Ayahuasca
Ayahuasca also known by the names of hoasca, oasca, caapi, kahpi, daime (which means “give me” in Portuguese), yajé or yage, cipó, natema or natem, dapa, mihi, or vegetal is a plant-based psychedelic or hallucinogen. Psychedelics also known as “classic hallucinogens” or more specifically “serotonergic hallucinogens”, are a class of psychoactive substances that produce changes in all your senses, mood, thinking, sense of time and emotions 1, 2, 3. Psychedelics can cause a person to hallucinate, seeing or hearing things that do not exist or are distorted. Psychedelics are substances that exert their effects primarily by an agonist (or partial agonist) action on brain serotonin 5-hydroxytryptamine (5-HT2A) receptors 1. Ayahuasca is a concentrated liquid (decoction) made by prolonged heating or boiling of leaves of the Psychotria viridis bush commonly known as chacruna supplying the hallucinogen DMT (N,N-dimethyltryptamine) (Figure 2) and the stem and/or bark of the Banisteriopsis caapi vine commonly called ayahuasca rich in beta-carboline harmala alkaloids (Figure 3), although there can be a variety of other plants from 90 different plants inhabiting the Amazon rainforest included in the decoction for different traditional purposes 4, 5, 6, 7. For example, in Colombian Putumayo and Ecuador, the leaves of Diplopterys cabrerana also known as chaliponga are used instead of Psychotria viridis 8; in Peru, several plants are frequently added to the aqueous concoction of Banisteriopsis caapi and Psychotria species, most commonly Nicotiana tabacum (tobacco), Brugmansia spp., and Brunfelsia spp. 8, 9.
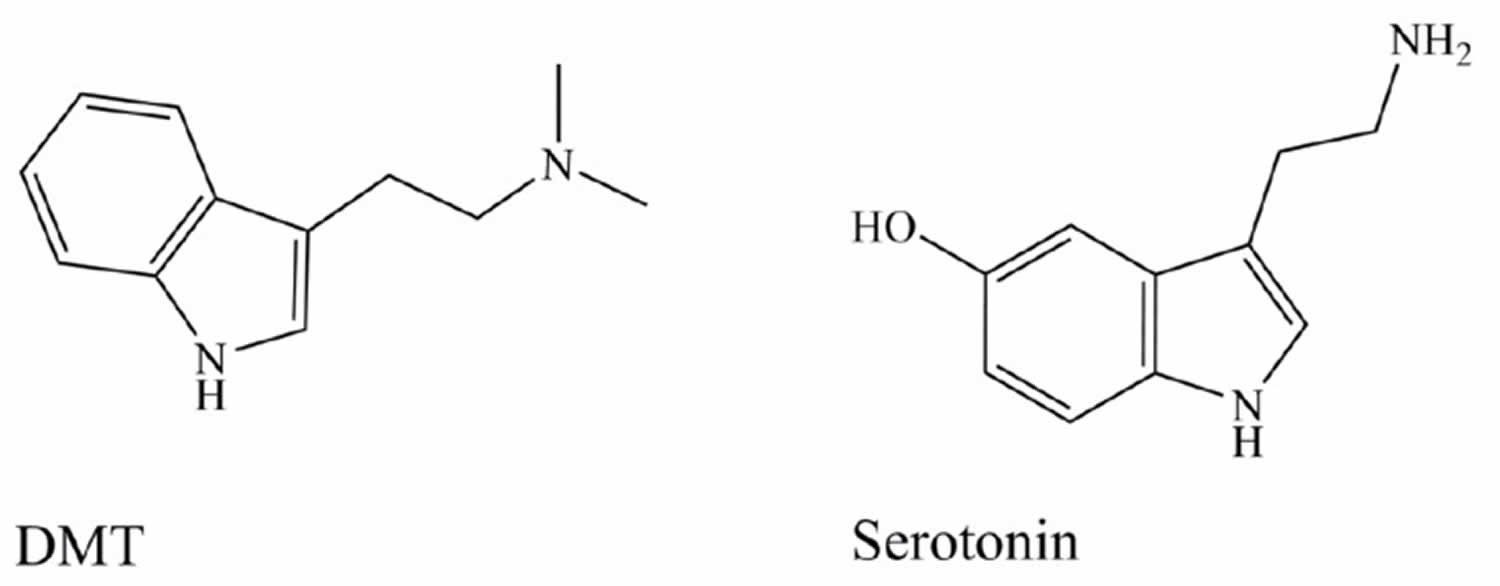
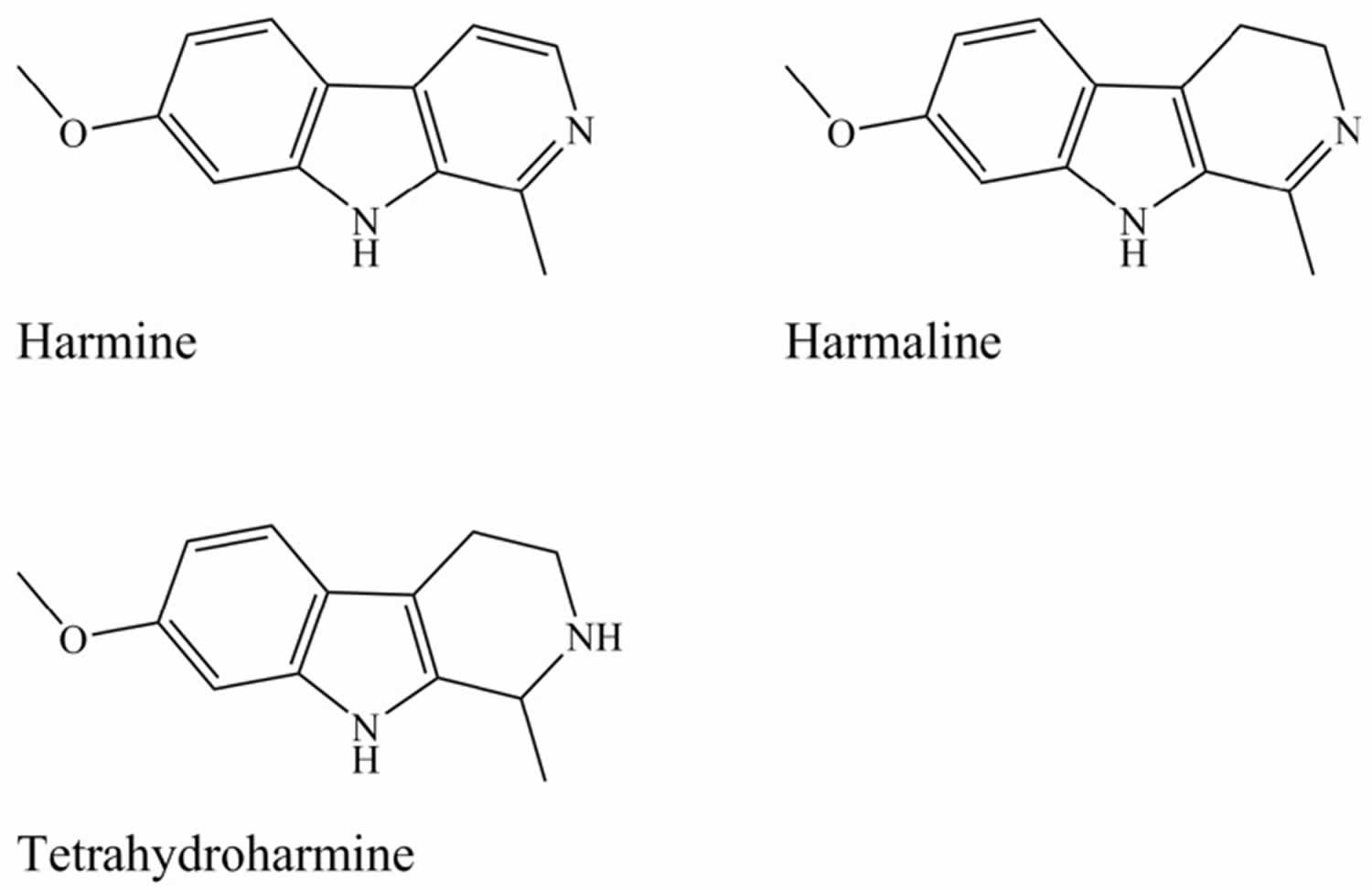
The main active alkaloids underlying the psychoactive effects of Ayahuasca were first characterised by Rivier and Lindgren 10. DMT (dimethyltryptamine) occurs in the leaves of Psychotria viridis bush 10, 11, while Banisteriopsis caapi vine stem and/or bark accumulates the harmala alkaloids also known as Beta-carbolines: harmine, harmaline, and tetrahydroharmine (see Figure 3) 12, 11, 13, 14.
The harmala alkaloids, harmine and harmaline, are monoamine oxidase inhibitors (MAOIs), without which the DMT (dimethyltryptamine) would be inactivated by your gut and liver monoamine oxidases (MAOs), allowing DMT (dimethyltryptamine) to reach your systemic circulation and your brain, where it activates serotonin 5-hydroxytryptamine 5-HT1A/2A/2C receptors in frontal and paralimbic areas 15, 16, 17. While tetrahydroharmine acts as a weak serotonin reuptake inhibitor without any MAOI action 18, 6. Ayahuasca is drunk as a liquid.
Ayahuasca means “vine of the soul” or “vine of the dead” in the Quecha language, is a traditional Amazonian decoction 19, 20, 21.
Ayahuasca can have varied effects depending on a person’s mood (often called the ‘set’) or the environment they are in (the ‘setting’) and can encompass a considerable degree of unpredictability 22:
- A person’s mood (the Set): a person’s state of mind, previous encounters with psychoactive drugs, and expectations of what’s going to happen 23. For example, feelings of anxiety or fear before using ayahuasca can be magnified and result in an unpleasant experience.
- The environment (the Setting): the environment in which someone consumes ayahuasca – whether it’s known and familiar, who they’re with, if they’re indoors or outdoors, the type of music and light 23. For example, using ayahuasca in a calm, quiet and relaxed environment can lead to a please experience, but being in a noisy, crowded place may result in a negative experience.
If you do decide to take ayahuasca, it’s important to consider the following.
- It is difficult to predict the strength and effects of ayahuasca. People can have very different experiences taking the same drug on different occasions.
- People with mental health conditions or a family history of these conditions should avoid using ayahuasca.
- Sometimes other drugs may be sold claiming to be ayahuasca that aren’t.
- Taking ayahuasca in an environment and with people where you feel safe may alleviate unpleasant emotional effects.
Use of ayahuasca is likely to be more dangerous when 24:
- taken in combination with alcohol or other drugs
- driving or operating heavy machinery
- judgement or motor coordination is required
- alone (in case medical assistance is required)
- the person has mental health issues.
Ayahuasca was originally used by Amazonian shamans in ritual ceremonies and by folk healers for a variety of psychosomatic complaints 25. The use of ayahuasca dates back to the earliest aboriginal inhabitants of the Amazonian basin, where it was used by indigenous shamans for communication with spirits, magical experiences, rites of initiation, and healing rituals 26. Ayahuasca was held in high regard among inhabitants of the Amazonian basin, particularly for religious and healing purposes 5. These were small private ceremonies where the patient and the shaman, and perhaps one or two others, would consume ayahuasca. Shortly after consumption, vomiting and often intense diarrhea occur 5. Vomiting results from increased serotonin (5-HT) stimulating the vagus nerve centrally, and diarrhea may be a result of excessive intestinal stimulation by serotonin (5-HT) peripherally 18. But after this, visions begin to appear, and the nature of the disease and curative plants are revealed to the shaman and the patient 27. Over the past several hundred years, the use of ayahuasca spread into Peru, Colombia, and Ecuador among indigenous Mestizo populations where it was integrated into folk medicine 26. These practices evolved during the early 1930s 28 for use as a religious ceremony or ritual in three Brazilian syncretic churches which combine indigenous and Christian traditions, the União do Vegetal (the largest, more meditative), the Santo Daime (the oldest, livelier, with music), and Barquinha (an Afro-Brazilian church), during twice monthly ceremonies lasting approximately four hours 20, 18, 29, 30, 15. Such religious groups use Ayahuasca both as a healing tool and as a way to “get in touch with the divine realm” 31. Ayahuasca therapy has also been used by witch doctors in treating addictions. For example, Lemlij 32 describes a group therapy model where participants come as many weeks as they need and may make a voluntary monetary contribution at the end.
In the last two decades the growing number of religious institutions as well as centers of alternative therapies that are allowed to use ayahuasca have led to a worldwide spread throughout the urban centers of South and North America, Europe, Asia, and Africa where Ayahuasca consumption have been used by users seeking a spiritual experience or a direct psychedelic effect 33, 34, 35, 36, 37, 38, 39. As such, Ayahuasca recreational use is sharply rising, with a global scale online survey showing an increased popularity on DMT consumption 40. In fact, a lot of foreigners travel to the Amazon also known as “ayahuasca tourism” to participate in ceremonial rituals organised by indigenous people 36. There is also an increased search of online “headshops” that legally trade-in ayahuasca ingredients 41, such as plants rich in DMT and harmala alkaloids, making them widely available to users.
However, DMT is illegal in most countries being classified as a Schedule 1 drug by the United Nations (UN) 1971 Convention on Psychotropic Substances 42. DMT is categorised as a Class A substance in the UK, as a Schedule 1 hallucinogenic substance by the Drug Enforcement Agency in the US and it is illegal to import, possess, sell, distribute, or consume ayahuasca 43, 40, 44, 45, as a Schedule 3 drug in Canada, a controlled substance in France and Portugal (the drug is included in the Table 2-A of the Decree Law no 15/93), and as a Schedule 1 drug under the German Narcotics Act; it is also banned in Japan 46, 47.
The harmala alkaloids are also regulated in a few countries, although not being subject to international control. In 2005, France added harmine, harmaline, tetrahydroharmine (THH), harmol, and harmalol to the list of controlled substances (Journal Officiel de la République Française no 102 du 3 mai 2005; NOR: SANP0521544A), being the only country in Europe where these substances are illegal. In Canada, harmalol and harmaline are also classified as Schedule III drugs under the 1996 Controlled Drugs and Substances Act 48.
On the other hand, the herbal products used for ayahuasca preparation, and known to contain Schedule 1 international controlled substances like DMT, are not subject to international control and have a lack of legal control in most countries 35, 42. An exception to this includes France, where non-licensed possession of P. viridis, B. caapi, P. harmala, and other plants containing DMT/harmala alkaloids have been banned since 2005 (Journal Officiel de la République Française no 102 du 3 mai 2005; NOR: SANP0521544A); and countries where all psychoactive compounds-containing plants are illegal, like Russia 49.
Lastly, Ayahuasca is potentially dangerous if taken by people with certain health conditions or taking certain medications, particularly ones that also release serotonin. Deaths have occurred 50, 51. People who should not take ayahuasca include those with 43, 52, 53:
- A family history of mental health issues
- Bipolar disorder
- Depression taking antidepressants
- Difficulty vomiting
- Epilepsy
- Fractures
- Gastrointestinal disorders
- Glaucoma
- Infection
- Recent surgery
- Retinal detachment
- Schizophrenia
- Seizures
- Serious liver, kidney, or gallbladder disease
- Severe cardiovascular disease
- Stroke
- Tuberculosis
- Parkinson’s disease
- Psychosis
- Uncontrolled or very high blood pressure
- Ayahuasca is not recommended for pregnant or lactating women
There is no safe level for ayahuasca and the use of this plant medicine always carries some risk. How ayahuasca affects you depends on:
- If you have taken it before
- Other medications you are taking
- The amount taken
- The strength of the decoction (varies from batch to batch)
- Your current state of health
- Weight
- Where you take it and the vibe.
Ayahuasca when consumed in unsupervised settings, the judgement of individuals can be compromised 43, leading to the belief of having superpowers or flying ability 54, having actions out of the ordinary like jump out of buildings 55 or staring at the sun during long periods of time resulting in eye damage 56, 57.
