of the constituents of ginger that serve as anti-inflammatory agents. All studies reported improvement following ginger intake compared to the control group. For instance, Mozaffari-Khosravi et al. 101 proposed that benefits of ginger were observed due to a reduction in the level of the proinflammatory cytokines after three months of consuming 500 mg of ginger powder. Other studies showed a promising benefit of ginger in relieving pain in osteoarthritis patients. Furthermore, no significant adverse effects were observed during the trials. An additional study that assessed ginger’s effects on rheumatoid arthritis demonstrated improvement by reducing symptoms via inducing FOXP3 gene expression. Finally, Kulkarni et al. 102 reported that ginger supplementation alone and combined with antitubercular treatment significantly helped to decrease tumor necrosis factor (TNF) alpha, ferritin, and malondialdehyde (MDA) levels compared to the control group.
Anti-Cancer Activities of Ginger
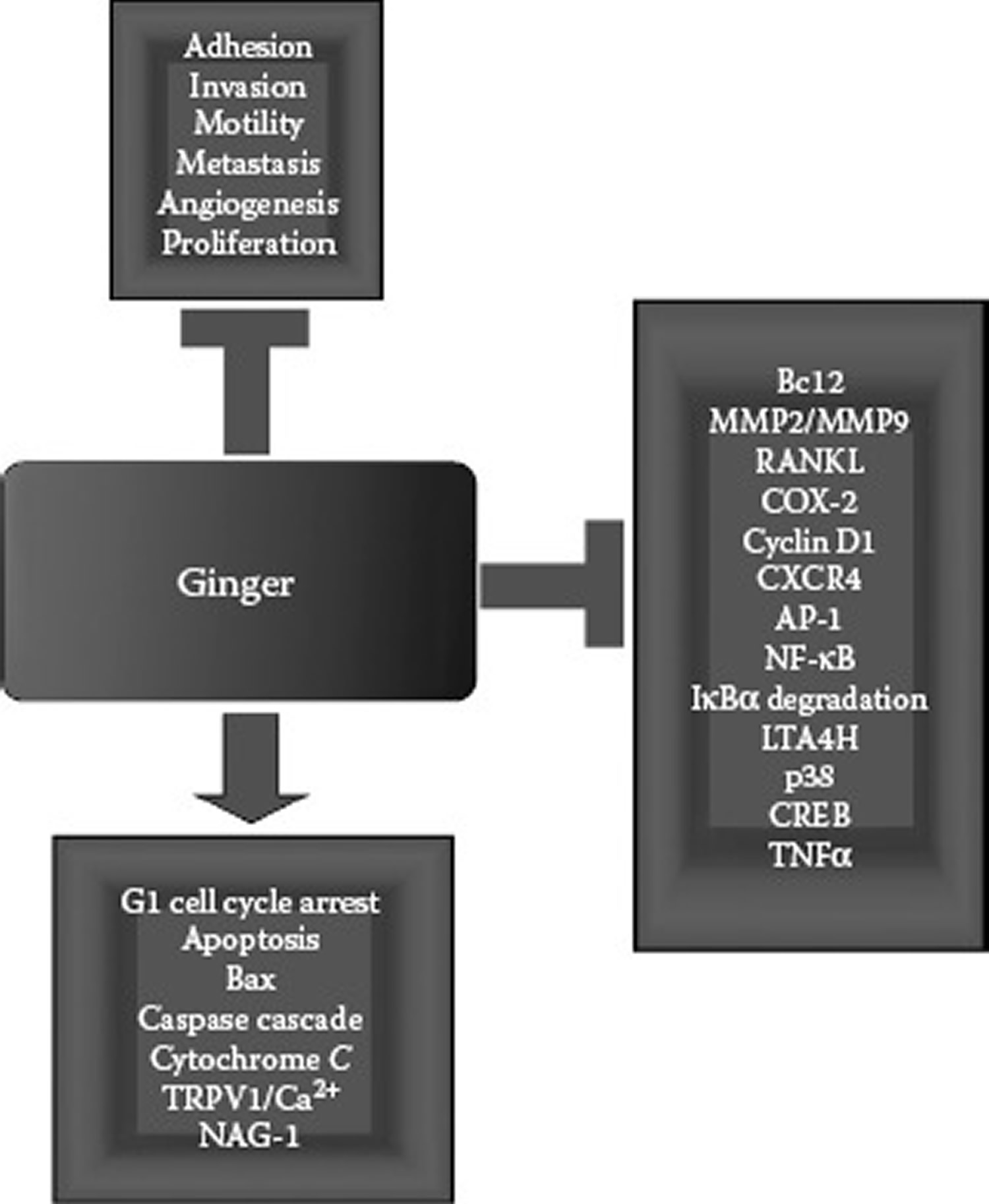
A great deal of interest by numerous research groups,is now being focused on the cancer-preventive and potential cancer therapeutic applications of ginger and its various components. Several aspects of the chemopreventive effects of numerous phytochemical dietary and medicinal substances, including ginger, have been reviewed previously 103, 104, 105, 106, 107, 108. Studies focused on the anticancer activities of various forms of ginger from a crude or partially purified extract to gingerols, especially 6-gingerol; shogaols, especially 6-shogaol; and zerumbone, a sesquiterpene compound derived from ginger and a number of minor components and metabolites. The effectiveness of ginger in preventing or suppressing cancer growth has been examined in a variety of cancer types, including lymphoma, hepatoma, colorectal cancer, breast cancer, skin cancer, liver cancer, and bladder cancer. The mechanisms proposed to explain the anticancer activities of ginger and its components include antioxidant activity and the ability to induce apoptosis, decrease proliferation, cause cell-cycle arrest, and suppress activator protein 1 (AP-1) and NF-κB/COX-2 signaling pathways (Figure 1).
Four randomized control trials examined the anticancer effect of ginger, all of which evaluated the risk of colorectal cancer according to the ginger treatment 64. Collectively, ginger has a beneficial effect on colorectal cancer by reducing tumorigenic risk factors. However, Jiang et al. 109 reported that participants with an average risk of colorectal cancer showed no significant aspect between the ginger and placebo groups. Citronberg et al. 110 investigated cell cycle markers with biopsies from the patients with increased risk of colorectal cancer and demonstrated the regulation of apoptotic and differentiation markers by ginger supplementation.
Figure 1. The anticancer activities exerted by ginger.
[Source 111]The anticancer activities of [6]-gingerol and zerumbone have been associated with their antioxidant activities. Several ginger components were reported to have effective anticancer promoter activity based on their ability to inhibit TPA-induced Epstein-Barr virus early antigen (EBV-EA) in Raji cells 112, 113. [6]-gingerol was reported to suppress the reactive oxygen species-potentiated invasive capacity of ascites hepatoma AH109A cells by reducing peroxide levels 114. In normal RL34 rat liver epithelial cells, zerumbone was found to induce glutathione S-transferase and the nuclear localization of the transcription factor Nrf2, which binds to the antioxidant response element (ARE) of phase II enzyme genes 115. Zerumbone potentiated the expression of several Nrf2/ARE-dependent phase II enzyme genes, including Y-glutamyl-cysteine synthetase, glutathione peroxidase, and hemeoxygenase-1 115. Others have reported that zerumbone decreases TPA-induced hydrogen peroxide formation and edema corresponding to enhanced levels of various antioxidant enzymes 116. These types of changes have been linked with lower 7,12-dimethylbenz[a]anthracene (DMBA)-initiated/TPA-promoted tumor incidence, number of tumors per mouse, and tumor volume 116.