The recreational use of psychedelics often results in what is called “bad trips” 2, characterized by symptoms like anxiety, heart palpitations, and visual distortions 58. “Too little” DMT was also associated with unpleasant feelings, not allowing the consumer to achieve the desired development of the characteristic perceptual effects, only giving them a tensely dysphoric state 59. However, it is sometimes difficult to assess the potential hazardous effects caused by the recreational consumption of DMT, since the drug is commonly used in combination with other illicit substances, such as psychostimulants, depressants, narcotics, cannabis, and alcohol 60. By virtue of MAO inhibition, severe adverse effects can occur when ayahuasca or the beta-carbolines alone are used concomitantly with selective serotonin reuptake inhibitors (SSRIs) such as antidepressants 61, as this combination leads to accumulation of serotonin (5-HT) at neuron synapses, resulting in a potentially fatal condition known as serotonin syndrome. Serotonin syndrome is a potentially life-threatening drug reaction 62, 63. Serotonin syndrome is caused by medications that build up high levels of serotonin in your body. Too much serotonin causes signs and symptoms that can range from mild shivering and diarrhea to severe muscle rigidity, fever and seizures 62, 63. Milder forms of serotonin syndrome may go away within a day or two of stopping the medications that cause symptoms and, sometimes, after taking drugs that block serotonin 62. Severe serotonin syndrome can cause death if not treated 62. Other compounds that may have serotonergic effects (e.g., lithium and triptans through the activation of serotonin receptors, levodopa through the increase in serotonin release) can also precipitate this fatal condition and should be avoided with ayahuasca 64, 65.
Vomiting and diarrhea are often reported as adverse effects resulting from ayahuasca consumption 66, which may be due to increased central serotonin (5-HT) stimulation of the vagus nerve and peripheral stimulation of the intestine 67. However, in a study conducted by Sanches et al. 68 in patients with depression, the vomiting effect was not considered as a cause of severe discomfort. Nausea and exhaustion are also commonly mentioned as side-effects. All of these effects are, however, considered to be transient, only persisting for one or two days, and easily manageable 69.
Traditional ayahuasca experiences are hosted by a shaman or curandero who prepares and concentrates the brew according to traditional methods and his experience. But there is no regulation of the industry and commercialization has taken over with money-grabbing imposters prevalent and hard to distinguish among legitimate ayahuasca centers. These may not have the same sense of ethics as some of the more genuine retreat centers or deliver the same experience, and at least 11 tourist deaths over the past decade have been associated with being part of an ayahuasca experience.
Ayahuasca should only be taken under the supervision of an experienced shaman or curandero because a trip can change your state of consciousness for several hours and you need to be kept safe.
Figure 1. Ayahuasca plant sources
Footnote: (A) Psychotria viridis bush commonly known as chacruna supplying the hallucinogen DMT (N,N-dimethyltryptamine) and (B & C) Banisteriopsis caapi bark and stem rich in beta-carboline harmala alkaloids, are the most common plant species used in ayahuasca preparations (decoction).
[Source 48 ]Figure 2. Dimethyltryptamine (DMT) and serotonin (5-HT)
[Source 5 ]Figure 3. Beta-carbolines (harmine, harmaline and tetrahydroharmine)
[Source 5 ]What does ayahuasca contain?
Ayahuasca is traditionally made using stem and/or bark of the Banisteriopsis caapi vine and leaves of the Psychotria viridis bush, both of which have hallucinogenic properties.
The main active ingredient, DMT (N, N-dimethyltryptamine) is a hallucinogen (psychedelic substance) that occurs naturally in Psychotria viridis. But DMT is easily broken down by monoamine oxidase (MAO) enzymes in the stomach, small intestine, and liver. This is why ayahuasca also contains Banisteriopsis caapi. Banisteriopsis caapi contains monoamine oxidase inhibitors (MAOIs) which in addition to having their own psychoactive effects, also stop DMT from being broken down by monoamine oxidase enzymes in the liver and gastrointestinal tract. This combination allows DMT to be absorbed and makes a powerful psychedelic drink.
What happens during an Ayahuasca experience?
If you are considering trying ayahuasca, always choose an experienced center with a guide you trust. Ayahuasca is not recommended for some people, and your guide should ask you to complete a questionnaire that should determine if it is safe for you to proceed with the experience.
Participants are asked to purify their bodies by abstaining from alcohol, caffeine, cigarettes, drugs, and sex for several weeks before a traditional ayahuasca experience. A healthy diet, preferably vegetarian should be followed for several days before the experience and the participant should be well-hydrated and in good physical health. The last meal should be eaten about 6 hours before ingesting ayahuasca.
What does an ayahuasca experience feel like?
Ayahuasca experiences tend to be a combination of both positive and negative effects, and some of the negative effects can be distressing. They are usually held at night and last until the effects of ayahuasca have worn off.
The effects usually come on within 20 to 60 minutes and can last 2 to 6 hours. Positive effects may include:
- An awareness of yourself and the way you look at yourself (introspection)
- Altered state of consciousness
- Connection with mythological, religious, or spiritual dimensions beyond what is usually experienced
- The emergence of memories and images from the past
- Feelings of euphoria
- Intense visual and auditory hallucinations
- Out-of-body experiences.
The emotional intensity related to these experiences is usually much greater than normal. Some describe the experience as a mirror of their own interior which can make you question your most fundamental assumptions about who you are and what the world is.
Most people also experience negative effects, such as diarrhea and vomiting, although you may have been told this is considered a normal part of the ayahuasca experience. Other negative effects that may alternate with the positive effects or become the main experience can include:
- Anxiety
- Confusion
- Dizziness
- Fainting
- Fear
- Panic
- Paranoia
- Psychosis.
Ayahuasca can also cause a moderate increase in blood pressure and heart rate and increased body temperature.
How does Ayahuasca work?
Ayahuasca is a concentrated liquid (decoction) made by prolonged heating or boiling of the stem and/or bark of the Banisteriopsis caapi vine rich in beta-carboline harmala alkaloids and leaves of the Psychotria viridis bush supplying the hallucinogen DMT (N,N-dimethyltryptamine), although there can be a variety of other plants from 90 different plants inhabiting the Amazon rainforest included in the decoction for different traditional purposes 4, 5, 6, 7. For example, in Colombian Putumayo and Ecuador, the leaves of Diplopterys cabrerana also known as chaliponga are used instead of Psychotria viridis 8; in Peru, several plants are frequently added to the aqueous concoction of Banisteriopsis caapi and Psychotria species, most commonly Nicotiana tabacum (tobacco), Brugmansia spp., and Brunfelsia spp. 8, 9.
The main active alkaloids underlying the psychoactive effects of Ayahuasca were first characterised by Rivier and Lindgren 10. DMT (dimethyltryptamine) occurs in the leaves of Psychotria viridis bush 10, 11, while Banisteriopsis caapi vine stem and/or bark accumulates the harmala alkaloids also known as Beta-carbolines: harmine, harmaline, and tetrahydroharmine (see Figure 3) 12, 11, 13, 14.
The harmala alkaloids, harmine and harmaline, are monoamine oxidase inhibitors (MAOIs), without which the DMT (dimethyltryptamine) would be inactivated by your gut and liver monoamine oxidases (MAOs), allowing DMT (dimethyltryptamine) to reach your systemic circulation and your brain, where it activates serotonin 5-hydroxytryptamine 5-HT1A/2A/2C receptors in frontal and paralimbic areas 15, 16, 17. While tetrahydroharmine acts as a weak serotonin reuptake inhibitor without any MAOI action 18, 6.
DMT (N,N-dimethyltryptamine) is a serotonin-like hallucinogen structurally resembling other indolealkylamines, including melatonin and psychedelic tryptamines such as psilocybin, and is known mostly for its activity as a serotonin 5-hydroxytryptamine (5-HT2A) receptor agonist 70, 71. DMT is found in fungi, marine sponges, tunicates, frogs, legumes, and grasses 72 and has been reported to be formed endogenously in human and rat brains 73 as well as to be found in human urine, blood, and CSF. DMT has affinity for 5-HT1A/1B/1D/2A/2B/2C/6/7 receptors, with proven partial agonist activity at the 5-HT1A/2A/2C receptors 74, 75, 76, 77. Carbonaro et al. 78 proposed that the mGluR2 glutamate receptors may have some involvement in DMT’s hallucinogenic effect. Current understanding is that psychedelic effects are mediated mainly by 5-HT2A/2C receptors. 5-HT2A receptor activation has also been associated with sympathetic activation which may explain some of the physiologic effects of ayahuasca administration 79, 80.
DMT activity was demonstrated at the rat trace amine-associated receptor 1 (TAAR1) by Bunzow et al. 81, where tryptamine is also thought to act as a neurotransmitter 72. Premont et al. 82 suggested that DMT may function endogenously as part of this system, and proposed that trace levels of endogenous DMT act to produce a calmer, more relaxed mental state and suppress psychosis.
DMT binds to sigma-1 receptors with a moderate affinity, and Fontanilla et al. 76 proposed that DMT is likely to serve as an endogenous sigma-1 receptor ligand. The function of this receptor is not well understood, although it is found in lung, prostate, colon, ovaries, breasts, and liver, and is most concentrated in the brain. This receptor may play a role in depression, anxiety, and cancer 83. Sigma-1 receptors are molecular chaperones situated on the mitonchondria-associated endoplasmic reticulum membrane, although when stimulated with high concentrations of ligands, may translocate to the cell’s plasma membrane, where they inhibit several ion channels. DMT has also shown affinity for α1- and α2-adrenergic receptors as well as the dopamine D1 receptor 84.
DMT is thought to have dose-dependent effects in humans, visual hallucinations predominating at high doses, while stimulant effects are mostly noted at low doses 59. After acute ayahuasca ingestion, psychoactive effects such as intense perceptual and cognitive changes, somatic effects, increased emotional liability, and positive mood are rapidly felt, being resolved within a maximum of 4 to 6 hours 85, 86, 87. Such consumption also results in moderate and transitory cardiovascular, autonomic, neuroendocrine, and immunological effects, being well tolerated when consumed by healthy individuals.
A cycle of experiences designated as the “transcendental circle” by Kjellgren et al. 88 are consistent among different individuals, following ayahuasca consumption. Changes in perceptions, visual field vibrating, and users feeling vulnerable are noted 30 min after ingestion, an experience known as the visionary state. Following this, terrifying feelings of confusion, paranoia, and fear can be experienced, which might be accompanied by nausea or vomiting. Then, participants usually mention contact with a spiritual world, characterised by feelings of oneness with the universe, profound peace, and ecstasy, and they are given lessons by spirit entities. The last phase involves fatigue and fading visuals 35. While participants are still able to speak and are aware of their environment during these experiences 22, the perception of time can be altered 87.
An ayahuasca experience resembles schizophrenic episodes; however, whether or not this herbal preparation or DMT are involved in a psychotic crisis is still a matter of debate. While some authors report that high levels of endogenous DMT are found in the urine and blood of individuals during a schizophrenic episode 89, 90, others found no significant difference in DMT levels between schizophrenic patients and normal individuals 91. It has been hypothesized that DMT is a homeostatic agent with psychotic suppression activity 92. Although having a resemblance, ayahuasca-DMT mainly produces visual hallucinations in healthy individuals, while auditory hallucinations are predominant in schizophrenia patients 66. The few cases where psychotic episodes occurred were found to be transient in nature and resolved spontaneously 22, 93. Case-control and cross-sectional studies with experienced Ayahuasca consumers revealed that this hallucinogenic herbal concoction is safe, with rare adverse effects, and not associated with psychopathological nor neuropsychological deficits 22, 39, 94.
Short-term emotional distress can be a psychological consequence of DMT or ayahuasca use. Development of long-lasting psychosis is infrequent, mostly occurring in individuals after the concomitant use of other drugs, personal or family history of psychosis/non-psychotic bipolar disorders, as well as ongoing psychotic or maniac symptomatology 95. In controlled clinical settings, factors that could predispose long-term psychological adverse effects are screened prior to administration of ayahuasca or DMT, their consumption being exceptionally safe in this scenario 95. However, when ayahuasca is administered outside clinical settings or established ceremonial rituals, severe and unpredictable adverse psychological reactions that remain to be elucidated can be triggered 96.
Beta-carbolines (Figure 3) are tricyclic indole alkaloids resembling tryptamines 28. 6-Methoxytetrahydro-beta-carboline has been found in the human pineal gland 97. Several beta-carbolines are found in the Banisteriopsis caapi vine, including harmine, harmaline, and tetrahydroharmine. The first two act as selective and reversible monoamine oxidase A inhibitors (MAO-AIs), while tetrahydroharmine acts as a weak serotonin reuptake inhibitor without any monoamine oxidase A inhibitor action 18. Beta-carbolines are found naturally in wheat, rice, corn, barley and throughout different body tissues 98. They elicit their effects through several mechanisms.
Beta-carbolines without DMT have been shown to produce psychological and physiologic effects, as in a case of intoxication following Paganum harmala seed extract 99. The effects, including nausea, vomiting 78, hallucinations, ataxia, confusion, and agitation were attributed to CNS stimulation by MAOI activity as well as the serotonin reuptake inhibition by tetrahydroharmine. Frison et al. 99 suggested that the hallucinogenic effects could be a result of the affinity of harmine and harmaline for 5-HT receptors. Beta-carbolines from the B. caapi vine taken without DMT are used by the Piaroa of Southern Venezuala. Piaroa shamans and people who use B. caapi describe enhanced empathy, stimulant-like effects, and increased visual acuity, and they also use it as a hunting aid 100, 101.
The main mechanisms of action proposed for beta-carbolines include the MAO-A inhibitory activity, dopamine efflux, and affinity for the serotonin 5-hydroxytryptamine (5-HT2A) binding site 102. Other less studied mechanisms include dopamine transporter (DAT) inhibition at high concentrations in particular of beta-carboline compounds 103, harmine as a specific tyrosine-phosphorylation-regulated kinase 1A (DYRK1A) inhibitor 104 and affinity for the imidazoline (I2) binding site 105. Harmine has also been found to regulate expression of the peroxisome proliferator-activated receptor gamma (PPAR-ɣ, also known as the glitazone receptor) and shows some antitrypanosomal activity 106. Harmine upregulates the glutamate transporter (GLT-1, also called excitatory amino acid transporter 2, or EAAT2), the primary mechanism for inactivating synaptic glutamate 107. Harmane, harmine, and norharmane have also been found to act as inverse agonists at the benzodiazepine binding site (between the α and ɣ subunits) of the GABA-A receptor 108, dopamine (D(2)) and benzodiazepine receptors. Drug Alcohol Depend. 2000 Aug 1;60(2):121-32. doi: 10.1016/s0376-8716(99)00148-9)). Another study showed that 4 beta-carbolines (1,2,3,4-tetrahydronorharmane, norharmane, harmane, and 6-methoxyharmalan) act as competitive antagonists at the glycine receptor ligand binding site, leading to inhibition at the glycine receptor 109.
A recent study on neurotransmitter concentrations in the amygdala and hippocampus of rats killed 40 minutes after administration of an ayahuasca infusion reported that ayahuasca reduced levels of glycine and GABA in rat amygdala 110. This is suggestive of an increased release of these neurotransmitters in the amygdala, leading to greater inhibition, while in the hippocampus, it increased GABA levels, suggestive of a decrease in GABA release and excitation at this structure 110. These opposite effects on the level of inhibitory neurotransmission in these two limbic structures may provide some explanation into the behavioural effects of ayahuasca, due to the importance of these structures in neural pathways involved in memory, learning, and emotion.
Harmine and harmaline affect dopamine pathways both by causing a significant increase in dopamine release from striatal cells and by acting as reversible MAO-A inhibitors. A study on the nucleus accumbens of rats 111 found that harmine increases electrically evoked dopamine efflux in the nucleus accumbens shell. Brierley & Davidson 102 proposed that, given harmine has some affinity for the 5-HT2A/2C receptors but not for the dopamine receptor 112, this effect has a 5-HT2A-mediated mechanism. Grella et al. 113 also found that certain beta-carbolines bind at the 5-HT2 receptor. In another experiment in rat striatum, dose dependent decreases in the levels of dopamine metabolites 3,4-dihydroxyphenylacetic acid (DOPAC) and homovanillic acid (HVA) were also seen, as well as decreases in levels of the serotonin breakdown product 5-hydroxyindoleacetic acid (5-HIAA), though not to the same extent as with the dopamine metabolites 114. These effects are additive when mixtures of various beta-carbolines are used 115. The dopamine transporter serves to actively shift dopamine from the synapse into the presynaptic neuron, acting as the primary mechanism for regulating dopaminergic activity 116. Harmine was found to inhibit dopamine uptake through the dopamine transporter (DAT) in rats. Dopaminergic neurotransmission is primarily modulated through regulation of the dopamine transporters, which act to shuttle extracellular dopamine back into the neurons. Harmine acts a potent ATP-competitive inhibitor of the DYRK1A enzyme, which inhibits synaptic vesicle endocytosis and dopamine transporter (DAT) membrane trafficking, possibly by phosphorylating vesicle proteins taking part in clathrin-mediated endocytosis that serves to regulate dopamine transporter trafficking 117, 118. DYRK1A overexpression has been implicated in defective neural development in Down Syndrome, and the protein has also been implicated in amyloid pathology as well as in tau protein phosphorylation (at serine 262/356/396) in both Down Syndrome and Alzheimer’s disease 119.
Ayahuasca was found to upregulate platelet serotonin transporters 120. The glutamate transporter GLT-1/EAAT2 is the main mechanism for extracellular glutamate uptake in the brain, and dysfunction may lead to excessive synaptic glutamate and excitotoxicity. Harmine has been found to activate the GLT-1 gene promoter, leading to increased gene expression and greater extracellular glutamate uptake 107. Certain beta-carbolines have been found to bind at imidazoline binding sites, including harmine and harmaline found in ayahuasca 105. Husbands et al. 105 suggested that the imidazoline type 2 (I2) receptors may play a role in the hallucinogenic nature of ayahuasca given that harmine and harmaline have much higher affinities for the I2 receptor than the 5-HT2A receptor. Harmine also increases superoxide dismutase and catalase activity, and these antioxidant effects may have relevance in depression and several neurodegenerative disorders 121.
In a study of brain blood flow using single photon emission computed tomography (SPECT) with 15 male volunteers, Riba et al. 16 reported that ayahuasca produced an activation of frontal and paralimbic brain regions and increased blood perfusion in both the anterior insula; greater intensity was observed in the right hemisphere and in the anterior cingulate and frontomedial cortex of the right hemisphere (areas involved in somatic awareness, subjective feelings, and arousal of emotion). Additional increases were observed in the left amygdala and parahippocampal gyrus, a structure also involved in emotional arousal. Sanches et al. 122 reported increased blood perfusion in the left nucleus accumbens, right insula, and left subgenual area 8 hours after ayahuasca ingestion and that ayahuasca was well tolerated. Riba et al. 16 indicated that these findings suggest an interaction of Ayahuasca with neural systems is important in introspection and processing of emotion and imply a modulatory role of serotonergic neurotransmission in these processes.
Ayahuasca causes a statistically significant increase in activation of many occipital, temporal, and frontal areas, including the primary visual area on magnetic resonance imaging (MRI) during closed eye imagery 17. Even with eyes closed, on Ayahuasca the levels of activation in the visual area were consistent with seeing a natural image. This action was seen in both the occipital cortex, which includes Brodmann areas 17, 18, 19, all involved in vision 17. Brodmann area 17 has also been correlated with perceptual changes and psychotic effects such as hallucinations. Areas involved in episodic memory were also activated, including the parahippocampal gyrus (Brodmann area 30) and the middle temporal cortex (Brodmann area 37) 17. The frontal cortex (Brodmann area 10) is also activated. Emotions and memories were intensified and past experiences were seen through vivid imagery, which gave the whole experience a “status of reality” 17. The posterior cingulate cortex is key component of the default mode network, a group of neural pathways involved in inwardly focussed thought, conception and awareness of self, remembering the past and envisioning the future. Bousa et al. 123 found an inverse correlation between cortical thickness in the posterior cingulate cortex and intensity and duration of previous ayahuasca use, as well as scores on a personality trait called self-transcendence, a leaning toward spirituality and religiosity and suggested that regular use of psychedelic drugs could result in structural changes in brain areas involved in attentional processes, self-referential thought, and internal mentation. Ayahuasca caused decreased activity in the default mode network, and also decreased connectivity between various components of the default mode network on functional magnetic resonance imaging (fMRI) 124.
Endocrine System effects
Callaway et al. 18 reported both growth hormone (GH) and prolactin increasing but returning to baseline by 360 minutes, and cortisol increasing to a maximum at 60 minutes, and dipping below basal levels at 360 minutes. Growth hormone (GH) and prolactin are also influenced by the serotonergic system, so their findings fit with other studies showing an increase in prolactin levels with DMT and other serotonergic drugs such as MDMA, fenfluramine, and citalopram 125, 126.
Immune System effects
Dos Santos et al. 127 found that relative to placebo, ayahuasca increased total lymphocytes at 1.5 hours, and decreased them at 4.5 hours compared to placebo and to amphetamine, although there was no difference at 24 hours. There were significant decreases in both CD3 and CD4 lymphocytes at 1.5 and 2 hours, no significant changes in CD8 and CD19 lymphocytes, and significant increases in natural killer (NK) cells at 1.5 and 2 hours compared to placebo. No tolerance or sensitization was found with repeat doses 128. Davydova et al. 129 and dos Santos 130 highlighted previous findings and postulated that DMT may activate peripheral 5-HT2A receptors on leukocytes with impacts on cytokine secretion and cell differentiation, and that increased glucocorticoid levels may have modulatory or inhibitory effects on immunity. Amphetamine and MDMA both induce changes similar to ayahuasca, with decreases in CD3 and CD4 lymphocyte levels and increases in NK cell levels 127. Frecska et al. 131 found that DMT caused significantly increased levels of secreted interferon-β and interferon-γ in cultured human NK cells, and suggested that DMT’s action at the sigma-1 receptor could be the mechanism for this effect. In an in vitro study on human primary monocyte-derived dendritic cells, DMT and 5-MeO-DMT reduced production of several pro-inflammatory cytokines (IL-1β, IL-6, TNF-α) and chemokine IL-8, while they increased the secretion of the antiinflammatory cytokine IL-10 132, 133. The authors found that these effects were mediated through the sigma-1 receptor, and also noted that both DMT and 5-MeO-DMT impaired the ability of T helper 1 and T helper 17 cells to activate immune responses. House et al. 134 noted that harmaline caused a dose-related suppression of CD8 activity, IL-2 and IL-4 production, B cell proliferation, and NK cell function.
Pupil Size and Body Temperature effects
DMT causes dose-dependent elevations in pupil size 128, 135. Callaway et al. 18 reported that pupillary diameter increased to a maximum of 4.9 ± 0.2 mm at 180 minutes, and returned to normal by 360 minutes. Mydriasis has been demonstrated in several intravenous DMT studies 135, 136. Mean pupillary light reflex (PLR) amplitude was reduced and PLR latency was increased significantly compared to placebo 127. A reduced PLR amplitude and increased PLR latency is typically associated with anticholinergics. Two studies found that the serotonin-noradrenaline reuptake inhibitor (SNRI) venlafaxine has the same effect, and they interpreted this as noradrenergic inhibition of parasympathetic transmission on the Edinger-Westphal nucleus, responsible for iris constriction 137, 138.
With respect to body temperature, dos Santos et al. 127 compared DMT (at a dose of 1 mg DMT/kg body weight), amphetamine, and placebo, and found that with placebo, body temperature steadily increased over the day, whereas both DMT and amphetamine caused a statistically significant decrease in body temperature during the first hour, followed by a gradual increase, which was larger for amphetamine. Studies involving IV DMT have shown inconsistent results, with one study showing increases and three others showing no change or ambiguous results 79, 136, 138.
Cardiovascular effects
In a study of 18 volunteers, Riba et al. 15 showed maximum increases in diastolic blood pressure (DBP) of approximately 10 mmHg at 15 minutes, and a maximal systolic BP (SBP) rise of approximately 8 mmHg at 75 minutes following ingestion of ayahuasca containing a 0.85 mg/kg dose of DMT. With respect to heart rate, the maximum increase was approximately 5 beats per minute (BPM) at 60 minutes. Only two of the 18 volunteers had a systolic BP over 140 (maximum 146) at any point in time, and two volunteers had a diastolic blood pressure over 90 125. The same volunteer with the high systolic BP and diastolic blood pressure had a heart rate of 101 at 60 minutes. Callaway et al. 18 found maximal increases in blood pressure (BP) at 40 minutes, 11 mmHg for systolic BP and 9 mmHg for diastolic blood pressure. Heart rate, at its maximal increase, was 7 beats per minute (BPM) above baseline at 20 minutes (79 BPM), decreased to a low of 7 BPM below baseline by 120 minutes (65 BPM), then returned toward baseline by 240 minutes. The return to baseline may be due to increasing levels of central serotonin (5-HT), mediating cardiac response through the vagus nerve 18. Another study demonstrated significant increases in heart rate (HR), systolic BP, and diastolic blood pressure relative to placebo, with a maximum HR of 150 and systolic BP of 147 mmHg, while no diastolic blood pressure values went above 90 mmHg 128.
Strassman and Qualls 79 found dose-dependent elevations in HR and BP with IV DMT. They found a larger and more rapid increase than with oral ingestion, reporting that a 0.4 mg/kg IV dose raised HR by approximately 26 BPM at 2 minutes, as well as systolic BP by 35 mmHg and diastolic blood pressure by 30 mmHg. In the same study, peak heart rates were approximately 150 BPM while some were no higher than 95 BPM. Gable 7 analyzed several studies to compare changes in HR, systolic BP, and diastolic blood pressure brought on by various psychoactive substances, and concluded that the hemodynamic effects of ayahuasca appear less hazardous than IV DMT, oral alcohol, insufflated cocaine, smoked marijuana, and oral MDMA.
As with any substance that causes acute hemodynamic changes, some adverse cardiac events are possible with the use of ayahuasca 7, although such minimal increases could be attributed to changes in physical activity or other reasons, along the same lines as suggested by Hartley et al. 139, who concluded that even just an anxiety-provoking stimulus increases these values more than caffeine; after 14 days of chronic administration, Pitol et al. 140 found flattening and stretching of vascular smooth muscle cells, and significant increases in media thickness as well as the ratio of the media thickness to the lumen diameter.
Psychiatric Symptoms
There is conflicting information on whether endogenous DMT levels are higher in psychotic disorders, and research thus far has been inconclusive 72. Checkley et al. 135 suggested that levels are higher during psychosis but normal after recovery, while Gillin et al. 141 argued that DMT levels do not differ significantly between schizophrenics and normal controls, and that DMT does not mimic symptoms of schizophrenia. Another theory proposed that DMT may even serve to suppress psychotic activity, acting as a homeostatic agent 72. Based on rates of psychotic episodes in the UDV, Gable 7 also concludes that ayahuasca is not a trigger for sustained psychosis. While ayahuasca and other psychedelics could precipate psychosis in predisposed individuals, rates of psychosis in the UDV are comparable to the general population in Brazil 142. Paterson et al. 143 provided a case report of a 42-year-old male without significant psychiatric history who presented with substance-induced psychosis in the context of recent and repeated DMT use as well as long term cannabis use. He improved with quetiapine, divalproex, and hydroxyzine. Warren et al. 144 also suggested that recreational DMT use could be a contributor to psychosis. Another case report discussed a man with preexisting bipolar disorder who had a manic episode following ayahuasca consumption 145.
Short Term Psychological Effects
The ayahuasca experience begins approximately 40 minutes following ingestion, peaking between 60 and 120 minutes, with subjective effects fading by approximately 4 hours. Mabit 21 reports that ayahuasca users do not lose consciousness but experience alterations in it, while Strassman 146) reported that with intravenous DMT injection, users experience a transient loss of their normal awareness lasting only a few minutes, with effects subsiding almost completely in half an hour. Some of the psychological effects during Ayahuasca ingestion are reported by Mabit 21 and include a powerful sense of self-confidence, a new perspective and reinterpretation of intrapsychic conflicts; users may reveal intimate truths, and ayahuasca may be powerful in facilitating psychotherapy.
Kjellgren et al. 20 described the “transcendental circle”, a cycle of experiences consistent among different users following ayahuasca ingestion. Approximately 30 minutes after ayahuasca ingestion, subjects noted changing perceptions and shaking, and felt vulnerable and easily influenced. Shortly after, participants developed feelings of confusion, paranoia and fear; psychological defenses were diminished and participants experienced traumatic memories and gained new insight into personal matters 147. This terrifying state peaks with intense vomiting, after which most participants noted an abrupt shift into an expansive state. Participants describe a transcendental experience in a spiritual world, encountering plant and animal spirits and even contact with a higher power, feelings of oneness with the universe, profound peace and ecstasy, and newly gained understandings of death and what comes after. Sense of time is altered, and users experience feelings of timelessness, time speeding up or slowing down, or traveling in time 148, 149. Users remain aware of their surroundings and are able to speak 7. Beyer 150 refers to a similar pattern and describes three phases, the first with visual imagery and sometimes nausea or vomiting; the second phase is contact with a spiritual world in which users report useful lessons from spirit teachers, and the third phase involves fading visuals and feeling physically drained.
Several studies used the Hallucinogen Rating Scale, which measures subjective effects of psychedelic ingestion on six scales, including Somaesthesia (somatic effects), Affect (emotion and affect), Volition (willful desire to interact), Cognition (thought process and content), Perception (sensory experiences), and Intensity (the strength of the experience). The Hallucinogen Rating Scale was developed by Dr. Rick Strassman 146) and is loosely based on the components of the mental status exam. Riba et al. 15 found that at least 75% of 18 healthy volunteers with experience in psychedelic use responded positively to 14 selected items in the Hallucinogen Rating Scale with a dose of 0.85 mg of DMT/kg of body weight, and described the effects of increased activation, euphoria, and wellbeing. They also reported perceptual changes and increased emotional lability. They also found a correlation between subjective effects of DMT and plasma concentration, and both peaked between 1.5 and 2 hours. Significant dose-dependent increases in all scores on subscales of the Hallucinogen Rating Scale have been found 127, 128, 151. When compared to intravenous DMT, ayahuasca produced a relatively mild high as measured by the Hallucinogen Rating Scale 26.
With regard to visual effects, objects appear to vibrate or increase in brightness, colours intensify, moving geometric patterns and intricate images occur with eyes closed or open 7; kaleidoscopic imagery or visions of people, beautiful scenery, or snakes or jungle animals are common 147, 152, 79. Effects peak between 60 and 120 minutes 18. Visual creativity may be heightened for some time even after acute effects wear off 153. Visual phenomena tend to linger even after acute effects subside, and this may be related to neurochemical changes in the visual cortex and the claustrum. The claustrum, a serotonergic nucleus in the brain, connects nearly all parts of the cerebral cortex. It is theorized that cortical areas with related functions tend to have overlapping claustral projections. Layer 6 (innermost) of the visual cortex and the claustrum have parallel circuits, both of which generate end-inhibition of layers 1 to 4 of the visual cortex through inhibitory interneurons. Lysergic acid diethylamide (LSD) and other hallucinogens are thought to also excite these inhibitory interneurons. Layers 1 to 4, important in interpreting shorter lines, have a property called end-stopping, in which they respond to lines up to a certain length; beyond these lengths, they are inhibited. Uncoupling of claustral and visual cortex sources of edge information, along with abnormal end-stopping properties and erroneous signalling, may explain some of the well known effects like trails, halos, wavy edges, and misinterpretation of contours 153. Synesthesia is common, particularly auditory to visual synesthetic effects, and usually they are associated with music. The tempo and feel of the music are often reflected in the movements of the visions and how often the images change 154. Shanon 154 also noted enhanced improvisation and improvements in their ability to play their instruments by the musicians during Santo Daime rituals, as well as in himself at the piano.