Treatment of cultured ovarian cancer cells with [6]-shogaol caused a marked growth inhibition that was associated with suppression of NF-κB activation as well as the diminished secretion of angiogenic factors, VEGF and IL-8 117, suggesting a role for this compound in preventing angiogenesis in cancer. In contrast to most reports, dietary consumption of ginger (0.5% or 1.0%) did not suppress aberrant crypt foci (ACF) formation or reduce the number of crypts per ACF in DMH-treated rats compared to untreated control rats 118. Dietary ginger did not significantly change the proliferative or apoptotic indexes of the colonic crypt cells induced by DMH 118. In marked contrast to many studies, ginger extract was not able to inhibit the development of N-butyl-N-(4-hydroxybutyl)-nitrosamine (BBN)/N-methyl-N-nitrosourea (MNU)-induced bladder cancer in male Swiss mice. In fact, in BBN/MNU/2% ginger-treated mice, the incidence of grade 2 transitional cell carcinoma was increased 118, 119.
Cardiovascular and Other Disease-Preventive Effects of Ginger
In addition to its effects in relation to cancer, some evidence supports a protective role for ginger in cardiovascular function and a number of other disease conditions. Ginger has gained interest for its potential to treat various aspects of cardiovascular disease, and the in vitro and animal data supporting the anti-inflammatory, antioxidant, antiplatelet, hypotensive, and hypolipidemic effects of this condiment have been reviewed 120. However, human trials are less convincing and more investigations are needed 120. Caution when taking ginger and other herbal extracts has been suggested because of an apparent association of ginger with reported incidences of increased risk of bleeding following surgery 121, 122 or if taken with anticoagulant drugs such as warfarin 123. However, the data are not conclusive 124. At least one study indicates that ginger has no effect on blood pressure, heart rate, or coagulation parameters and does not interact with anticoagulant drugs such as warfarin 125. These findings were supported in a later study in which ginger was reported to have no effect on clotting status or the pharmacokinetics or pharmacodynamics of warfarin in healthy subjects 126. An aqueous ginger extract was reported to induce a dose-dependent decrease in arterial blood pressure in a variety of animal models 127.
At least one group found that administration or consumption of standardized ginger extract decreased aortic atherosclerotic lesion areas, plasma triglycerides and cholesterol, low-density lipoprotein (LDL)-associated lipid peroxides, and LDL aggregation in mice 128. In rabbits that were fed a high-cholesterol diet, administration of ginger extract resulted in a significant antihyperlipidemic effect and a lower degree of atherosclerosis compared to the group that was fed cholesterol alone 129. Importantly, ginger powder (3 g/day in 1-g capsule 3xd) significantly lowered lipid levels in volunteer patients in a double-blind, controlled clinical trial study 130. Triglyceride and cholesterol were substantially decreased as was LDL levels compared to placebo group. Notably, the high-density lipoprotein (HDL) level of the ginger group was higher than that of the placebo group, whereas the very-low-density lipoprotein (VLDL) level of the placebo group was higher than that of the ginger group 130. Dried ginger powder (0.1 g/kg BW, per oral administration [p.o.] for 75 days) significantly lowered (50%) the development of atheroma in the aorta and coronary arteries of rabbits that were fed cholesterol 131. This effect was associated with decreased lipid peroxidation and increased fibrinolytic activity with ginger, but blood lipid levels were not different from control animals 131. Another compound isolated from ginger, (E)-8 β,17-epoxylabd-12-ene-15,16-dial, was reported to inhibit cholesterol biosynthesis 132, and ginger meal (1%) decreased serum cholesterol levels significantly 118. Ginger was also reported to slightly reduce retinoid-binding protein mRNA expression levels in liver and visceral fat in male rats that were fed cholesterol to induce hyperlipidemia 133. These results hint that ginger consumption might improve lipid metabolism 133.
Ginger Side Effects
Ginger, when used as a spice, is believed to be generally safe 134, 2. Seventeen studies provided information about side effects in their research papers, most of which were not considered to be severely harmful to the participants. Among the adverse effects, gastrointestinal-related symptoms were mostly reported to reverse the gastrointestinal protective effect of ginger in other aspects 64. Heartburn, a general symptom of gastroesophageal reflux disease, was reported in sixteen studies. Five studies reported nausea as a side effect of ginger treatment, which was the primary topic evaluated to observe the clinical effect of ginger. Diarrhea was reported in two studies in groups of patients with heavy menstrual bleeding and after elective cesarean section. Other gastrointestinal symptoms included abdominal pain, bloating, gas, and epigastric distress. Furthermore, cardiovascular symptoms and respiratory symptoms were observed in a ginger-treated patient group who underwent laparoscopic surgery.
Observational studies in humans suggest no evidence of teratogenicity from treatments for early pregnancy nausea that included ginger 135. These results were confirmed in a similar trial showing that administration of ginger beginning at the first trimester of pregnancy did not appear to increase the rates of major malformations above the baseline rate of 1-3% 136. Overall, these data indicate that ginger consumption appears to be very safe with very limited side effects.
In some people, ginger can have mild side effects such as abdominal discomfort, heartburn, diarrhea, and gas.
Some experts recommend that people with gallstone disease use caution with ginger because it may increase the flow of bile.
Research has not definitely shown whether ginger interacts with medications, but concerns have been raised that it might interact with anticoagulants (blood thinners).
Although several studies have found no evidence of harm from taking ginger during pregnancy, it’s uncertain whether ginger is always safe for pregnant women. If you’re considering using ginger while you’re pregnant, consult your health care provider.
Ginger Summary
Ginger is an extremely popular dietary condiment used for flavoring food but also an herb that has been used for thousands of years as a medicinal herb to treat a variety of ailments. Chemical and metabolic analyses have revealed that ginger comprises hundreds of compounds and metabolites. The most extensively studied bioactive components include gingerols and shogaols, especially 6-gingerol and 6-shogaol, respectively. The content of each component is clearly dependent on the source and preparation of the ginger rhizome. Research interest in determining the role of natural compounds in preventing disease has increased markedly over the last few years. In spite of the abundance of research studies, many of the results are phenomenon based and provide data that are descriptive and observational on laboratory animals. More studies are needed humans on the kinetics of ginger and its constituents and on the effects of consumption over a long period of time. Specific molecular targets and mechanisms of action need to be identified. Ginger clearly has a vast number of components and metabolites, many of which have not been studied in detail. The lack of standardization of ginger supplements is disconcerting, and whether consumption of high levels of isolated components (e.g., 6-gingerol) is advisable is uncertain. 6-gingerol or other ginger components might require inter-reactivity or dependency on other components in the whole food source to exert their positive effects 137.