Long-term Psychological Effects
Grob et al. 26 performed a small study comparing 15 syncretic church (União do Vegetal church) ayahuasca users versus 15 matched controls as a part of their Hoasca Project. They found that among the ayahuasca users, all alcohol, depressive, and anxiety disorders remitted after joining the União do Vegetal church. As with the adolescent studies, it is hard to separate the effects of a strong supportive community and religious belonging from the actual effects of the substance, and to determine whether people with particular traits are drawn toward ayahuasca use or church involvement. In the same study, the Tridimensional Personality Questionnaire revealed that users scored significantly lower in the areas of novelty seeking and harm avoidance, but similarly on reward dependence compared to controls 155. On neuropsychological testing with the World Health Organization, University of California, Los Angeles Auditory Verbal Learning Test (WHO-UCLA AVLT), users scored significantly higher in the area of word recall on the fifth trial. They also scored better in number of words recalled, delayed recall, and words recalled after interference, though these were not statistically significant. Grob et al. 26 reported that long term ceremonial use does not appear to negatively affect neuropsychological function. Regular users of ayahuasca scored lower on two of the Addiction Severity Index subscales, Alcohol Use and Psychiatric Status, and ritual use does not seem to be associated with the negative psychosocial impacts of many other drugs of abuse 156. Of 32 members belonging to the American Santo Daime church, 19 reported previous psychiatric histories, but all reported good mental health and only two currently had an active psychiatric disorder 19. Harris and Gurel 157 reported that ayahuasca users scored higher in the areas of Joy in Life and Relationship to the Sacred, and had an experience just as spiritual as the Catholic retreat participants, and also had reduced alcohol consumption, healthier eating, better mood, and self-acceptance.
Barbosa et al. 158 reported on 23 subjects just prior to their first ayahuasca experience in a religious setting and six months following, using three surveys, the Clinical Interview Schedule-Revised Edition (CIS-R), Short Form-36 Health Survey (SF-36), and the Temperament and Character Inventory-125 items (TCI-125). They found no adverse effects on quality of life, measured by the Clinical Interview Schedule-Revised Edition (CIS-R), and some participants showed significant improvements in mental health on the Short Form-36 Health Survey (SF-36) as well as in minor psychiatric symptoms on the Clinical Interview Schedule-Revised Edition (CIS-R). They also found that regular users (>9 sessions in six months) scored significantly higher on social and emotional functioning domains of the Short Form-36 Health Survey (SF-36) questionnaire than less frequent users. In a previous study, Barbosa et al. 29 reported on 28 first time ritual users and also found the same reduction of minor psychiatric symptoms in a shorter time frame of 1 to 2 weeks following use. In another more recent study, Barbosa et al. 159 looked at regular ayahuasca users within a religious setting. Using assessments including the Profile of Mood States (POMS), Big Five Inventory (BFI), Medical Outcomes Study Short Form-36 (SF-36), Addiction Severity Index (ASI), and the California Verbal Learning Test (CVLT), the authors showed that the regular ayahuasca users scored better in terms of mood, having more positive personality traits, better health, improved addiction problems, and better scores on the California Verbal Learning Test (CVLT). Barbosa et al. 159 concluded that religious use of ayahuasca “does not adversely affect neuropsychological functioning and may have positive effects on substance abuse and mood”. One study assessed the effects of ayahuasca on creativity using the Torrance Tests of Creative thinking, and found that ingestion had no effect on the areas of “fluency,” “relative flexibility,” or “relative originality”; however, it increased participants’ ability to come up with “highly original solutions” to tasks 160. In addressing the possibility that more creative individuals may seek out a consciousness-altering experience, they found that baseline creativity scores did not differ when compared to controls. Soler et al. 161 found that ayahuasca intake resulted in increased decentering ability (measured by the Experiences Questionnaire), as well as reduced inner reactivity and reduced judgmental processing of experiences on the Five Facets Mindfulness Questionnaire.
In a 2012 study, Bouso et al. 162 compared a variety of psychological measures in ayahuasca users against matched controls. They found that ayahuasca users scored lower on psychopathology measures (on The Symptom Check-List-90-Revised/SCL-90-R psychopathology questionnaire), performed better on cognitive tests (such as the Stroop Colour and Word Test and the Wisconsin Card Sorting Test), and scored higher on the Purpose in Life Test, Spiritual Orientation Inventory, and Psychosocial Well-Being Test. These differences remained the same at one year followup, and there was no evidence of any deleterious effect on mental health and no signs of cognitive impairment among ritual ayahuasca users. Kuypers et al. 163 looked at ayahuasca’s effect on creative divergent thinking, a way of thinking believed to enhance psychological flexibility and allow for new cognitive, emotional, and behavioural strategies. Assessing participants before and during the acute effects of ayahuasca, the authors found significantly increased divergent thinking while the subjects were on ayahuasca, and suggested this may facilitate psychotherapeutic interventions. In a research study based on results from the Ayahuasca Researcher’s Behavioral Observation Scale (ARBOS), Shamanic Experience and Net Benefit scales, and the Temperament and Character Inventory Predictor scale, Burton 164 suggests we can predict which patients would benefit from or be harmed by participating in an ayahuasca ceremony.
In a questionnaire, 25 Northern European ayahuasca users reported increased self-awareness, being more loving, more empathetic, having greater creativity and new interests especially with nature, and having a more meaningful inner world 20. Winkelman 165 reported similar effects including new insights and access to deeper levels of the self. Serious reflection on life, nature, and consciousness were consistent themes 7. Cakic et al. 70 reported that increased “psychospiritual insight” was the most commonly reported positive effect among 12 Australian recreational DMT users, a finding in keeping with studies of religious and ceremonial ayahuasca use. Many users reported that prior to entering the church, they had alcohol problems and violent behaviour, and described themselves as impulsive, disrespectful, oppositional, and irresponsible. All 15 members involved in the Hoasca Project reported that ayahuasca had a profound influence on their lives, allowing them new insight into their self-destructive ways and motivating them to take control of their lives. They also reported better memory and concentration, and a consistently positive mood; however they all recognized the importance of the sense of community and guidance provided by ritual use within the church 26.
Dr. Jacques Mabit 21 runs an addiction clinic in Peru and uses ayahuasca as a part of the treatment. He reports many positive effects: that ayahuasca increases intellectual capacity and concentration, reduces anxiety, increases tolerance of frustration, improves self-esteem, facilitates individuation processes, allows users to see beyond their own worldview and increases openness to new perspectives. Reports from his patients indicate that ayahuasca facilitates introspection and self-discovery, forgiveness without blame, recognition of mistakes, improved decision making ability, motivation to change, increased quality and quantity of dreams, reflections on life as a part of nature and discovery of previously unknown dimensions of life. As well, users seem to benefit from a structured, spiritual, religious, ritual manner of use 21. Loizaga-Velder and Verres 166 interviewed 14 ritual participants who had long histories of severe substance dependence, and many had several unsuccessful treatments prior to ayahuasca assisted therapy. All participants reported ayahuasca rituals were pivotal to attaining abstinence or achieving less harmful patterns of drug use; they also reported ayahuasca was instrumental in understanding the causes of their addictions. Over half reported reduced cravings. In a study by Cavnar 167, self-identified gay and lesbian ayahuasca users reported feeling affirmation of their sexual orientation.
Ayahuasca uses
Ayahuasca is drunk as a liquid and it has been used for centuries by First Nations peoples from contemporary Peru, Brazil, Colombia and Ecuador for religious ritual and therapeutic purposes 6. Ayahuasca is now being used by tourists seeking a spiritual experience, and by recreational users all over the world 20. Research into medical use of ayahuasca indicates potential as a treatment in addictions, depression and anxiety, with a variety of other possible medical uses, though these require more research 168. Ayahuasca use for depression may be associated with a reduction in short-term depression symptoms and suicidality 169. However, confidence in these findings is also low due to a smaller number of trials overall with small sample sizes and some inconsistency 169. Cakic et al. 170 found that a group of Australian ayahuasca users (n=121) gained psychospiritual insigh from use. Cardenas and Gomez 171 examined motives for modern urban use by 40 residents of Bogota, Colombia. They found that subjects used ayahuasca to achieve mental wellbeing and also to enhance their ability to solve personal problems; in another study 158, the participants cited “healing” and “equilibrium” as reasons for Ayahuasca use. Kjellgren et al. 20 found similar motives among northern European users, including exploring their inner world, personal development, increasing self-awareness, examining psychological patterns, and enhancing creativity. Fiedler et al. 172 studied motives for use among Santo Daime members, and found that reasons were consistently religious or spiritual, as well as self-treatment.
Anecdotal evidence from studies conducted among ayahuasca consumers, and preliminary studies in patients suggest that Ayahuasca has broad therapeutic potential, especially for the treatment of substance dependence and anxiety and mood disorders 26, 173, 19, 156, 174, 162, 175, 176, 177.
Ayahuasca Potential Benefits
Ayahuasca is a mixture of several active ingredients, the ratio of which in the final drink is very variable 178 and therefore its effects depend on both the specific plants used and the method of preparation. The effects are remarkable on a physiological and mental level. However, the dosage of ayahuasca is extremely individual. It depends on the tolerance of the individual and his personal sensitivity, on the intention with which it is to be used, and above all on the intuition of the presiding mestre who works with the substance.
Ayahuasca was and is considered by Native Americans to be the most powerful medicinal plant on Earth 179. They attribute the ability to teach and heal people to the decoction made out of this “magical” liana, and some even believe that it mediates the connection with the universe and spiritual beings 179. Because of these reasons, the use of ayahuasca has spread from South America not only to Europe and to the United States, but throughout the whole world.
Studies and research have raised hopes for Ayahuasca therapeutic potential, but also concerns about its possible toxicity 180, 181, 182. Authors Galvão Ana et al. 183 emphasized in their work the importance of the effect of ayahuasca on the production of salivary cortisol, which acts in the regulation of various physiological, cognitive and emotional pathways. Their opinion was based on studies that suggested that regulating salivary cortisol levels to normal values was considered an important part of the treatment of depression 183.
Some studies suggest that the main active ingredients of ayahuasca (DMT and beta-carbolines) have anti-inflammatory, neuroprotective and memory-improving effects 184. Galvão-Coelho et al. 185 observed a reduction in C-reactive protein (CRP) levels 48 h after the intake in ayahuasca-treated patients but not in placebo-treated subjects. However, the exact mechanism is unknown. The use of ayahuasca during the day subsequently affects the sleep cycle; however, does not worsen the quality of sleep 186. A prolongation of the second sleep stage, shortening of the duration of rapid eye movement (REM) sleep phases and prolongation of non-REM phases was described 186.
Multiple studies suggest that the use of ayahuasca may be beneficial for people with mood disorders, depression, anxiety or post-traumatic stress disorder 187, 188. At the same time, the studies focused on the possibilities of ayahuasca use in the fight against various addictions (alcoholism, nicotinism, cocainism etc.) 8, 189, 190, 191. Some authors assume that the use of ayahuasca could be implemented into the controlled treatment with health benefits 192, 193, 194, 195. Recent studies suggest that regular intake of psychedelic microdoses leads to positive effects on health, mood, cognitive function, and reduced depression and anxiety 192, 193, 194, 195, 196. Yet the outcome of which is ambiguous and still debatable at this time 179. Furthermore, the number of publications focused on the investigation of toxicological risks of ayahuasca is not small. In particular, the limits of the relevant evaluation of its benefits and risks lie in an insufficient number of studies dealing with the analysis of the concentrations of individual components in the organism after the ingestion of ayahuasca decoction with their subsequent correlation to the effects of the plant 179. Last but not least, thorough investigation of pathological changes on tissues and organs in cases of long-term use of ayahuasca is also insufficient and therefore requirable 179.
DMT-containing ayahuasca appears to be less toxic while retaining psychological effects. Based on the studies of the health status of ayahuasca users, the use of ayahuasca may be considered safe and even beneficial to health 183. Adverse results have been reported extremely rarely and are considered to be the result of uncontrolled intake of non-traditional ayahuasca preparations. However, there is still a need for more extensive clinical research on the use of these substances. Such studies should be done by recognized, credible researchers and must include a comprehensive recording of side effects as well as beneficial effects. These studies should be registered with the appropriate global clinical databases 197.
Addictions treatment
Ayahuasca appears to be beneficial in treatment of addictions, and when used appropriately does not appear to carry risks of abuse or dependence 198. Ayahuasca may enable sustained abstinence from alcohol, barbiturates, sedatives, cocaine, amphetamines, and solvents, though most continue to use marijuana 156. Compared to matched controls, regular participants in Brazilian ayahuasca church ceremonies scored significantly lower on the Addictions Severity Index subscales of Alcohol Use and Psychiatric Status, although the authors noted that it is hard to separate whether these effects are from the ayahuasca, involvement in a supportive community, or both 156. Barbosa et al. 158 suggest that based on their findings, administration of hallucinogens in both clinical settings and religious settings can provide benefits.
Any drug that affects dopamine has potential for abuse, and although harmine does, it does not cause dependence 5. Ayahuasca does not show activation in reward-related regions of the striatum or ventral-tegmental area on SPECT imaging 156, 122 and only causes increased blood flow in the frontal and paralimbic areas.
Liester & Prickett suggest 4 hypotheses to explain ayahuasca’s proposed antiaddictive properties 199:
- Ayahuasca reduces brain dopamine levels or activity in the mesolimbic dopamine pathway, decreasing the reward associated with an addictive substance. DMT is a known 5-HT2A receptor agonist and 5-HT2A receptor agonism is known to inhibit dopamine release in the mesolimbic, nigrostriatal, and mesocortical pathways. Reduced brain dopamine also fits with elevated prolactin levels with ayahuasca use 18. The opposite is also true as illustrated by atypical antipsychotics, which have 5-HT2A receptor antagonist activity and exhibit reduced dopamine blockade (70-80% blockade) compared to typical antipsychotics (90%) which have little action at serotonin receptors 200.
- Reduced dopamine in reward pathways impairs the synaptic plasticity involved in addiction development and maintenance.
- The introspection, self-realizations, and healing of past traumas afforded by an ayahuasca experience offer better understanding of consequences and improved decision-making, empowering the individual to abstain.
- Ayahuasca facilitates transcendent experiences; the authors give the example of Bill Wilson, founder of Alcoholics Anonymous, having such an experience (not ayahuasca induced) and being able to give up alcohol.
Pain treatment and Opioid dependence treatment
Beta-carbolines may prove useful in treating opioid addiction. Harmine and harmaline act as imidazoline type 2 receptor agonists I2 105. Harmane and harmine have both been reported to reduce the symptoms of morphine withdrawal 201. Miralles et al. 202 assessed the affinity of various beta-carbolines for the I2 binding site in brain and liver and also found that norharmane prevents the stimulatory effects of opioid withdrawal as measured by withdrawal symptom severity, and attenuated L-3,4-dihydroxyphenylalanine (L-dopa) synthesis normally associated with withdrawal.
Cocaine dependence treatment
A study by Glick et al. 203 reported that harmaline led to significantly reduced cocaine and morphine self-administration in rats. While cocaine increases dopamine efflux and reuptake inhibition in both the shell and core of the nucleus accumbens, harmine only augments efflux in the shell of the nucleus accumbens 102, 112, perhaps demonstrating one mechanism of harmine that is similar to cocaine that can be useful in treatment and has far less addictive potential. A Canadian study by Thomas et al. 177 showed that ayahuasca holds promise as a potential treatment for cocaine dependence, with a statistically significant reduction in use (by self-report) that is greater than the reduction in either tobacco or alcohol use.
Alcoholism treatment
Halpern 204 touched on promising past research involving LSD in the treatment of alcoholism and anecdotal evidence of peyote containing mescaline used in the Native American Church being potentially useful in treating drug dependence and alcohol addiction, and suggested it is time to start studying hallucinogens again. As mentioned previously, Doering-Silveira et al. 205 found that adolescents from a Brazilian ayahuasca-using church had less recent alcohol use (32.5%) compared to adolescents who had never used ayahuasca (65.1%). Oliveira-Lima et al. 206 showed that in mice, ayahuasca inhibited some of the early behaviours that were associated with developing alcohol addiction.
Depression treatment
Osorio et al. 176 in an open label trial in an inpatient psychiatric unit, found that a single dose of ayahuasca has rapid acting anxiolytic and antidepressant effects in patients with recurrent depression. dos Santos et al. 207 reviewed several clinical trials on ayahuasca, psilocybin, LSD, and their effects and concluded that all these drugs could be beneficial in treatment of depression (especially in treatment-resistant subjects), as well as anxiety and alcohol and tobacco dependence. The results also seemed to confirm that both the DMT and the beta-carbolines in ayahuasca show promise as effective depression and anxiety treatments. They highlighted findings that serotonin 5-HT1A receptor agonists have shown antidepressive and anxiolytic effects in both humans and animals, and 5-HT2A/2C agonists had antidepressive and anxiolytic effects in animal studies. In addition, 5-HT1A/2A/2C receptor agonists have shown anti-inflammatory properties, and there is growing evidence that inflammation is another process implicated in the pathogenesis of anxiety and depression 173.