- Trease GE, Evans WC, editors. Pharmacognosy. 12th ed. London (UK): Bailliere Tindall; 1983.[↩]
- U.S. Department of Health & Human Services, National Institutes of Health. National Center for Complementary and Integrative Health. Ginger. https://nccih.nih.gov/health/ginger[↩][↩][↩]
- Langner ES, Griefenberg S, Gruenwald J. Ingwer: eine Heilpflanze mit Geschichte [Ginger: a medicinal plant with a history]. Balance Z Prax Wiss Komplementarer Ther 1997; 1: 5–16.[↩]
- Grant KL, Lutz RB. Ginger. Am J Health SystPharm 2000;57:945-947.[↩]
- Chang CP, Chang JY, Wang FY, Chang JG.The effect of Chinese medicinal herbZingiberis rhizoma extract on cytokinesecretion by human peripheral blood mono-nuclear cells. J Ethnopharmacol 1995;48:13-19.[↩]
- Ali B. H, Blunden G, Tanira M. O, Nemmar A. Some phytochemical, pharmacological and toxicological properties of ginger (Zingiber officinale Roscoe): A review of recent research. Food Chem Toxicol. 2008;46(2):409–20. https://www.ncbi.nlm.nih.gov/pubmed/17950516 [↩]
- Khorasani F, Aryan H, Sobhi A, Aryan R, Abavi-Sani A, Ghazanfarpour M, Saeidi M, Rajab Dizavandi F. A systematic review of the efficacy of alternative medicine in the treatment of nausea and vomiting of pregnancy. J Obstet Gynaecol. 2020 Jan;40(1):10-19. doi: 10.1080/01443615.2019.1587392[↩]
- McParlin C, O’Donnell A, Robson SC, Beyer F, Moloney E, Bryant A, Bradley J, Muirhead CR, Nelson-Piercy C, Newbury-Birch D, Norman J, Shaw C, Simpson E, Swallow B, Yates L, Vale L. Treatments for Hyperemesis Gravidarum and Nausea and Vomiting in Pregnancy: A Systematic Review. JAMA. 2016 Oct 4;316(13):1392-1401. doi: 10.1001/jama.2016.14337[↩]
- Crichton M, Marshall S, Marx W, McCarthy AL, Isenring E. Efficacy of Ginger (Zingiber officinale) in Ameliorating Chemotherapy-Induced Nausea and Vomiting and Chemotherapy-Related Outcomes: A Systematic Review Update and Meta-Analysis. J Acad Nutr Diet. 2019 Dec;119(12):2055-2068. doi: 10.1016/j.jand.2019.06.009[↩]
- Tóth B, Lantos T, Hegyi P, Viola R, Vasas A, Benkő R, Gyöngyi Z, Vincze Á, Csécsei P, Mikó A, Hegyi D, Szentesi A, Matuz M, Csupor D. Ginger (Zingiber officinale): An alternative for the prevention of postoperative nausea and vomiting. A meta-analysis. Phytomedicine. 2018 Nov 15;50:8-18. doi: 10.1016/j.phymed.2018.09.007[↩]
- Xu, Y., Yang, Q., & Wang, X. (2020). Efficacy of herbal medicine (cinnamon/fennel/ginger) for primary dysmenorrhea: a systematic review and meta-analysis of randomized controlled trials. The Journal of international medical research, 48(6), 300060520936179. https://doi.org/10.1177/0300060520936179[↩]
- Pattanittum, P., Kunyanone, N., Brown, J., Sangkomkamhang, U. S., Barnes, J., Seyfoddin, V., & Marjoribanks, J. (2016). Dietary supplements for dysmenorrhoea. The Cochrane database of systematic reviews, 3(3), CD002124. https://doi.org/10.1002/14651858.CD002124.pub2[↩]
- Araya-Quintanilla F, Gutierrez-Espinoza H, Munoz-Yanez MJ, Sanchez-Montoya U, Lopez-Jeldes J. Effectiveness of Ginger on Pain and Function in Knee Osteoarthritis: A PRISMA Systematic Review and Meta-Analysis. Pain Physician. 2020 Mar;23(2):E151-E161.[↩]
- United States Department of Agriculture, Agriculture Research Service. USDA Food Composition Databases. https://ndb.nal.usda.gov/ndb/[↩]
- Azam F, Amer AM, Abulifa AR, Elzwawi MM. Ginger components as new leads for the design and development of novel multi-targeted anti-Alzheimer’s drugs: a computational investigation. Drug Design, Development and Therapy. 2014;8:2045-2059. doi:10.2147/DDDT.S67778. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4211852/[↩][↩][↩][↩]
- Mahomoodally M.F., Aumeeruddy M.Z., Rengasamy K.R.R., Roshan S., Hammad S., Pandohee J., Hu X., Zengin G. Ginger and its active compounds in cancer therapy: From folk uses to nano-therapeutic applications. Semin. Cancer Biol. 2019 doi: 10.1016/j.semcancer.2019.08.009[↩]
- Kubra I.R., Rao L.J.M. An impression on current developments in the technology, chemistry, and biological activities of ginger (Zingiber officinale Roscoe) Crit. Rev. Food Sci. Nutr. 2012;52:651–688. doi: 10.1080/10408398.2010.505689[↩]
- Wang S, Zhang C, Yang G, Yang Y. Nat Prod Commun. 2014 Jul;9(7):1027-30. Biological properties of 6-gingerol: a brief review. https://www.ncbi.nlm.nih.gov/pubmed/25230520[↩][↩]
- Nile S.H., Park S.W. Chromatographic analysis, antioxidant, anti-inflammatory, and xanthine oxidase inhibitory activities of ginger extracts and its reference compounds. Ind. Crop. Prod. 2015;70:238–244. doi: 10.1016/j.indcrop.2015.03.033[↩]
- Ann M. Bode and Zigang Dong. Herbal Medicine: Biomolecular and Clinical Aspects. 2nd edition. Chapter 7: The Amazing and Mighty Ginger. https://www.ncbi.nlm.nih.gov/books/NBK92775/ [↩]
- Shukla Y, Singh M. Cancer preventive properties of ginger: a brief review. Food Chem Toxicol. 2007;45(5):683–690. https://www.ncbi.nlm.nih.gov/pubmed/17175086[↩]
- Surh Y. Molecular mechanisms of chemopreventive effects of selected dietary and medicinal phenolic substances. Mutat Res. 1999;428(1–2):305–327. https://www.ncbi.nlm.nih.gov/pubmed/10518003[↩]
- Ali BH, Blunden G, Tanira MO, Nemmar A. Some phytochemical, pharmacological and toxicological properties of ginger (Zingiber officinale Roscoe): a review of recent research. Food Chem Toxicol. 2008;46(2):409–420. https://www.ncbi.nlm.nih.gov/pubmed/17950516[↩]
- Pan MH, Hsieh MC, Hsu PC, et al. 6-Shogaol suppressed lipopolysaccharide-induced up-expression of iNOS and COX-2 in murine macrophages. Mol Nutr Food Res. 2008;52(12):1467–1477. https://www.ncbi.nlm.nih.gov/pubmed/18683823[↩]