Several studies of harmine have shown an antidepressant effect 208, 209, 210, 211, 212, 213. One known mechanism through which harmine and harmaline may exert an antidepressant effect is reversible inhibition of MAO-A 214, resulting in increased neurotransmission. Their reversibility for MAO-A inhibition makes them safer than the traditional nonselective, irreversible MAO inhibitors 28. Fortunato et al. 212, 213 have conducted several animal studies assessing the antidepressant effect of harmine. Using the forced swim test, it was shown that the animals treated with harmine had decreased immobility and more swimming and climbing, and they had increased levels of brain-derived neurotrophic factor (BDNF) which has an antidepressant effect in the brain 209, 210, 211, 212, 213. Harmine was also able to reverse the anhedonic effects of the chronic mild stress test 213. Harmine acts to decrease synaptic glutamate via increased GLT-1/EAAT2 expression and subsequently increasing glutamate transport 107.
DMT activates sigma-1 receptors. Other antidepressants, though not all, of the SSRI, MAOI and TCA classes have been found to do so as well. These receptors are found throughout the nervous system, and are concentrated in the hippocampus, frontal cortex, and olfactory bulb, consistent with a possible role in depression 215. Past experiments have shown an antidepressant-like effect in mice administered sigma-1 receptor agonists 216 and attenuation of these effects with sigma receptor antagonists [181]. Agonists of the sigma receptor are being studied as potential antidepressant drugs 214. More work into the functions of sigma receptors and their role in depression treatment is needed. A possible connection lies in the inhibitory effect of DMT on the NMDA receptor through sigma receptor activation 83.
Both I1 and I2 imidazoline receptors have been associated with the pathology of depression 217. I1 sites are decreased in brains of depressed suicide victims, notably in the hippocampus and prefrontal cortex 217. Imidazoline I1 binding sites are found throughout the human brain, and the highest density areas include in the striatum, pallidum, hippocampus, amygdala, and substantia nigra 218. Imidazoline I1 receptors are thought to be involved in the central inhibition of sympathetic outflow, which can be altered in depression and hypertension 219. Interestingly, the number of I1 binding sites are reported to be increased on platelets of patients experiencing depression and premenstrual dysphoric disorder. This effect was highly correlated with severity of symptoms, but there was a consistent return to normal levels following treatment with fluoxetine, citalopram, bupropion, desipramine, clomipramine, imipramine, and lithium, even though several of these drugs act through different mechanisms, which suggests platelet I1 density could be used as a possible biological marker of depression 219. Halaris and Piletz 219 also described an unpublished finding that in nondepressed patients, desipramine failed to produce the same effect. Therefore, platelet I1 sites could have potential as a biological marker of depression, as well as a measure of response to treatment. A downregulation of I2 binding sites has been found in frontal cortices and hippocampi of depressed humans postmortem. Harmine and harmaline have high affinity for the I2 binding site in rat brains [188]. In terms of clinical use, the selective I2 ligand BU224 showed antidepressant-like activity in rats and increased 5-HT levels in the frontal cortex and hypothalamus 218. Antidepressant treatment caused upregulation of I2 sites in rat brains 220. Most I2 selective ligands have been found to be allosteric inhibitors of both MAO-A and MAO-B 221.
Evidence is now suggesting that reactive oxygen species (ROS) may be involved in the pathogenesis of depression and anxiety 222. Harmine has shown to be of benefit as it increased levels of both superoxide dismutase and catalase enzymes, and attenuated oxidative stress parameters of lipid and protein oxidation in rat brain hippocampus, a structure involved in mood regulation 121.
Anxiety treatment
Jacob and Presti 223 suggested that DMT action at a trace amine receptor may produce an anxiolytic effect. Anxiety, like depression, is another disorder which has been linked to oxidative stress 222. Sarris et al.224 highlight ayahuasca as a treatment of potential use in their review of plant based medicines for anxiety. A double blind study showed a statistically significant reduction of hopelessness and panic-like parameters using standardized questionnaires, the Beck Hopelessness Scale and the Revised Anxiety Sensitivity Index upon acute ayahuasca ingestion 173. Furthermore, it has been suggested that DMT acts in a manner similar to serotonin, and serotonin (5-HT2) receptor activation has been shown to alleviate panic symptoms 225. It is important to note that some beta-carbolines may have a possible anxiogenic effect, given their inverse agonist effect at the benzodiazepine receptor site of the GABA-A receptor 108, dopamine (D(2)) and benzodiazepine receptors. Drug Alcohol Depend. 2000 Aug 1;60(2):121-32. doi: 10.1016/s0376-8716(99)00148-9)), 109.
Psychotherapy
There are many reported psychotherapeutic benefits of ayahuasca, however most studies stress that this is only when it is used in specific settings 28. Like LSD in the 1950s, ayahuasca is now being considered as a tool to facilitate psychotherapy, by dissolving the ego, promoting introspection, and aiding in processes of self-analysis 152. Barbosa et al. 29 suggested that hallucinogens may also act to facilitate association and memory processing. However, regulations governing the use of psychoactive substances often limit the ability to undertake scientific investigations of such novel approaches.
Ayahuasca side effects
There is no safe level of Ayahuasca. Use of any drug always carries some risk. It’s important to be careful when taking any type of drug.
Ayahuasca affects everyone differently, based on 22:
- your size, weight and health
- whether the person is used to taking Ayahuasca
- whether other drugs are taken around the same time as Ayahuasca
- the amount of Ayahuasca taken
- the strength of the Ayahuasca decoction (varies from batch to batch)
- the environment where Ayahuasca is taken.
The effects of ayahuasca can last between 4- to 6-hours and may include:
- nausea and vomiting (induced by drinking the decoction). The most frequently reported adverse physical health effect was vomiting and nausea (68.2%), while the frequency of other adverse effects was 17.8% headache. It is important to clarify that vomiting and nausea is considered a normal effect of ayahuasca for experienced users. In the case of traditional ayahuasca ceremonies and even in non-traditional ceremonies, not only is vomiting and nausea not considered an adverse effect, but it is even sought out for its purging and perceived spiritual cleansing benefits 226
- diarrhea
- euphoria
- feelings of connection and unity
- introspection
- intense visual and auditory hallucinations
- experiencing powerful emotions
- anxiety
- panic and fear
- moderate increase in blood pressure and heart rate
- increased body temperature 6, 227
When Ayahuasca is taken in a traditional or ritual setting, these effects may be perceived as cleansing or purging and a part of the spiritual or healing journey 6.
Although there is consensus regarding the general safety of ayahuasca 228, 229, 230, 231, the following reports of adverse effects have been identified in the literature:
- The American Association of Poison Controls Centers’ (AAPCC) National Poison Data System (NPDS) collected 538 calls related to exposure to ayahuasca botanical products between 2005 and 2015 230. Forty-one cases (7%) reported major clinical manifestations and 296 cases moderate ones (55%). The most common clinical effects were hallucinations (190, 35%), agitation (181, 34%), tachycardia (180, 34%), confusion (99, 18%), hypertension (87, 16%), mydriasis (72, 13%), and vomiting (32, 6%). The most severe effects were seizures (12, 2%), respiratory arrest (7, 1%), and cardiac arrest (4, 1%). Three fatalities were reported. In this study, patient’s characteristic, previous medical conditions, concomitant medication use, and what subjects actually took were unknown 230.
- A systematic review of the published case reports describing psychotic episodes associated with ayahuasca use found three case series and two case reports describing psychotic episodes 232 and at least three other cases have been reported since then 233, 234, 235. Although most of the cases had previous psychiatric diagnoses, a few did not. Some of the cases required antipsychotic medication, but all of them returned to their pre-crisis mental states after a variable amount of time.
- A study involving 32 subjects of a US Santo Daime church also identified side-effects, most commonly nausea (11 subjects), vomiting (9 subjects), and exhaustion in the following days (9 subjects) 19. Other somatic effects, like headache, tachycardia, and muscle spasms, were reported by a few subjects [57]. In a recent retrospective study involving 614 members (regular uses) of the União do Vegetal, a Brazilian religion where ayahuasca is a sacrament, the most common physical effects, as expected, were vomiting (96.74%) and nausea (91.53%) 19. Diarrhea, shivers, rapid heart rate (tachycardia), tremor, and tinnitus were other common effects 19. Surprisingly, persistent physical effects were reported by 41.86% of the participants, among whom the daily lives of 3.89% (10 individuals) were reasonably (n = 9) or very (n = 1) affected in a negative way 19. Adverse psychological effects (anxiety, disorientation, or distress) were less common; however, persistent adverse psychological effects were reported by 20.68% of the participants (127 individuals), among which 11.81% (n = 15) indicated that their daily lives were moderately (n = 14) or very (n = 1) negatively affected by persistent effects 19. The total duration of the persistent adverse effects was not recorded. Participants with a reported psychiatric diagnosis experienced adverse effects more frequently than those without 19.
- In a prospective longitudinal study involving 40 subjects who participated in ritual ayahuasca ceremonies 236, 7 referred to an extremely challenging psychological reaction, involving psychotic symptoms in some cases. Among those 7 subjects, 4 had a previous psychiatric diagnosis. The condition of all subjects with psychiatric antecedents improved, and none of those without were found to be worse during the follow-ups 229.
- In a series of controlled clinical trials, while characterizing the pharmacology of ayahuasca with a total of 24 subjects using low (0.5mg/DMT/kg), medium (0.75mg/DMT/kg), and high (1mg/DMT/kg) doses of ayahuasca 237, 238, subjects scored active doses higher than a placebo in terms of the Visual Analogue Scales “liking,” and the ayahuasca was well tolerated. However, one subject who took the medium dose experienced an intensely dysphoric reaction with transient disorientation and anxiety that lasted 20 minutes without the need for medication 237, 239. In another clinical study using functional MRI (fMRI) and involving 10 subjects, effects were significant only at 40- and 80-minutes post‐intake in the brief psychiatric ratings scale (BPRS) used to detect psychotic symptoms and the Young Mania Rating Scale (YMRS) used to measure mania symptoms, but subjects were not incapacitated from performing the imagery task involved 240. A recent controlled clinical trial studying the effects of ayahuasca on the recognition of facial expressions of emotions in naive healthy volunteers did not find differences in clinical variables between ayahuasca and placebo 241.
Ayahuasca Mental adverse effects
The frequency of adverse mental health effects was relatively high (55.4%), with a similar frequency being found for the emotional-cognitive and altered perception factors (42% and 38.3%, respectively) 242. Although the frequency of any adverse mental health effects was high, only “hearing or seeing things that other people do not hear or see” commonly known as auditory and visual hallucination was observed in 28.5% of the ayahuasca users 242. In relation to this response and also “visual distortions” it is important to note some respondents reporting these changes specifically mentioned in subsequent qualitative responses that they had considered these positive and not adverse effects 242. Other mental health adverse effects were reported in less than 21.0% (“feeling disconnected or alone”) of the participants. However, there was a low frequency of severe adverse effects (4.4% was the highest frequency, for “visual distortions”). Moreover, 11.9% of the participants reportedly needed professional mental health support for the adverse effects they experienced. For most participants reporting adverse mental health effects, the duration was identified as being for less than a week. While previous research has found long-lasting, severe adverse mental health effects in rare cases 232, other research has found that especially challenging adverse effects may improve psychiatric conditions 229, while other work from this study has reported the number of adverse mental health effects reported to be negatively associated with current mental health and perceived improvement in psychological wellbeing 243. Other research has also found that adverse effects can be important enough to interfere in individual daily lives 231. So, future studies should focus on follow-up and the evolution of the adverse effects when they appear.
History of ayahuasca use, age at initial ayahuasca use and lifetime use were not related with the adverse mental health effects 242. Regarding the clinical variables, only a previous diagnosis of anxiety disorder increased the likelihood of adverse emotional-cognitive effects, and previous physical health conditions increased the likelihood of altered perception adverse effects 242. All non-religious contexts (traditional, non-traditional and non-supervised) increased the likelihood of emotional-cognitive and altered perception adverse effects 242. Adverse emotional-cognitive and altered perception effects were more likely to be observed in females, younger participants, and those unmarried 242.
Adverse mental health effects were not significantly associated with ayahuasca history of use variables. This finding, while intriguing, is in accordance with the early age of ayahuasca initiation in traditional contexts, both Indigenous and in ayahuasca churches, where researchers failed to find long-term neuropsychiatric alterations 244, 245, 246, 247. Over the last decade ayahuasca has increasingly been perceived to have therapeutic effects, leading to an increasing number of people accessing ceremonies as a complementary medicine or self-care practice. This situation has resulted in rare physical and/or mental health issues being more commonly seen in non-traditional contexts 248, 249.
Ayahuasca drug interactions
There is a possibility that any substances, including beta-carbolines, that act as MAO-A inhibitors can produce serotonin syndrome 213. Serotonin syndrome is a potentially life-threatening drug reaction 62, 63. Serotonin syndrome is caused by medications that build up high levels of serotonin in the body. Too much serotonin causes signs and symptoms that can range from mild shivering and diarrhea to severe muscle rigidity, fever and seizures 62, 63. Milder forms of serotonin syndrome may go away within a day or two of stopping the medications that cause symptoms and, sometimes, after taking drugs that block serotonin 62. Severe serotonin syndrome can cause death if not treated 62.
McKenna 28 suggested that DMT and the beta-carbolines in ayahuasca, when combined with an selective serotonin reuptake inhibitor (SSRI) antidepressant could cause serotonin syndrome. Callaway and Grob 250 also report a case of serotonin syndrome in a patient using the SSRI fluoxetine in conjunction with ayahuasca. The irreversible, nonselective MAOIs phenelzine and tranylcypromine are associated with serotonin syndrome, and there are also cases with opiates, analgesics, tricyclic antidepressants (TCAs), SSRIs, and antimigraine drugs, and it is suggested that those who have recently used any ginseng, St. John’s wort, dextromethorphan, or MDMA should be cautious 7. A chart of potential interactions exists on the harm reduction organization TripSit’s website (TripSit.me). Balikova 251 reported on an incident of 30 people who ingested a brew containing harmine, atropine, and scopolamine in a meditation session, and found tachycardia, amnesia, hallucinations, hyperthermia, hypotension, mydriasis, collapse, coma, and even respiratory depression requiring mechanical ventilation. A synergistic effect was found with beta-carbolines in combination with atropine and scopolamine; the ingested doses of the three substances were all 8 to 13 times less than estimated lethal doses 7.
Harmine, harmol, and harmane have been found to be noncompetitive inhibitors of CYP3A4; they are also both substrates and inhibitors of CYP2D6. Harmaline, harmine and harmol showed competitive inhibition of CYP2D6 252. With two major CYP enzymes inhibited, users should be cautious of drug interactions. As well, genetic polymorphisms can affect the efficacy of these enzymes.
Ayahuasca bad trips
Some people may have negative experiences taking psychedelics, or experiences they find challenging. This can include experiencing:
- frightening or confronting hallucinations
- intense anxiety and confusion
- fear and paranoia 6
These experiences may be understood or interpreted differently in a traditional or ritual context, where they may be seen as lessons and part of a spiritual or healing journey rather than wholly negative.
Ayahuasca dependence and tolerance
As a Schedule 1 controlled substance, DMT can be seen as an addictive substance, associated with substantial health risks 66. Studies have been contradicting this finding, as no compulsive drug-seeking precipitated by consumption of DMT or ayahuasca has been reported in humans 22. Repeated use of Ayahuasca does not appear to result in tolerance to the effects, and it appears to pose an extremely limited risk for dependence 156. Psychedelics, including DMT and ayahuasca, are seen as safer substances than cocaine, opiates, or even the widely used nicotine and alcohol, with the advantage of lacking the abuse potential, characteristic of these former drugs 22. As reviewed by Gable et al. 22, reports of abstinence syndrome after termination of DMT consumption are unknown.