- Jung HW, Yoon CH, Park KM, Han HS, Park YK. Hexane fraction of Zingiberis Rhizoma Crudus extract inhibits the production of nitric oxide proinflammatory cytokines in LPS-stimulated BV2 microglial cells via the NF kappaB pathway. Food Chem Toxicol. 2009;47(6):1190–1197. https://www.ncbi.nlm.nih.gov/pubmed/19233241[↩]
- Tripathi S, Maier KG, Bruch D, Kittur DS. Effect of 6-gingerol on proinflammatory cytokine production and costimulatory molecule expression in murine peritoneal macrophages. J Surg Res. 2007;138(2):209–213. https://www.ncbi.nlm.nih.gov/pubmed/17291534[↩]
- Ha SK, Moon E, Ju MS, et al. 6-Shogaol a ginger product modulates neuroinflammation: a new approach to neuroprotection. Neuropharmacology. 2012;63(2):211–223. https://www.ncbi.nlm.nih.gov/pubmed/22465818[↩]
- Kim DS, Kim DS, Oppel MN. Shogaols from Zingiber officinale protect human neuroblastoma and normal human umbilical vein endothelial cells from beta-amyloid (25–35) insult. Planta Med. 2002;68(4):375–376. https://www.ncbi.nlm.nih.gov/pubmed/11988870[↩]
- Jolad S. D, Lantz R. C, Chen G. J, Bates R. B, Timmermann B. N. Commercially processed dry ginger (Zingiber officinale): Composition and effects on LPS-stimulated PGE2 production. Phytochemistry. 2005;66(13):1614–35. https://www.ncbi.nlm.nih.gov/pubmed/15996695[↩]
- Jiang H, Solyom A. M, Timmermann B. N, Gang D. R. Characterization of gingerol-related compounds in ginger rhizome (Zingiber officinale Rosc.) by high-performance liquid chromatography/electrospray ionization mass spectrometry. Rapid Commun Mass Spectrom. 2005;19(20):2957–64. https://www.ncbi.nlm.nih.gov/pubmed/16189817[↩]
- Koh E. M, Kim H. J, Kim S, editors. et al. Modulation of macrophage functions by compounds isolated from Zingiber officinale. Planta Med. 2009;75(2):148–51. https://www.ncbi.nlm.nih.gov/pubmed/19031369[↩][↩]
- Schwertner H. A, Rios D. C, Pascoe J. E. Variation in concentration and labeling of ginger root dietary supplements. Obstet Gynecol. 2006;107(6):1337–43. https://www.ncbi.nlm.nih.gov/pubmed/16738161[↩][↩][↩]
- Bailey-Shaw Y. A, Williams L. A, Junor G. A, Green C. E, Hibbert S. L, Salmon C. N, Smith A. M. Changes in the contents of oleoresin and pungent bioactive principles of Jamaican ginger (Zingiber officinale Roscoe) during maturation. J Agric Food Chem. 2008;56(14):5564–71. https://www.ncbi.nlm.nih.gov/pubmed/18564850[↩]
- Jiang H, Xie Z, Koo H. J, McLaughlin S. P, Timmermann B. N, Gang D. R. Metabolic profiling and phylogenetic analysis of medicinal Zingiber species: Tools for authentication of ginger (Zingiber officinale Rosc) Phytochemistry. 2006;67(15):1673–85. https://www.ncbi.nlm.nih.gov/pubmed/16169024[↩]
- Crichton M., Marshall S., Marx W., McCarthy A.L., Isenring E. Efficacy of ginger (Zingiber officinale) in ameliorating chemotherapy-induced nausea and vomiting and chemotherapy-related outcomes: A systematic review update and meta-analysis. J. Acad. Nutr. Diet. 2019 doi: 10.1016/j.jand.2019.06.009[↩]
- Mao Q.-Q., Xu X.-Y., Cao S.-Y., Gan R.-Y., Corke H., Beta T., Li H.-B. Bioactive compounds and bioactivities of ginger (Zingiber officinale Roscoe) Foods. 2019;8:185. doi: 10.3390/foods8060185[↩]
- Chen C. Y, Li Y. W, Kuo S. Y. Effect of [10]-gingerol on [Ca2+]i and cell death in human colorectal cancer cells. Molecules. 2009;14(3):959–69. https://www.ncbi.nlm.nih.gov/pubmed/19255554[↩]
- Ghayur M. N, Gilani A. H, Janssen L. J. Ginger attenuates acetylcholine-induced contraction and Ca2+ signalling in murine airway smooth muscle cells. Can J Physiol Pharmacol. 2008;86(5):264–71. https://www.ncbi.nlm.nih.gov/pubmed/18432287[↩]
- Ahui M. L, Champy P, Ramadan A, editors. et al. Ginger prevents Th2-mediated immune responses in a mouse model of airway inflammation. Int Immunopharmacol. 2008;8(12):1626–32. https://www.ncbi.nlm.nih.gov/pubmed/18692598[↩]
- Islam M. S, Choi H. Comparative effects of dietary ginger (Zingiber officinale) and garlic (Allium sativum) investigated in a type 2 diabetes model of rats. J Med Food. 2008;11(1):152–9. https://www.ncbi.nlm.nih.gov/pubmed/18361751[↩]
- Kadnur S. V, Goyal R. K. Beneficial effects of Zingiber officinale Roscoe on fructose induced hyperlipidemia and hyperinsulinemia in rats. Indian J Exp Biol. 2005;43(12):1161–4. https://www.ncbi.nlm.nih.gov/pubmed/16359128 [↩]
- Al-Amin Z. M, Thomson M, Al-Qattan K. K, Peltonen-Shalaby R, Ali M. Anti-diabetic and hypo- lipidaemic properties of ginger (Zingiber officinale) in streptozotocin-induced diabetic rats. Br J Nutr. 2006;96(4):660–6. https://www.ncbi.nlm.nih.gov/pubmed/17010224[↩]
- Ghayur M. N, Gilani A. H, Ahmed T, Khalid A, Nawaz S. A, Agbedahunsi J. M, Choudhary M. I, Houghton P. J. Muscarinic, Ca(+ +) antagonist and specific butyrylcholinesterase inhibitory activity of dried ginger extract might explain its use in dementia. J Pharm Pharmacol. 2008;60(10):1375–83. https://www.ncbi.nlm.nih.gov/pubmed/18812031[↩]
- El-Abhar H. S, Hammad L. N, Gawad H. S. Modulating effect of ginger extract on rats with ulcerative colitis. J Ethnopharmacol. 2008;118(3):367–72. https://www.ncbi.nlm.nih.gov/pubmed/18571884[↩]
- Ding M, Leach M, Bradley H. The effectiveness and safety of ginger for pregnancy-induced nausea and vomiting: a systematic review. Women and Birth. 2013;26(1):e26-e30. https://www.ncbi.nlm.nih.gov/pubmed/22951628[↩][↩]
- Ensiyeh J., Sakineh M.-A.C. Comparing ginger and vitamin B6 for the treatment of nausea and vomiting in pregnancy: A randomised controlled trial. Midwifery. 2009;25:649–653. doi: 10.1016/j.midw.2007.10.013[↩]
- Heitmann K, Nordeng H, Holst L. Safety of ginger use in pregnancy: results from a large population-based cohort study. European Journal of Clinical Pharmacology. 2013;69(2):269-277. https://www.ncbi.nlm.nih.gov/pubmed/22706624[↩][↩]
- Leach MJ, Kumar S. Int J Evid Based Healthc. 2008 Sep;6(3):311-20. doi: 10.1111/j.1744-1609.2008.00106.x. The clinical effectiveness of Ginger (Zingiber officinale) in adults with osteoarthritis[↩][↩][↩]
- Mozaffari‐Khosravi, H. , Naderi, Z. , Dehghan, A. , Nadjarzadeh, A. , & Fallah Huseini, H. (2016). Effect of ginger supplementation on proinflammatory cytokines in older patients with osteoarthritis: Outcomes of a randomized controlled clinical trial. Journal of Nutrition in Gerontology and Geriatrics, 35(3), 209–218. 10.1080/21551197.2016.1206762[↩]
- Naderi, Z. , Mozaffari‐Khosravi, H. , Dehghan, A. , Nadjarzadeh, A. , & Huseini, H. F. (2016). Effect of ginger powder supplementation on nitric oxide and C‐reactive protein in elderly knee osteoarthritis patients: A 12‐week double‐blind randomized placebo‐controlled clinical trial. Journal of Traditional and Complementary Medicine, 6(3), 199–203. 10.1016/j.jtcme.2014.12.007[↩]
- Niempoog, S. , Siriarchavatana, P. , & Kajsongkram, T. (2012). The efficacy of plygersic gel for use in the treatment of osteoarthritis of the knee. Journal of the Medical Association of Thailand = Chotmaihet Thangphaet, 95(Suppl 1), S113–S119.[↩][↩][↩]
- Yip, Y. B. , & Tam, A. C. Y. (2008). An experimental study on the effectiveness of massage with aromatic ginger and orange essential oil for moderate‐to‐severe knee pain among the elderly in Hong Kong. Complementary Therapies in Medicine, 16(3), 131–138. 10.1016/j.ctim.2007.12.003[↩]
- Chopra, A. , Saluja, M. , Tillu, G. , Sarmukkaddam, S. , Venugopalan, A. , Narsimulu, G. , … Patwardhan, B. (2013). Ayurvedic medicine offers a good alternative to glucosamine and celecoxib in the treatment of symptomatic knee osteoarthritis: A randomized, double‐blind, controlled equivalence drug trial. Rheumatology, 52(8), 1408–1417. 10.1093/rheumatology/kes414[↩]
- Maghbooli M., Golipour F., Moghimi Esfandabadi A., Yousefi M. Comparison between the efficacy of ginger and sumatriptan in the ablative treatment of the common migraine. Phytother. Res. 2014;28:412–415. doi: 10.1002/ptr.4996[↩]
- Martins L.B., Rodrigues A., Rodrigues D.F., Dos Santos L.C., Teixeira A.L., Ferreira A.V.M. Double-blind placebo-controlled randomized clinical trial of ginger (Zingiber officinale Rosc) addition in migraine acute treatment. Cephalalgia. 2019;39:68–76. doi: 10.1177/0333102418776016[↩]
- Pain Medicine Volume 16, Issue 12, December 2015; Pages 2243–2255. Efficacy of Ginger for Alleviating the Symptoms of Primary Dysmenorrhea: A Systematic Review and Meta-analysis of Randomized Clinical Trials. http://onlinelibrary.wiley.com/doi/10.1111/pme.12853/full[↩][↩]
- Kashefi, F. , Khajehei, M. , Tabatabaeichehr, M. , Alavinia, M. , & Asili, J. (2014). Comparison of the effect of ginger and zinc sulfate on primary dysmenorrhea: A placebo‐controlled randomized trial. Pain Management Nursing, 15(4), 826–833. 10.1016/j.pmn.2013.09.001[↩]
- Rahnama P., Montazeri A., Huseini H.F., Kianbakht S., Naseri M. Effect of zingiber officinale R. rhizomes (ginger) on pain relief in primary dysmenorrhea: A placebo randomized trial. BMC Complement. Altern. Med. 2012;12:92. doi: 10.1186/1472-6882-12-92[↩]
- Shirvani M.A., Motahari-Tabari N., Alipour A. Use of ginger versus stretching exercises for the treatment of primary dysmenorrhea: A randomized controlled trial. J. Integr. Med. 2017;15:295–301. doi: 10.1016/S2095-4964(17)60348-0[↩]
- National Cancer Institute. Ginger Helps Reduce Nausea from Chemotherapy. https://prevention.cancer.gov/news-and-events/news/ginger-helps-reduce-nausea[↩]
- Ryan JL, Heckler CE, Roscoe JA, et al. Ginger (Zingiber officinale) reduces acute chemotherapy-induced nausea: A URCC CCOP study of 576 patients. Supportive Care in Cancer. 2012;20(7):1479-1489. doi:10.1007/s00520-011-1236-3. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3361530/[↩][↩][↩][↩]
- Pillai AK, Sharma KK, Gupta YK, et al. Anti-emetic effect of ginger powder versus placebo as an add-on therapy in children and young adults receiving high emetogenic chemotherapy. Pediatric Blood & Cancer. 2011;56(2):234-238. http://onlinelibrary.wiley.com/wol1/doi/10.1002/pbc.22778/full[↩][↩]
- Ebrahimzadeh Attari V., Ostadrahimi A., Asghari Jafarabadi M., Mehralizadeh S., Mahluji S. Changes of serum adipocytokines and body weight following zingiber officinale supplementation in obese women: A RCT. Eur. J. Nutr. 2016;55:2129–2136. doi: 10.1007/s00394-015-1027-6[↩]
- Anh, N. H., Kim, S. J., Long, N. P., Min, J. E., Yoon, Y. C., Lee, E. G., Kim, M., Kim, T. J., Yang, Y. Y., Son, E. Y., Yoon, S. J., Diem, N. C., Kim, H. M., & Kwon, S. W. (2020). Ginger on Human Health: A Comprehensive Systematic Review of 109 Randomized Controlled Trials. Nutrients, 12(1), 157. https://doi.org/10.3390/nu12010157[↩][↩][↩]
- Aeschbach R, Loliger J, Scott B. C, Murcia A, Butler J, Halliwell B, Aruoma O. I. Antioxidant actions of thymol, carvacrol, [6]-gingerol, zingerone and hydroxytyrosol. Food Chem Toxicol. 1994;32(1):31–6. https://www.ncbi.nlm.nih.gov/pubmed/7510659[↩]
- Ahmad N, Katiyar S.K, Mukhtar H. Antioxidants in chemoprevention of skin cancer. Curr Probl Dermatol. 2001;29:128–39. https://www.ncbi.nlm.nih.gov/pubmed/11225193[↩]