Studies with repeated administration of DMT to volunteers have seen little or no drug tolerance 253, 91. Comparing long-term users and occasional consumers of ayahuasca, Bouso et al. 254 found that following ingestion of a single dose, both groups were associated with lower scores on working memory and performance improvement, but only the occasional users had an impaired performance in strategic planning. Therefore, greater prior exposure to ayahuasca by long-term users was associated with drug-induced neuromodulatory or compensatory effects, resulting in reduced cognitive incapacitation. Further reports on tolerance solely included slight changes in the release of growth hormone (GH), adrenocorticotropic hormone, and prolactin, which were found to be decreased following a second administration, and lower response in the systolic blood pressure and heart rate 255, 256. DMT did not elicit tolerance in animal models like squirrel monkeys 257 and cats 258. Additionally, LSD is not capable to produce cross-tolerance to DMT 259, contrary to what happens with other classic hallucinogens like mescaline or psilocybin 260, 261. The absence of cross-tolerance suggests a distinguished pharmacodynamic behavior for DMT, which makes it a quite unique substance among classic hallucinogens.
Morgenstern et al. 198 reported that almost no hallucinogen users had difficulty cutting down or controlling use, unlike many other drugs. In a study of rhesus monkeys, Fantegrossi et al. 262 found reinforcing effects of the hallucinogens DMT, mescaline, and psilocybin, and suggest that the patterns of self-administration demonstrate weak reinforcing effects, and possibly mixed reinforcing and aversive effects. Ayahuasca does not seem to have the negative psychosocial implications caused by many drugs of abuse 156. Mixed results were found in studies of drug tolerance in animal studies 263, 258, 264 as well as in human studies 7, particularly to the psychoactive effects, which is unique among other known hallucinogens. Callaway et al. 120 found that some physical tolerance may develop in humans with regular use. In a study by dos Santos et al. 128, acute tolerance failed to develop for any measures aside from growth hormone (GH), which showed decreased release on second administration, as well as a slightly lower response in the systolic blood pressure (SBP) and heart rate (HR). Another study similarly showed tolerance with heart rate, adrenocorticotropic hormone (ACTH) and prolactin 79. Another study found that there was little to no tolerance with DMT in cats 258.
Is Ayahuasca safe?
The stem and/or bark of the Banisteriopsis caapi vine rich in beta-carboline harmala alkaloids can induce tremor in mice, thought to be due to the interaction of these compounds with tryptamine binding receptors 265. An experiment by Louis et al. 98 suggests people should be cautious with beta-carbolines, as they are found endogenously in higher levels in essential tremor patients, and that harmine and harmane may be tremorigenic. Bouso et al. 266 compared two groups of ayahuasca users, one with long term experience and the other with occasional use. The study found that acute ayahuasca use does impair working memory, but having greater prior ayahuasca exposure was associated with less incapacitation during administration, and detrimental effects on cognition were mainly seen in the occasional ayahuasca use group. Those findings suggest there may be neuromodulatory or compensatory effects induced by long term ayahuasca use.
In an attempt to investigate ayahuasca toxicity, Pic-Taylor et al. 267 administered ayahuasca doses 30 and 50 times higher than that typically used in religious rituals to female Wistar rats. While the authors could not calculate an LD50 (the amount of a solid or liquid material that it takes to kill 50% of test animals in one dose) based on their findings, they determined that the lethal dose (corresponding to 15.1 mg/kg DMT) is higher than 50 times a typical Ayahuasca dose used during religious ceremonies. Pic-Taylor et al. 267 also found that the increased serotonergic activation from these high doses led to some neural degeneration, but no permanent alteration in brain structure or number of cells was found. Other rodent studies allowed to estimate that the LD50 values for humans are approximately of 1.6 mg/kg for DMT i.v. administration (a total dose of 112 mg for a typical 70 kg individual), and 8 mg/kg for DMT per os (a dose of 560 mg) 22, which is significantly higher than the average ceremonial dose of DMT (27 mg), giving DMT/ayahuasca a safety margin of approximately 20-fold.
The toxic dose of ayahuasca would be approximately 7.8 liters for a 75 kg person 35. Given its highly unpleasant taste, it is unlikely anyone would ever reach this dose. In addition, vomiting and diarrhea occur long before this limit is reached 21.
In a literature search, Hamill et al 5 found no reports of deaths directly attributable to ayahuasca use. There are a few reported deaths associated with ayahuasca-like herbal preparations, but in these cases it appears coingestion with other substances was to blame. The only two cases of death reported in the literature that involved ayahuasca/DMT/harmala alkaloids were of a 71-year-old diabetic female who consumed B. caapi mixed with tobacco leaves (no information on quantities) 50 and a 25-year-old man who consumed herbal extracts containing harmala alkaloids and tryptamines (no information on quantities) 51, with no anatomical cause of death found in the autopsies. In the first case, blood analysis revealed only the presence of nicotine (710–1900 ng/mL) and the cause of death was determined as acute nicotine intoxication. In the second case, reported by Sklerov et al. 51, the following concentration ranges were obtained in the blood analysis: 0.01–0.02 mg/L for DMT, 0.04–0.07 mg/L for harmaline, 0.08–0.17 mg/L for harmine, 0.24–0.38 for tetrahydroharmine (THH) and 1.20–1.88 mg/L for 5-MeO-DMT. The cause of death was undetermined. However, a few cases of suspected deaths involving ayahuasca consumption have been reported in the media.
A review by dos Santos 268 covers ayahuasca use in pregnancy, and concludes that while some animal studies show toxicity from in utero ayahuasca exposure, what little information there is on the topic indicates that there are no serious adverse effects of ayahuasca exposure in utero in humans, but more information is needed before its safety in pregnancy is fully understood 269. Pregnant rats consuming high doses of ayahuasca (10 times the normal human Ayahuasca dose) displayed decreased food consumption. The rats displayed decreased weight gain but increased relative liver weight, possibly indicating some liver toxicity 270. These researchers found a ayahuasca dose-response relationship, and fetal effects at 10 times the normal human dose include visceral and skeletal malformations, dilated lateral and third ventricles, along with decreased body weight, thought to represent intrauterine growth restriction (IUGR). A more recent study by Gardner et al. 271 discussed congenital malformations in livestock in parts of South America and Mexico, which have long been attributed to ingestion of the Mimosa tenuiflora shrub containing N-methyltryptamine (NMT) and DMT. Their findings showed increased rates of cleft palate, scoliosis, and skeletal deformities in rat pups compared to controls when pregnant mothers were fed diets including DMT, N-methyltryptamine (NMT), both, or extracts of the Mimosa tenuiflora seeds and leaves.
In a recent study, Colaço et al. 272 submitted Wistar rats to a chronic 28-day treatment using the same ayahuasca samples as a previous Pic-Taylor study 273 at doses 2 times higher (corresponding to 4.28 mL/kg of ayahuasca, 0.52 mg/kg of DMT, 5.16 mg/kg of harmine, 0.342 mg/kg of harmaline, and 0.66 mg/kg of tetrahydroharmine [THH]) than the common ritual dose of ayahuasca. Blood analysis (i.e., hemoglobin, total hematocrit, erythrogram, leukogram, corpuscular volumes), and biochemical analysis for hepatic function (aspartate transaminase, alanine transaminase, and alkaline phosphatase), kidney function (urea and serum creatine), and tissue damage (lactate dehydrogenase) were performed, with no reports on toxic effects 272. Furthermore, a 1-year study comparing regular ayahuasca users with controls 94 showed no indication that long-term ayahuasca use could induce psychologic maladjustment, mental health deterioration, or cognitive impairment. No decreased cognitive function nor increased mental health issues were associated with populations who have a life-time use of these mind-altering substances, particularly as part of religious ceremonies 274, 275.
There have been a few reported cases of poisoning from Peganum harmala seeds. These included a 35-year-old male with abdominal pain, low blood pressure (hypotension) and convulsions, but this was after ingesting 150 g of Peganum harmala seeds 276. His symptoms resolved after several hours 276. It is possible the convulsions could have been related to the inverse agonist effect of some of the beta-carbolines at the benzodiazepine receptor site on the GABA-A receptor 108, dopamine (D(2)) and benzodiazepine receptors. Drug Alcohol Depend. 2000 Aug 1;60(2):121-32. doi: 10.1016/s0376-8716(99)00148-9)), 109. Another case involved a 27-year-old woman who had ingested 50 g of Peganum harmala seeds in a cup of coffee. She presented with hallucinations, slow heart rate (bradycardia), nausea, and vomiting, but was discharged several hours later, and laboratory investigation results were all normal 276. Another case was a 45 years old woman who ingested about 50 grams seed of Peganum harmala for heavy periods (menorrhagia) 277. She suffered nausea, vomiting, dizziness, tremor, ataxia, and confusion. On physical examination, she had hypotension (BP=90/60 mmHg) with normal heart rate (60 beat/min) and impaired knee to heel test. Her consciousness was reduced without any hallucination. Her laboratory test was normal. She was discharged at good condition 18 hours later 277. Several similar cases have been reported 278, 279, 280.
Harmaline, which constitutes 3% of Peganum harmala seeds and is present in low amounts in ayahuasca, is two times more toxic than harmine, inducing tremor, convulsion, respiratory paralysis, hypothermia, central nervous system depression, visual trouble, delirium, loss of coordination, paralysis, and sometimes hallucinations, when consumed in high doses 277. Toxic effects usually appear 3 to 4 hours after ingestion, with the first symptoms being nausea and vomiting, followed by an altered mental state and other neurological presentations.
Pharmacological studies of acute ayahuasca administration to healthy volunteers and mental health assessments of long-term ayahuasca consumers suggest that ayahuasca is relatively safe 15, 16, 26, 173, 19, 156, 174, 162, 127, 128, 281.
Ayahuasca during Pregnancy
As pregnant women also participate in ceremonial ayahuasca consumption, studies should be pursued in order to assess possible toxic effects that might occur during pregnancy 268. Experimental data recorded in pregnant rats dealing with the in utero toxicity of ayahuasca alkaloids are contradictory, with some authors suggesting toxicity 282 while others do not 283. Oliveira et al. 284 first reported the evidence of toxic effects in pregnant rats due to chronic ayahuasca consumption during gestational days 6–20, at doses 10 times higher (14 mL/kg of ayahuasca) than the normal human dose. These animals presented decreased food consumption and weight gain, accompanied with increased relative liver weight, an indication of hepatotoxicity. Effects at the fetal level included visceral and skeletal malformations, dilated lateral and third ventricles, and decreased body weight 284. Nevertheless, such data appear to be of limited relevance and should be extrapolated to humans with caution since, from field observations, pregnant women attending the religious ceremonies tend to use ayahuasca less frequently, and in reduced quantity 268. Although more investigation on this subject is needed, there are a few scientific evidences suggesting that no psychiatric nor neuropsychological problems are seen in adolescents who were exposed to ayahuasca before birth 268.
Is Ayahuasca dangerous?
Yes, Ayahuasca is potentially dangerous if taken by people with certain health conditions or taking certain medications, particularly ones that also release serotonin. Deaths have occurred 50, 51. People who should not take ayahuasca include those with:
- A family history of mental health issues
- Bipolar disorder
- Depression taking antidepressants
- Difficulty vomiting
- Epilepsy
- Fractures
- Gastrointestinal disorders
- Glaucoma
- Infection
- Recent surgery
- Retinal detachment
- Schizophrenia
- Seizures
- Serious liver, kidney, or gallbladder disease
- Severe cardiovascular disease
- Stroke
- Tuberculosis
- Parkinson’s disease
- Psychosis
- Uncontrolled or very high blood pressure
- Ayahuasca is not recommended for pregnant or lactating women
There is no safe level for ayahuasca and the use of this plant medicine always carries some risk. How ayahuasca affects you depends on:
- If you have taken it before
- Other medications you are taking
- The amount taken
- The strength of the decoction (varies from batch to batch)
- Your current state of health
- Weight
- Where you take it and the vibe.
Traditional ayahuasca experiences are hosted by a shaman or curandero who prepares and concentrates the brew according to traditional methods and his experience. But there is no regulation of the industry and commercialization has taken over with money-grabbing imposters prevalent and hard to distinguish among legitimate ayahuasca centers. These may not have the same sense of ethics as some of the more genuine retreat centers or deliver the same experience, and at least 11 tourist deaths over the past decade have been associated with being part of an ayahuasca experience.
Ayahuasca should only be taken under the supervision of an experienced shaman or curandero because a trip can change your state of consciousness for several hours and you need to be kept safe.
DMT, the active ingredient in ayahuasca is classed as a Schedule I substance in the United States and it is illegal to import, possess, sell, distribute, or consume ayahuasca.
References- Nichols D. E. (2016). Psychedelics. Pharmacological reviews, 68(2), 264–355. https://doi.org/10.1124/pr.115.011478
- Nichols D.E. Hallucinogens. Pharmacol. Ther. 2004;101:131–181. doi: 10.1016/j.pharmthera.2003.11.002
- Strassman R.J. DMT: The Spirit Molecule: A Doctor’s Revolutionary Research into the Biology of Near-Death and Mystical Experiences. Park Street Press; Rochester, NY, USA: 2001.
- Ott J. Pharmacotheon: Entheogenic Drugs Their Plant Sources and History. Natural Products Company; Kennewick, WA, USA: 1993.
- Hamill J, Hallak J, Dursun SM, Baker G. Ayahuasca: Psychological and Physiologic Effects, Pharmacology and Potential Uses in Addiction and Mental Illness. Curr Neuropharmacol. 2019;17(2):108-128. doi: 10.2174/1570159X16666180125095902
- Santos, R. G. dos ., Bouso, J. C., & Hallak, J. E. C.. (2017). Ayahuasca: what mental health professionals need to know. Archives of Clinical Psychiatry (são Paulo), 44(4), 103–109. https://doi.org/10.1590/0101-60830000000130
- Gable RS. Risk assessment of ritual use of oral dimethyltryptamine (DMT) and harmala alkaloids. Addiction. 2007 Jan;102(1):24-34. doi: 10.1111/j.1360-0443.2006.01652.x
- McKenna D.J. Clinical investigations of the therapeutic potential of ayahuasca: Rationale and regulatory challenges. Pharmacol. Ther. 2004;102:111–129. doi: 10.1016/j.pharmthera.2004.03.002
- Schultes R.E. Ethnotoxocological significance of additives to New World hallucinogens. Plant Sci. Bull. 1972;18:34–41.
- Rivier L., Lindgren J. Ayahuasca, the South American hallucinogenic drink: Ethnobotanical and chemical investigations. Econ. Bot. 1972;29:101–129. doi: 10.1007/BF02860772
- McIlhenny E.H., Riba J., Barbanoj M.J., Strassman R., Barker S.A. Methodology for and the determination of the major constituents and metabolites of the Amazonian botanical medicine ayahuasca in human urine. Biomed. Chromatogr. 2011;25:970–984. doi: 10.1002/bmc.1551
- McKenna D.J., Towers G.H., Abbott F. Monoamine oxidase inhibitors in South American hallucinogenic plants: Tryptamine and beta-carboline constituents of ayahuasca. J. Ethnopharmacol. 1984;10:195–223. doi: 10.1016/0378-8741(84)90003-5
- Callaway J.C., Raymon L.P., Hearn W.L., McKenna D.J., Grob C.S., Brito G.S., Mash D.C. Quantitation of N,N-dimethyltryptamine and harmala alkaloids in human plasma after oral dosing with ayahuasca. J. Anal. Toxicol. 1996;20:492–497. doi: 10.1093/jat/20.6.492
- Wang Y.H., Samoylenko V., Tekwani B.L., Khan I.A., Miller L.S., Chaurasiya N.D., Rahman M.M., Tripathi L.M., Khan S.I., Joshi V.C., et al. Composition, standardization and chemical profiling of Banisteriopsis caapi, a plant for the treatment of neurodegenerative disorders relevant to Parkinson’s disease. J. Ethnopharmacol. 2010;128:662–671. doi: 10.1016/j.jep.2010.02.013
- Riba J, Valle M, Urbano G, Yritia M, Morte A, Barbanoj MJ. Human pharmacology of ayahuasca: subjective and cardiovascular effects, monoamine metabolite excretion, and pharmacokinetics. J Pharmacol Exp Ther. 2003 Jul;306(1):73-83. doi: 10.1124/jpet.103.049882
- Riba J, Romero S, Grasa E, Mena E, Carrió I, Barbanoj MJ. Increased frontal and paralimbic activation following ayahuasca, the pan-Amazonian inebriant. Psychopharmacology (Berl). 2006 May;186(1):93-8. doi: 10.1007/s00213-006-0358-7
- de Araujo DB, Ribeiro S, Cecchi GA, Carvalho FM, Sanchez TA, Pinto JP, de Martinis BS, Crippa JA, Hallak JE, Santos AC. Seeing with the eyes shut: neural basis of enhanced imagery following Ayahuasca ingestion. Hum Brain Mapp. 2012 Nov;33(11):2550-60. doi: 10.1002/hbm.21381
- Callaway JC, McKenna DJ, Grob CS, Brito GS, Raymon LP, Poland RE, Andrade EN, Andrade EO, Mash DC. Pharmacokinetics of Hoasca alkaloids in healthy humans. J Ethnopharmacol. 1999 Jun;65(3):243-56. doi: 10.1016/s0378-8741(98)00168-8
- Halpern JH, Sherwood AR, Passie T, Blackwell KC, Ruttenber AJ. Evidence of health and safety in American members of a religion who use a hallucinogenic sacrament. Med Sci Monit. 2008 Aug;14(8):SR15-22. https://medscimonit.com/abstract/index/idArt/865802
- Kjellgren A, Eriksson A, Norlander T. Experiences of encounters with ayahuasca–“the vine of the soul”. J Psychoactive Drugs. 2009 Dec;41(4):309-15. doi: 10.1080/02791072.2009.10399767
- Mabit J. Psychedelic Medicine (Vol 2): New Evidence for Hallucinogenic Substances as Treatments. 2007. Ayahuasca in the treatment of addictions.
- Gable R.S. Risk assessment of ritual use of oral dimethyltryptamine (DMT) and harmala alkaloids. Addiction. 2007;102:24–34. doi: 10.1111/j.1360-0443.2006.01652.x
- Nutt DJ. Drugs Without the Hot Air : Making Sense of Legal and Illegal Drugs. Revised & updated second edition. [International territories version] ed. Cambridge: UIT Cambridge; 2020.
- Ayahuasca. https://adf.org.au/drug-facts/ayahuasca
- Dobkin de Rios M. Ayahuasca–the healing vine. Int J Soc Psychiatry. 1971 Winter;17(4):256-69. doi: 10.1177/002076407101700402
- Grob CS, McKenna DJ, Callaway JC, Brito GS, Neves ES, Oberlaender G, Saide OL, Labigalini E, Tacla C, Miranda CT, Strassman RJ, Boone KB. Human psychopharmacology of hoasca, a plant hallucinogen used in ritual context in Brazil. J Nerv Ment Dis. 1996 Feb;184(2):86-94. doi: 10.1097/00005053-199602000-00004
- Desmarchelier C, Gurni A, Ciccia G, Giulietti AM. Ritual and medicinal plants of the Ese’ejas of the Amazonian rainforest (Madre de Dios, Perú). J Ethnopharmacol. 1996 May;52(1):45-51. doi: 10.1016/0378-8741(96)01390-6
- McKenna D.J. In: The healing vine: Ayahuasca as medicine in the 21st century. Psychedelic Medicine: New Evidence for Hallucinogenic Substances as Treatments; Winkelman, M.J. Roberts T.B., editor. Vol. 1. Praeger Publishers; 2007. pp. 21–44.
- Barbosa PC, Giglio JS, Dalgalarrondo P. Altered states of consciousness and short-term psychological after-effects induced by the first time ritual use of ayahuasca in an urban context in Brazil. J Psychoactive Drugs. 2005 Jun;37(2):193-201. doi: 10.1080/02791072.2005.10399801
- Labate BC. Consumption of ayahuasca by children and pregnant women: medical controversies and religious perspectives. J Psychoactive Drugs. 2011 Jan-Mar;43(1):27-35. doi: 10.1080/02791072.2011.566498
- Santos R.G., Landeira-Fernandez J., Strassman R.J., Motta V., Cruz A.P. Effects of ayahuasca on psychometric measures of anxiety, panic-like and hopelessness in Santo Daime members. J. Ethnopharmacol. 2007;112:507–513. doi: 10.1016/j.jep.2007.04.012
- Lemlij M. Primitive group treatment. Psychiatr Clin (Basel). 1978;11(1):10-4. doi: 10.1159/000283720
- Labate B.C., Cavnar C. Ayahuasca Shamanism in the Amazon and Beyond. Oxford University Press; Oxford, UK: 2014.
- Yritia M., Riba J., Ortuno J., Ramirez A., Castillo A., Alfaro Y., de la Torre R., Barbanoj M.J. Determination of N,N-dimethyltryptamine and beta-carboline alkaloids in human plasma following oral administration of Ayahuasca. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2002;779:271–281. doi: 10.1016/S1570-0232(02)00397-5
- Hamill J., Hallak J., Dursun S.M., Baker G. Ayahuasca: Psychological and physiologic effects, pharmacology and potential uses in addiction and mental illness. Curr. Neuropharmacol. 2019;17:108–128. doi: 10.2174/1570159X16666180125095902
- Halpern J.H. Hallucinogens and dissociative agents naturally growing in the United States. Pharmacol. Ther. 2004;102:131–138. doi: 10.1016/j.pharmthera.2004.03.003
- Tupper K.W. The globalization of ayahuasca: Harm reduction or benefit maximization? Int. J. Drug Policy. 2008;19:297–303. doi: 10.1016/j.drugpo.2006.11.001
- Pelaez A.I.E. Personality traits in former spanish substance users recovered with ayahuasca. J. Psychoact. Drugs. 2020:1–9. doi: 10.1080/02791072.2020.1752960
- Barbosa P.C., Mizumoto S., Bogenschutz M.P., Strassman R.J. Health status of ayahuasca users. Drug Test Anal. 2012;4:601–609. doi: 10.1002/dta.1383
- Winstock A.R., Kaar S., Borschmann R. Dimethyltryptamine (DMT): Prevalence, user characteristics and abuse liability in a large global sample. J. Psychopharmacol. 2014;28:49–54. doi: 10.1177/0269881113513852
- Dalgarno P. Buying ayahuasca and other entheogens online: A word of caution. Addict. Res. Theory. 2008;16:1–4. doi: 10.1080/16066350701663672
- Lanaro R., Calemi D.B., Togni L.R., Costa J.L., Yonamine M., Cazenave Sde O., Linardi A. Ritualistic use of ayahuasca versus street use of similar substances seized by the police: A key factor involved in the potential for intoxications and overdose? J. Psychoact. Drugs. 2015;47:132–139. doi: 10.1080/02791072.2015.1013202
- Nichols D.E. Psychedelics. Pharmacol. Rev. 2016;68:264–355. doi: 10.1124/pr.115.011478
- Arunotayanun W., Gibbons S. Natural product ‘legal highs’ Nat. Prod. Rep. 2012;29:1304–1316. doi: 10.1039/c2np20068f
- Stiffler J.D. Ayahuasca: From the Amazon to a city near you. Am. J. Addict. 2018;27:648–649. doi: 10.1111/ajad.12833
- International Center for Ethnobotanical Education Research & Service Ayahuasca’s Legal Status. https://www.iceers.org/ayahuasca-legal-status
- Kikura-Hanajiri R., Hayashi M., Saisho K., Goda Y. Simultaneous determination of nineteen hallucinogenic tryptamines/beta-calbolines and phenethylamines using gas chromatography-mass spectrometry and liquid chromatography-electrospray ionisation-mass spectrometry. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2005;825:29–37. doi: 10.1016/j.jchromb.2005.01.041
- Brito-da-Costa AM, Dias-da-Silva D, Gomes NGM, Dinis-Oliveira RJ, Madureira-Carvalho Á. Toxicokinetics and Toxicodynamics of Ayahuasca Alkaloids N,N-Dimethyltryptamine (DMT), Harmine, Harmaline and Tetrahydroharmine: Clinical and Forensic Impact. Pharmaceuticals (Basel). 2020 Oct 23;13(11):334. doi: 10.3390/ph13110334
- Aixalà M., Bouso J.C. Technical Report on Psychoactove Ethnobotanicals. International Center for Ethnobotanical Education Research & Service; Barcelona, Spain: 2018.
- Warren R.J. Fatal nicotine intoxication resulting from the ingestion of “ayahuasca” J. Anal. Toxicol. 2004;28:287.
- Sklerov J., Levine B., Moore K.A., King T., Fowler D. A fatal intoxication following the ingestion of 5-methoxy-N,N-dimethyltryptamine in an ayahuasca preparation. J. Anal. Toxicol. 2005;29:838–841. doi: 10.1093/jat/29.8.838
- Strassman R.J. Adverse reactions to psychedelic drugs. A review of the literature. J. Nerv. Ment. Dis. 1984;172:577–595. doi: 10.1097/00005053-198410000-00001
- Halpern J.H., Pope H.G., Jr. Do hallucinogens cause residual neuropsychological toxicity? Drug Alcohol. Depend. 1999;53:247–256. doi: 10.1016/S0376-8716(98)00129-X
- Reynolds P.C., Jindrich E.J. A mescaline associated fatality. J. Anal. Toxicol. 1985;9:183–184. doi: 10.1093/jat/9.4.183
- Keeler M.H., Reifler C.B. Suicide during an LSD reaction. Am. J. Psychiatry. 1967;123:884–885. doi: 10.1176/ajp.123.7.884
- Schatz H., Mendelblatt F. Solar retinopathy from sun-gazing under the influence of LSD. Br. J. Ophthalmol. 1973;57:270–273. doi: 10.1136/bjo.57.4.270
- Fuller D.G. Severe solar maculopathy associated with the use of lysergic acid diethylamide (LSD) Am. J. Ophthalmol. 1976;81:413–416. doi: 10.1016/0002-9394(76)90295-6
- Ikeda A., Sekiguchi K., Fujita K., Yamadera H., Koga Y. 5-methoxy-N,N-diisopropyltryptamine-induced flashbacks. Am. J. Psychiatry. 2005;162:815. doi: 10.1176/appi.ajp.162.4.815
- Strassman R.J., Qualls C.R., Uhlenhuth E.H., Kellner R. Dose-response study of N,N-dimethyltryptamine in humans. II. Subjective effects and preliminary results of a new rating scale. Arch. Gen. Psychiatry. 1994;51:98–108. doi: 10.1001/archpsyc.1994.03950020022002
- Cakic V., Potkonyak J., Marshall A. Dimethyltryptamine (DMT): Subjective effects and patterns of use among Australian recreational users. Drug Alcohol. Depend. 2010;111:30–37. doi: 10.1016/j.drugalcdep.2010.03.015
- Callaway J.C., Grob C.S. Ayahuasca preparations and serotonin reuptake inhibitors: A potential combination for severe adverse interactions. J. Psychoact. Drugs. 1998;30:367–369. doi: 10.1080/02791072.1998.10399712
- Serotonin syndrome. https://www.mayoclinic.org/diseases-conditions/serotonin-syndrome/symptoms-causes/syc-20354758
- Serotonin syndrome. https://medlineplus.gov/ency/article/007272.htm
- Malcolm B.J., Lee K.C. Ayahuasca: An ancient sacrament for treatment of contemporary psychiatric illness? Ment. Health Clin. 2017;7:39–45. doi: 10.9740/mhc.2017.01.039
- Volpi-Abadie J, Kaye AM, Kaye AD. Serotonin syndrome. Ochsner J. 2013 Winter;13(4):533-40. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3865832
- Cameron L.P., Olson D.E. Dark classics in chemical neuroscience: N,N-dimethyltryptamine (DMT) ACS Chem. Neurosci. 2018;9:2344–2357. doi: 10.1021/acschemneuro.8b00101
- Callaway J.C., McKenna D.J., Grob C.S., Brito G.S., Raymon L.P., Poland R.E., Andrade E.N., Andrade E.O., Mash D.C. Pharmacokinetics of Hoasca alkaloids in healthy humans. J. Ethnopharmacol. 1999;65:243–256. doi: 10.1016/S0378-8741(98)00168-8
- Sanches R.F., de Osorio L.F., Dos Santos R.G., Macedo L.R., Maia-de-Oliveira J.P., Wichert-Ana L., de Araujo D.B., Riba J., Crippa J.A., Hallak J.E. Antidepressant effects of a single dose of ayahuasca in patients with recurrent depression: A SPECT study. J. Clin. Psychopharmacol. 2016;36:77–81. doi: 10.1097/JCP.0000000000000436
- Halpern J.H., Sherwood A.R., Passie T., Blackwell K.C., Ruttenber A.J. Evidence of health and safety in American members of a religion who use a hallucinogenic sacrament. Med. Sci. Monit. 2008;14:Sr15–Sr22. https://medscimonit.com/abstract/index/idArt/865802
- Cakic V, Potkonyak J, Marshall A. Dimethyltryptamine (DMT): subjective effects and patterns of use among Australian recreational users. Drug Alcohol Depend. 2010 Sep 1;111(1-2):30-7. doi: 10.1016/j.drugalcdep.2010.03.015
- Carbonaro TM, Gatch MB. Neuropharmacology of N,N-dimethyltryptamine. Brain Res Bull. 2016 Sep;126(Pt 1):74-88. doi: 10.1016/j.brainresbull.2016.04.016
- Araújo AM, Carvalho F, Bastos Mde L, Guedes de Pinho P, Carvalho M. The hallucinogenic world of tryptamines: an updated review. Arch Toxicol. 2015 Aug;89(8):1151-73. doi: 10.1007/s00204-015-1513-x
- Saavedra JM, Axelrod J. Psychotomimetic N-methylated tryptamines: formation in brain in vivo and in vitro. Science. 1972 Mar 24;175(4028):1365-6. doi: 10.1126/science.175.4028.1365
- Freedland CS, Mansbach RS. Behavioral profile of constituents in ayahuasca, an Amazonian psychoactive plant mixture. Drug Alcohol Depend. 1999 May 3;54(3):183-94. doi: 10.1016/s0376-8716(98)00154-9
- Riba J, McIlhenny EH, Valle M, Bouso JC, Barker SA. Metabolism and disposition of N,N-dimethyltryptamine and harmala alkaloids after oral administration of ayahuasca. Drug Test Anal. 2012 Jul-Aug;4(7-8):610-6. doi: 10.1002/dta.1344
- Fontanilla D, Johannessen M, Hajipour AR, Cozzi NV, Jackson MB, Ruoho AE. The hallucinogen N,N-dimethyltryptamine (DMT) is an endogenous sigma-1 receptor regulator. Science. 2009 Feb 13;323(5916):934-7. doi: 10.1126/science.1166127
- Keiser MJ, Setola V, Irwin JJ, Laggner C, Abbas AI, Hufeisen SJ, Jensen NH, Kuijer MB, Matos RC, Tran TB, Whaley R, Glennon RA, Hert J, Thomas KL, Edwards DD, Shoichet BK, Roth BL. Predicting new molecular targets for known drugs. Nature. 2009 Nov 12;462(7270):175-81. doi: 10.1038/nature08506
- Carbonaro TM, Eshleman AJ, Forster MJ, Cheng K, Rice KC, Gatch MB. The role of 5-HT2A, 5-HT 2C and mGlu2 receptors in the behavioral effects of tryptamine hallucinogens N,N-dimethyltryptamine and N,N-diisopropyltryptamine in rats and mice. Psychopharmacology (Berl). 2015 Jan;232(1):275-84. doi: 10.1007/s00213-014-3658-3
- Strassman RJ, Qualls CR. Dose-response study of N,N-dimethyltryptamine in humans. I. Neuroendocrine, autonomic, and cardiovascular effects. Arch Gen Psychiatry. 1994 Feb;51(2):85-97. doi: 10.1001/archpsyc.1994.03950020009001
- Strassman RJ, Qualls CR, Berg LM. Differential tolerance to biological and subjective effects of four closely spaced doses of N,N-dimethyltryptamine in humans. Biol Psychiatry. 1996 May 1;39(9):784-95. doi: 10.1016/0006-3223(95)00200-6
- Bunzow JR, Sonders MS, Arttamangkul S, Harrison LM, Zhang G, Quigley DI, Darland T, Suchland KL, Pasumamula S, Kennedy JL, Olson SB, Magenis RE, Amara SG, Grandy DK. Amphetamine, 3,4-methylenedioxymethamphetamine, lysergic acid diethylamide, and metabolites of the catecholamine neurotransmitters are agonists of a rat trace amine receptor. Mol Pharmacol. 2001 Dec;60(6):1181-8. doi: 10.1124/mol.60.6.1181
- Premont RT, Gainetdinov RR, Caron MG. Following the trace of elusive amines. Proc Natl Acad Sci U S A. 2001 Aug 14;98(17):9474-5. doi: 10.1073/pnas.181356198
- Schenberg EE. Ayahuasca and cancer treatment. SAGE Open Med. 2013 Oct 18;1:2050312113508389. doi: 10.1177/2050312113508389
- Tittarelli R, Mannocchi G, Pantano F, Romolo FS. Recreational use, analysis and toxicity of tryptamines. Curr Neuropharmacol. 2015 Jan;13(1):26-46. doi: 10.2174/1570159X13666141210222409
- Dos Santos R.G., Balthazar F.M., Bouso J.C., Hallak J.E. The current state of research on ayahuasca: A systematic review of human studies assessing psychiatric symptoms, neuropsychological functioning, and neuroimaging. J. Psychopharmacol. 2016;30:1230–1247. doi: 10.1177/0269881116652578
- Dos Santos R.G., Valle M., Bouso J.C., Nomdedeu J.F., Rodriguez-Espinosa J., McIlhenny E.H., Barker S.A., Barbanoj M.J., Riba J. Autonomic, neuroendocrine, and immunological effects of ayahuasca: A comparative study with d-amphetamine. J. Clin. Psychopharmacol. 2011;31:717–726. doi: 10.1097/JCP.0b013e31823607f6
- Riba J., Valle M., Urbano G., Yritia M., Morte A., Barbanoj M.J. Human pharmacology of ayahuasca: Subjective and cardiovascular effects, monoamine metabolite excretion, and pharmacokinetics. J. Pharmacol. Exp. Ther. 2003;306:73–83. doi: 10.1124/jpet.103.049882
- Kjellgren A., Eriksson A., Norlander T. Experiences of encounters with ayahuasca—“The vine of the soul” J. Psychoact. Drugs. 2009;41:309–315. doi: 10.1080/02791072.2009.10399767
- Checkley S.A., Murray R.M., Oon M.C., Rodnight R., Birley J.L. A longitudinal study of urinary excretion of N,N-dimethyltryptamine in psychotic patients. Br. J. Psychiatry. 1980;137:236–239. doi: 10.1192/bjp.137.3.236
- Lipinski JF, Mandel LR, Ahn HS, Vanden Heuvel WJ, Walker RW. Blood dimethyltryptamine concentrations in psychotic disorders. Biol Psychiatry. 1974 Aug;9(1):89-91.