- Topic B, Tani E, Tsiakitzis K, Kourounakis P. N, Dere E, Hasenohrl R. U, Hacker R, Mattern C. M, Huston J. P. Enhanced maze performance and reduced oxidative stress by combined extracts of zingiber officinale and ginkgo biloba in the aged rat. Neurobiol Aging. 2002;23(1):135–43. https://www.ncbi.nlm.nih.gov/pubmed/11755028[↩]
- Mallikarjuna K, Sahitya Chetan P, Sathyavelu Reddy K, Rajendra W. Ethanol toxicity: Rehabilitation of hepatic antioxidant defense system with dietary ginger. Fitoterapia. 2008;79(3):174–8. https://www.ncbi.nlm.nih.gov/pubmed/18182172[↩]
- Halvorsen B. L, editor. et al. A systematic screening of total antioxidants in dietary plants. J Nutr. 2002;132(3):461–71. https://www.ncbi.nlm.nih.gov/pubmed/11880572[↩]
- Kim H. W, Murakami A, Nakamura Y, Ohigashi H. Screening of edible Japanese plants for suppressive effects on phorbol ester-induced superoxide generation in differentiated HL-60 cells and AS52 cells. Cancer Lett. 2002;176(1):7–16. https://www.ncbi.nlm.nih.gov/pubmed/11790448[↩]
- Krishnakantha T. P, Lokesh B. R. Scavenging of superoxide anions by spice principles. Indian J Biochem Biophys. 1993;30(2):133–4. https://www.ncbi.nlm.nih.gov/pubmed/8394839[↩]
- Reddy A. C, Lokesh B. R. Studies on spiceprinciples as antioxidants in the inhibition of lipid peroxidation of rat liver microsomes. Mol Cell Biochem. 1992;111(1-2):117–24. https://www.ncbi.nlm.nih.gov/pubmed/1588934[↩]
- Ahmed R. S, Seth V, Banerjee B. D. Influence of dietary ginger (Zingiber officinales Rosc.) on antioxidant defense system in rat: Comparison with ascorbic acid. Indian J Exp Biol. 2000;38(6):604–6. https://www.ncbi.nlm.nih.gov/pubmed/11116533[↩]
- Ahmed R. S, Seth V, Pasha S. T, Banerjee B. D. Influence of dietary ginger (Zingiber officinales Rosc.) on oxidative stress induced by malathion in rats. Food Chem Toxicol. 2000;38(5):443–50. https://www.ncbi.nlm.nih.gov/pubmed/10762730[↩]
- Shobana S, Naidu K. A. Antioxidant activity of selected Indian spices. Prostaglandins Leukot Essent Fatty Acids. 2000;62(2):107–10. https://www.ncbi.nlm.nih.gov/pubmed/10780875[↩]
- Ahmed R. S, Suke S. G, Seth V, Chakraborti A, Tripathi A. K, Banerjee B. D. Protective effects of dietary ginger (Zingiber officinales Rosc.) on lindane-induced oxidative stress in rats. Phytother Res. 2008;22(7):902–6. https://www.ncbi.nlm.nih.gov/pubmed/18389491[↩][↩]
- El-Sharaky A. S, Newairy A. A, Kamel M. A, Eweda S. M. Protective effect of ginger extract against bromobenzene-induced hepatotoxicity in male rats. Food Chem Toxicol. 2009;47(7):1584–90. https://www.ncbi.nlm.nih.gov/pubmed/19371770[↩][↩]
- Ippoushi K, Azuma K, Ito H, Horie H, Higashio H. [6]-gingerol inhibits nitric oxide synthesis in activated J774.1 mouse macrophages and prevents peroxynitrite-induced oxidation and nitration reac-tions. Life Sci. 2003;73(26):3427–37. https://www.ncbi.nlm.nih.gov/pubmed/14572883[↩][↩]
- Ippoushi K, Ito H, Horie H, Azuma K. Mechanism of inhibition of peroxynitrite-induced oxidation and nitration by [6]-gingerol. Planta Med. 2005;71(6):563–6. https://www.ncbi.nlm.nih.gov/pubmed/15971130[↩]
- Uz E, Karatas O. F, Mete E, Bayrak R, Bayrak O, Atmaca A. F, Atis O, Yildirim M. E, Akcay A. The effect of dietary ginger (Zingiber officinals Rosc.) on renal ischemia/reperfusion injury in rat kidneys. Ren Fail. 2009;31(4):251–60. https://www.ncbi.nlm.nih.gov/pubmed/19462272[↩]
- Jagetia G. C, Baliga M. S, Venkatesh P, Ulloor J. N. Influence of ginger rhizome (Zingiber officinale Rosc.) on survival, glutathione and lipid peroxidation in mice after whole-body exposure to gamma radiation. Radiat Res. 2003;160(5):584–92. https://www.ncbi.nlm.nih.gov/pubmed/14565823[↩]
- Jagetia G, Baliga M, Venkatesh P. Ginger (Zingiber officinale Rosc.), a dietary supplement, protects mice against radiation-induced lethality: Mechanism of action. Cancer Biother Radiopharm. 2004;19(4):422–35. https://www.ncbi.nlm.nih.gov/pubmed/15453957[↩]
- Kim S. O, Chun K. S, Kundu J. K, Surh Y. J. Inhibitory effects of [6]-gingerol on PMA- induced COX-2 expression andactivation of NF-kappaB and p38 MAPK in mouse skin. Biofactors. 2004;21(1-4):27–31. https://www.ncbi.nlm.nih.gov/pubmed/15630166[↩]
- Young H. Y, Luo Y. L, Cheng H. Y, Hsieh W. C, Liao J. C, Peng W. H. Analgesic and anti-inflammatory activities of [6]-gingerol. J Ethnopharmacol. 2005;96(1-2):207–10. https://www.ncbi.nlm.nih.gov/pubmed/15588672[↩]
- Minghetti P, Sosa S, Cilurzo F, editors. et al. Evaluation of the topical anti-inflammatory activity of ginger dry extracts from solutions and plasters. Planta Med. 2007;73(15):1525–30. https://www.ncbi.nlm.nih.gov/pubmed/18058610[↩]
- Reginster J. Y, Gillot V, Bruyere O, Henrotin Y. Evidence of nutriceutical effectiveness in the treatment of osteoarthritis. Curr Rheumatol Rep. 2000;2(6):472–7. https://www.ncbi.nlm.nih.gov/pubmed/11123100[↩]
- Marcus D. M, Suarez-Almazor M. E. Is there a role for ginger in the treatment of osteoarthritis? Arthritis Rheum. 2001;44(11):2461–2. https://www.ncbi.nlm.nih.gov/pubmed/11710700[↩]
- Eldershaw T. P, Colquhoun E. Q, Dora K. A, Peng Z. C, Clark M. G. Pungent principles of gin-ger (Zingiber officinale) are thermogenic in the perfused rat hind limb. Int J Obes Relat Metab Disord. 1992;16(10):755–63. https://www.ncbi.nlm.nih.gov/pubmed/1330955[↩][↩]
- Ueki S, Miyoshi M, Shido O, Hasegawa J, Watanabe T. Systemic administration of [6]-gingerol, a pungent constituent of ginger, induces hypothermia in rats via an inhibitory effect on metabolic rate. Eur J Pharmacol. 2008;584(1):87–92. https://www.ncbi.nlm.nih.gov/pubmed/18295202[↩]
- Swain AR, Duton SP, Truswell AS. Salicylates in foods. J Am Diet Assoc 1985; 85: 950–60. https://www.ncbi.nlm.nih.gov/pubmed/4019987[↩]
- Mustafa T, Srivastava KC, Jensen KB. Drug development: report 9. Pharmacology of ginger, Zingiber officinale. J Drug Dev 1993; 6: 25–89.[↩]
- Kiuchi F, Iwakami S, Shibuya M, Hanaoka F, Sandawa U. Inhibition of prostaglandin and leukotriene biosynthesis by gingeroles and diarylheptanoids. Chem Pharm Bull (Tokyo) 1992; 40: 187–91.[↩]
- Mascolo N, Jain R, Jain SC, Capasso F. Ethnopharmacological investigation of Ginger (Zingiber officinale). J Ethnopharmacol 1989; 27: 129–40. https://www.ncbi.nlm.nih.gov/pubmed/2615416[↩]
- Jana U, Chattopadhayay RN, Shaw BP. Preliminary studies on anti-inflammatory activity of Zingiber officinale Rosc., Vitex negundo Linn. and Tinospora cordifolia (Willid) miers in albino rats. Indian J Pharmacol 1999; 31: 232–3[↩]
- Srivastava K. C, Mustafa T. Ginger (Zingiber officinale) in rheumatismand musculoskeletal disorders. Med Hypotheses. 1992;39(4):342–8. https://www.ncbi.nlm.nih.gov/pubmed/1494322[↩]
- Kiuchi F, Iwakami S, Shibuya M, Hanaoka F, Sankawa U. Inhibition of prostaglandin and leukotriene biosynthesis by gingerols and diarylheptanoids. Chem Pharm Bull (Tokyo) 1992;40(2):387–91. https://www.ncbi.nlm.nih.gov/pubmed/1606634[↩]
- Tjendraputra E, Tran V. H, Liu-Brennan D, Roufogalis B. D, Duke C. C. Effect of ginger constit-uentsand synthetic analogues on cyclooxygenase-2 enzyme in intact cells. Bioorg Chem. 2001;29(3):156–63. https://www.ncbi.nlm.nih.gov/pubmed/11437391[↩]
- Frondoza C. G, Sohrabi A, Polotsky A, Phan P. V, Hungerford D. S, Lindmark L. An in vitro screening assay for inhibitors of proinflammatory mediators in herbal extracts using human synoviocyte cultures. Vitro Cell Dev Biol Anim. 2004;40(3-4):95–101. https://www.ncbi.nlm.nih.gov/pubmed/15311968[↩]
- Tripathi S, Bruch D, Kittur D. S. Ginger extract inhibits LPS induced macrophage activation and function. BMC Complement Altern Med. 2008;8:1. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2234390/[↩][↩]
- Aktan F, Henness S, Tran V. H, Duke C. C, Roufogalis B. D, Ammit A. J. Gingerol metabolite and a synthetic analogue Capsarol inhibit macrophage NF-kappaB-mediated iNOS gene expression and enzyme activity. Planta Med. 2006;72(8):727–34. https://www.ncbi.nlm.nih.gov/pubmed/16732525[↩]
- Mozaffari-Khosravi H., Naderi Z., Dehghan A., Nadjarzadeh A., Fallah Huseini H. Effect of ginger supplementation on proinflammatory cytokines in older patients with osteoarthritis: Outcomes of a randomized controlled clinical trial. J. Nutr. Gerontol. Geriatr. 2016;35:209–218. doi: 10.1080/21551197.2016.1206762[↩]
- Kulkarni R.A., Deshpande A.R. Anti-inflammatory and antioxidant effect of ginger in tuberculosis. J. Complement. Integr. Med. 2016;13:201–206. doi: 10.1515/jcim-2015-0032[↩]
- Surh Y. J, Lee E, Lee J. M. Chemoprotective properties of somepungent ingredients present in red pepper and ginger. Mutat Res. 1998;402(1-2):259–67. https://www.ncbi.nlm.nih.gov/pubmed/9675305[↩]
- Surh Y. J. Molecular mechanisms of chemopreventive effects of selected dietary and medicinal phenolic substances. Mutat Res. 1999;428(1-2):305–27. https://www.ncbi.nlm.nih.gov/pubmed/10518003[↩]
- Surh Y. J. Anti-tumor promoting potential of selected spice ingredients with antioxidative and anti- inflammatory activities: A short review. Food Chem Toxicol. 2002;40(8):1091–7. https://www.ncbi.nlm.nih.gov/pubmed/12067569 [↩]
- Bode A. M, Dong Z. Ginger. Packer L, Ong C.N, Halliwell B. New York: Marcel Dekker; Herbal and Traditional Medicine: Molecular Aspects of Health. 2004[↩]
- Shukla Y, Singh M. Cancer preventive properties of ginger: A brief review. Food Chem Toxicol. 2007;45(5):683–90. https://www.ncbi.nlm.nih.gov/pubmed/17175086[↩]
- Aggarwal B. B, Kunnumakkara A. B, Harikumar K. B, Tharakan S. T, Sung B, Anand P. Potential of spice-derived phytochemicals for cancer prevention. Planta Med. 2008;74(13):1560–9. https://www.ncbi.nlm.nih.gov/pubmed/18612945[↩]
- Jiang Y., Turgeon D.K., Wright B.D., Sidahmed E., Ruffin M.T., Brenner D.E., Sen A., Zick S.M. Effect of ginger root on cyclooxygenase-1 and 15-hydroxyprostaglandin dehydrogenase expression in colonic mucosa of humans at normal and increased risk for colorectal cancer. Eur. J. Cancer Prev. 2013;22:455–460. doi: 10.1097/CEJ.0b013e32835c829b[↩]
- Citronberg J., Bostick R., Ahearn T., Turgeon D.K., Ruffin M.T., Djuric Z., Sen A., Brenner D.E., Zick S.M. Effects of ginger supplementation on cell-cycle biomarkers in the normal-appearing colonic mucosa of patients at increased risk for colorectal cancer: Results from a pilot, randomized, and controlled trial. Cancer Prev. Res. 2013;6:271–281. doi: 10.1158/1940-6207.CAPR-12-0327[↩]
- Ann M. Bode and Zigang Dong. Herbal Medicine: Biomolecular and Clinical Aspects. 2nd edition. Boca Raton (FL): CRC Press/Taylor & Francis; 2011. Chapter 7, The Amazing and Mighty Ginger. https://www.ncbi.nlm.nih.gov/books/NBK92775/ [↩]
- Vimala S, Norhanom A. W, Yadav M. Anti-tumour promoter activity in Malaysian ginger rhizobia used in traditional medicine. Br J Cancer. 1999;80(1-2):110–6. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2362999/[↩]
- Kapadia G. J, Azuine M. A, Tokuda H, Hang E, Mukainaka T, Nishino H, Sridhar R. Inhibitory effect of herbalremedies on 12 -O -tetradecanoylphorbol-13-acetate-promoted Epstein-Barr virus early antigen activation. Pharmacol Res. 2002;45(3):213–20. https://www.ncbi.nlm.nih.gov/pubmed/11884218[↩]
- Yagihashi S, Miura Y, Yagasaki K. Inhibitory effect of gingerol on the proliferation and invasion of hepatoma cells in culture. Cytotechnology. 2008;57(2):129–36. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2553670/[↩]
- Nakamura Y, Yoshida C, Murakami A, Ohigashi H, Osawa T, Uchida K. Zerumbone, a tropical ginger sesquiterpene, activates phase II drug metabolizing enzymes. FEBS Lett. 2004;572(1-3):245–50. https://www.ncbi.nlm.nih.gov/pubmed/15304356[↩][↩]
- Murakami A, Tanaka T, Lee J. Y, editors. et al. Zerumbone, a sesquiterpene in subtropical ginger, suppresses skin tumor initiation and promotion stages in ICR mice. Int J Cancer. 2004;110(4):481–90. https://www.ncbi.nlm.nih.gov/pubmed/15122579[↩][↩]
- Rhode J, Fogoros S, Zick S, Wahl H, Griffith K. A, Huang J, Liu J. R. Ginger inhibits cell growth and modulates angiogenic factors in ovarian cancer cells. BMC Complement Altern Med. 2007;7:44. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2241638/[↩]
- Dias M. C, Spinardi-Barbisan A. L, Rodrigues M. A, de Camargo J. L, Teran E, Barbisan L. F. Lack of chemopreventive effects of ginger on colon carcinogenesis induced by 1,2-dimethylhydrazine in rats. Food Chem Toxicol. 2006;44(6):877–84. https://www.ncbi.nlm.nih.gov/pubmed/16442687[↩][↩][↩][↩]
- Bidinotto L. T, Spinardi-Barbisan A. L, Rocha N. S, Salvadori D. M, Barbisan L. F. Effects of ginger (Zingiber officinale Roscoe) on DNA damage and development of urothelial tumors in a mouse bladder carcinogenesis model. Environ Mol Mutagen. 2006;47(8):624–30. https://www.ncbi.nlm.nih.gov/pubmed/16878317[↩]
- Nicoll R, Henein M. Y. Ginger (Zingiber officinale Roscoe): A hot remedy for cardiovascular disease? Int J Cardiol. 2009;131(3):408–9. https://www.ncbi.nlm.nih.gov/pubmed/18037515[↩][↩]
- Chang L. K, Whitaker D. C. The impact of herbal medicines on dermatologic surgery. Dermatol Surg. 2001;27(8):759–63. https://www.ncbi.nlm.nih.gov/pubmed/11493302[↩]
- Pribitkin E. D, Boger G. Herbal therapy: What every facial plastic surgeon must know. Arch Facial Plast Surg. 2001;3(2):127–32. https://www.ncbi.nlm.nih.gov/pubmed/11368667[↩]
- Heck A. M, De Witt B. A, Lukes A. L. Potential interactions between alternative therapies and warfarin. Am J Health Syst Pharm. 2000;57(13):1221–7. https://www.ncbi.nlm.nih.gov/pubmed/10902065[↩]
- Vaes L. P, Chyka P. A. Interactions of warfarin with garlic, ginger, ginkgo, or ginseng: Nature of the evidence. Ann Pharmacother. 2000;34(12):1478–82. https://www.ncbi.nlm.nih.gov/pubmed/11144706[↩]
- Weidner M. S, Sigwart K. The safety of a ginger extract in the rat. J Ethnopharmacol. 2000;73(3):513–20. https://www.ncbi.nlm.nih.gov/pubmed/11091007[↩]
- Jiang X, Williams K. M, Liauw W. S, Ammit A. J, Roufogalis B. D, Duke C. C, Day R. O, McLachlan A. J. Effect of ginkgo and ginger on the pharmacokinetics and pharmacodynamics of warfarin in healthy subjects. Br J Clin Pharmacol. 2005;59(4):425–32. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1884814/[↩]
- Ghayur M. N, Gilani A. H. Ginger lowers blood pressure through blockade of voltage-dependent calcium channels. J Cardiovasc Pharmacol. 2005a;45(1):74–80. https://www.ncbi.nlm.nih.gov/pubmed/15613983[↩]
- Fuhrman B, Rosenblat M, Hayek T, Coleman R, Aviram M. Ginger extract consumption reduces plasma cholesterol, inhibits LDL oxidation and attenuates development of atherosclerosis in atherosclerotic, apolipoprotein E-deficient mice. J Nutr. 2000;130(5):1124–31. https://www.ncbi.nlm.nih.gov/pubmed/10801908[↩]
- Bhandari U, Sharma J. N, Zafar R. The protective action of ethanolic ginger (Zingiber officinale) extract in cholesterol fed rabbits. J Ethnopharmacol. 1998;61(2):167–71. https://www.ncbi.nlm.nih.gov/pubmed/9683348[↩]
- Alizadeh-Navaei R, Roozbeh F, Saravi M, Pouramir M, Jalali F, Moghadamnia A. A. Investigation of the effect of ginger on the lipid levels. A double blind controlled clinical trial. Saudi Med J. 2008;29(9):1280–4. https://www.ncbi.nlm.nih.gov/pubmed/18813412[↩][↩]
- Verma S. K, Singh M, Jain P, Bordia A. Protective effect of ginger, Zingiber officinale Rosc. on experimental atherosclerosis in rabbits. Indian J Exp Biol. 2004;42(7):736–8. https://www.ncbi.nlm.nih.gov/pubmed/15339040[↩][↩]
- Tanabe M, Chen Y. D, Saito K, Kano Y. Cholesterol biosynthesis inhibitory component from Zingiber officinale Roscoe. Chem Pharm Bull (Tokyo) 1993;41(4):710–3. https://www.ncbi.nlm.nih.gov/pubmed/8508473[↩]
- Matsuda A, Wang Z, Takahashi S, Tokuda T, Miura N, Hasegawa J. Upregulation of mRNA of retinoid binding protein and fattyacid binding protein by cholesterol enriched-diet and effect of ginger on lipid metabolism. Life Sci. 2009;84(25-26):903–7. https://www.ncbi.nlm.nih.gov/pubmed/19379761[↩][↩]
- Kaul P. N, Joshi B. S. Alternative medicine: Herbal drugs and their critical appraisal-part II. Prog Drug Res. 2001;57:1–75. https://www.ncbi.nlm.nih.gov/pubmed/11727999[↩]
- Jewell D, Young G. Interventions for nausea and vomiting in early pregnancy. Cochrane Database Syst Rev. 2003;(4) CD000145. https://www.ncbi.nlm.nih.gov/pubmed/14583914[↩]
- Portnoi G, Chng L. A, Karimi-Tabesh L, Koren G, Tan M. P, Einarson A. Prospective compara-tivestudy of the safety and effectiveness of ginger for the treatment of nausea and vomiting in pregnancy. Am J Obstet Gynecol. 2003;189(5):1374–7. https://www.ncbi.nlm.nih.gov/pubmed/14634571[↩]
- Ann M. Bode and Zigang Dong. Herbal Medicine: Biomolecular and Clinical Aspects. 2nd edition. Boca Raton (FL): CRC Press/Taylor & Francis; 2011. Chapter 7, The Amazing and Mighty Ginger. https://www.ncbi.nlm.nih.gov/books/NBK92775[↩]