- Gillin J.C., Kaplan J., Stillman R., Wyatt R.J. The psychedelic model of schizophrenia: The case of N,N-dimethyltryptamine. Am. J. Psychiatry. 1976;133:203–208. doi: 10.1176/ajp.133.2.203
- Araujo A.M., Carvalho F., Bastos Mde L., de Pinho G.P., Carvalho M. The hallucinogenic world of tryptamines: An updated review. Arch. Toxicol. 2015;89:1151–1173. doi: 10.1007/s00204-015-1513-x
- De Araujo D.B., Ribeiro S., Cecchi G.A., Carvalho F.M., Sanchez T.A., Pinto J.P., de Martinis B.S., Crippa J.A., Hallak J.E., Santos A.C. Seeing with the eyes shut: Neural basis of enhanced imagery following Ayahuasca ingestion. Hum. Brain Mapp. 2012;33:2550–2560. doi: 10.1002/hbm.21381
- Bouso J.C., Gonzalez D., Fondevila S., Cutchet M., Fernandez X., Ribeiro Barbosa P.C., Alcazar-Corcoles M.A., Araujo W.S., Barbanoj M.J., Fabregas J.M., et al. Personality, psychopathology, life attitudes and neuropsychological performance among ritual users of Ayahuasca: A longitudinal study. PLoS ONE. 2012;7:e42421. doi: 10.1371/journal.pone.0042421
- Dos Santos R.G., Bouso J.C., Hallak J.E.C. Ayahuasca, dimethyltryptamine, and psychosis: A systematic review of human studies. Ther. Adv. Psychopharmacol. 2017;7:141–157. doi: 10.1177/2045125316689030
- De Rios M.D. Interview with Guillermo Arrevalo, a Shipibo urban shaman, by Roger Rumrrill. J. Psychoact. Drugs. 2005;37:203–207. doi: 10.1080/02791072.2005.10399802
- Langer SZ, Lee CR, Segonzac A, Tateishi T, Esnaud H, Schoemaker H, Winblad B. Possible endocrine role of the pineal gland for 6-methoxytetrahydro-beta-carboline, a putative endogenous neuromodulator of the [3H]imipramine recognition site. Eur J Pharmacol. 1984 Jul 13;102(2):379-80. doi: 10.1016/0014-2999(84)90275-9
- Louis ED, Zheng W, Jurewicz EC, Watner D, Chen J, Factor-Litvak P, Parides M. Elevation of blood beta-carboline alkaloids in essential tremor. Neurology. 2002 Dec 24;59(12):1940-4. doi: 10.1212/01.wnl.0000038385.60538.19
- Frison G, Favretto D, Zancanaro F, Fazzin G, Ferrara SD. A case of beta-carboline alkaloid intoxication following ingestion of Peganum harmala seed extract. Forensic Sci Int. 2008 Aug 6;179(2-3):e37-43. doi: 10.1016/j.forsciint.2008.05.003
- Anderson B.T. Ayahuasca as antidepressant? Psychedelics and styles of reasoning in psychiatry. Anthropol. Consciousness. 2012;23(1):44–59. doi.org/10.1111/j.1556-3537.2012.01056.x
- Rodd R. Reassessing the cultural and psychopharmacological significance of Banisteriopsis caapi: preparation, classification and use among the Piaroa of Southern Venezuela. J Psychoactive Drugs. 2008 Sep;40(3):301-7. doi: 10.1080/02791072.2008.10400645
- Brierley DI, Davidson C. Developments in harmine pharmacology–implications for ayahuasca use and drug-dependence treatment. Prog Neuropsychopharmacol Biol Psychiatry. 2012 Dec 3;39(2):263-72. doi: 10.1016/j.pnpbp.2012.06.001
- Drucker G, Raikoff K, Neafsey EJ, Collins MA. Dopamine uptake inhibitory capacities of beta-carboline and 3,4-dihydro-beta-carboline analogs of N-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) oxidation products. Brain Res. 1990 Feb 12;509(1):125-33. doi: 10.1016/0006-8993(90)90318-6
- Bain J, Plater L, Elliott M, Shpiro N, Hastie CJ, McLauchlan H, Klevernic I, Arthur JS, Alessi DR, Cohen P. The selectivity of protein kinase inhibitors: a further update. Biochem J. 2007 Dec 15;408(3):297-315. doi: 10.1042/BJ20070797
- Husbands SM, Glennon RA, Gorgerat S, Gough R, Tyacke R, Crosby J, Nutt DJ, Lewis JW, Hudson AL. beta-carboline binding to imidazoline receptors. Drug Alcohol Depend. 2001 Oct 1;64(2):203-8. doi: 10.1016/s0376-8716(01)00123-5
- Hopp KH, Cunningham LV, Bromel MC, Schermeister LJ, Khalil SK. In vitro antitrypanosomal activity of certain alkaloids against Trypanosoma lewisi. Lloydia. 1976 Sep-Oct;39(5):375-7.
- Li Y, Sattler R, Yang EJ, Nunes A, Ayukawa Y, Akhtar S, Ji G, Zhang PW, Rothstein JD. Harmine, a natural beta-carboline alkaloid, upregulates astroglial glutamate transporter expression. Neuropharmacology. 2011 Jun;60(7-8):1168-75. doi: 10.1016/j.neuropharm.2010.10.016
- Glennon RA, Dukat M, Grella B, Hong S, Costantino L, Teitler M, Smith C, Egan C, Davis K, Mattson MV. Binding of beta-carbolines and related agents at serotonin (5-HT(2) and 5-HT(1A
- Chen X, Cromer BA, Lynch JW. Molecular determinants of beta-carboline inhibition of the glycine receptor. J Neurochem. 2009 Sep;110(5):1685-94. doi: 10.1111/j.1471-4159.2009.06273.x
- de Castro-Neto EF, da Cunha RH, da Silveira DX, Yonamine M, Gouveia TL, Cavalheiro EA, Amado D, Naffah-Mazzacoratti Mda G. Changes in aminoacidergic and monoaminergic neurotransmission in the hippocampus and amygdala of rats after ayahuasca ingestion. World J Biol Chem. 2013 Nov 26;4(4):141-7. doi: 10.4331/wjbc.v4.i4.141
- Schwarz MJ, Houghton PJ, Rose S, Jenner P, Lees AD. Activities of extract and constituents of Banisteriopsis caapi relevant to parkinsonism. Pharmacol Biochem Behav. 2003 Jun;75(3):627-33. doi: 10.1016/s0091-3057(03)00129-1
- Brierley DI, Davidson C. Harmine augments electrically evoked dopamine efflux in the nucleus accumbens shell. J Psychopharmacol. 2013 Jan;27(1):98-108. doi: 10.1177/0269881112463125
- Grella B, Dukat M, Young R, Teitler M, Herrick-Davis K, Gauthier CB, Glennon RA. Investigation of hallucinogenic and related beta-carbolines. Drug Alcohol Depend. 1998 Apr 1;50(2):99-107. doi: 10.1016/s0376-8716(97)00163-4
- Iurlo M, Leone G, Schilström B, Linnér L, Nomikos G, Hertel P, Silvestrini B, Svensson H. Effects of harmine on dopamine output and metabolism in rat striatum: role of monoamine oxidase-A inhibition. Psychopharmacology (Berl). 2001 Dec;159(1):98-104. doi: 10.1007/s002130100879
- McKenna DJ, Towers GH, Abbott F. Monoamine oxidase inhibitors in South American hallucinogenic plants: tryptamine and beta-carboline constituents of ayahuasca. J Ethnopharmacol. 1984 Apr;10(2):195-223. doi: 10.1016/0378-8741(84)90003-5
- Schmitt KC, Reith ME. Regulation of the dopamine transporter: aspects relevant to psychostimulant drugs of abuse. Ann N Y Acad Sci. 2010 Feb;1187:316-40. doi: 10.1111/j.1749-6632.2009.05148.x
- Adayev T, Wegiel J, Hwang YW. Harmine is an ATP-competitive inhibitor for dual-specificity tyrosine phosphorylation-regulated kinase 1A (Dyrk1A). Arch Biochem Biophys. 2011 Mar 15;507(2):212-8. doi: 10.1016/j.abb.2010.12.024
- Göckler N, Jofre G, Papadopoulos C, Soppa U, Tejedor FJ, Becker W. Harmine specifically inhibits protein kinase DYRK1A and interferes with neurite formation. FEBS J. 2009 Nov;276(21):6324-37. doi: 10.1111/j.1742-4658.2009.07346.x
- Frost D, Meechoovet B, Wang T, Gately S, Giorgetti M, Shcherbakova I, Dunckley T. β-carboline compounds, including harmine, inhibit DYRK1A and tau phosphorylation at multiple Alzheimer’s disease-related sites. PLoS One. 2011 May 6;6(5):e19264. doi: 10.1371/journal.pone.0019264
- Callaway JC, Airaksinen MM, McKenna DJ, Brito GS, Grob CS. Platelet serotonin uptake sites increased in drinkers of ayahuasca. Psychopharmacology (Berl). 1994 Nov;116(3):385-7. doi: 10.1007/BF02245347
- Réus GZ, Stringari RB, de Souza B, Petronilho F, Dal-Pizzol F, Hallak JE, Zuardi AW, Crippa JA, Quevedo J. Harmine and imipramine promote antioxidant activities in prefrontal cortex and hippocampus. Oxid Med Cell Longev. 2010 Sep-Oct;3(5):325-31. doi: 10.4161/oxim.3.5.13109
- Sanches RF, de Lima Osório F, Dos Santos RG, Macedo LR, Maia-de-Oliveira JP, Wichert-Ana L, de Araujo DB, Riba J, Crippa JA, Hallak JE. Antidepressant Effects of a Single Dose of Ayahuasca in Patients With Recurrent Depression: A SPECT Study. J Clin Psychopharmacol. 2016 Feb;36(1):77-81. doi: 10.1097/JCP.0000000000000436
- Bouso JC, Palhano-Fontes F, Rodríguez-Fornells A, Ribeiro S, Sanches R, Crippa JA, Hallak JE, de Araujo DB, Riba J. Long-term use of psychedelic drugs is associated with differences in brain structure and personality in humans. Eur Neuropsychopharmacol. 2015 Apr;25(4):483-92. doi: 10.1016/j.euroneuro.2015.01.008
- Palhano-Fontes F, Andrade KC, Tofoli LF, Santos AC, Crippa JA, Hallak JE, Ribeiro S, de Araujo DB. The psychedelic state induced by ayahuasca modulates the activity and connectivity of the default mode network. PLoS One. 2015 Feb 18;10(2):e0118143. doi: 10.1371/journal.pone.0118143
- Flory JD, Manuck SB, Perel JM, Muldoon MF. A comparison of d, l-fenfluramine and citalopram challenges in healthy adults. Psychopharmacology (Berl). 2004 Jul;174(3):376-80. doi: 10.1007/s00213-003-1763-9
- Mas M, Farré M, de la Torre R, Roset PN, Ortuño J, Segura J, Camí J. Cardiovascular and neuroendocrine effects and pharmacokinetics of 3, 4-methylenedioxymethamphetamine in humans. J Pharmacol Exp Ther. 1999 Jul;290(1):136-45.
- Dos Santos RG, Valle M, Bouso JC, Nomdedéu JF, Rodríguez-Espinosa J, McIlhenny EH, Barker SA, Barbanoj MJ, Riba J. Autonomic, neuroendocrine, and immunological effects of ayahuasca: a comparative study with d-amphetamine. J Clin Psychopharmacol. 2011 Dec;31(6):717-26. doi: 10.1097/JCP.0b013e31823607f6
- Dos Santos RG, Grasa E, Valle M, Ballester MR, Bouso JC, Nomdedéu JF, Homs R, Barbanoj MJ, Riba J. Pharmacology of ayahuasca administered in two repeated doses. Psychopharmacology (Berl). 2012 Feb;219(4):1039-53. doi: 10.1007/s00213-011-2434-x
- Davydova SM, Cheido MA, Gevorgyan MM, Idova GV. Effects of 5-HT2A receptor stimulation and blocking on immune response. Bull Exp Biol Med. 2010 Dec;150(2):219-21. English, Russian. doi: 10.1007/s10517-010-1109-7
- dos Santos RG. Immunological effects of ayahuasca in humans. J Psychoactive Drugs. 2014 Nov-Dec;46(5):383-8. doi: 10.1080/02791072.2014.960113
- Frecska E, Szabo A, Winkelman MJ, Luna LE, McKenna DJ. A possibly sigma-1 receptor mediated role of dimethyltryptamine in tissue protection, regeneration, and immunity. J Neural Transm (Vienna). 2013 Sep;120(9):1295-303. doi: 10.1007/s00702-013-1024-y
- Szabo A. Psychedelics and Immunomodulation: Novel Approaches and Therapeutic Opportunities. Front Immunol. 2015 Jul 14;6:358. doi: 10.3389/fimmu.2015.00358
- Szabo A, Kovacs A, Frecska E, Rajnavolgyi E. Psychedelic N,N-dimethyltryptamine and 5-methoxy-N,N-dimethyltryptamine modulate innate and adaptive inflammatory responses through the sigma-1 receptor of human monocyte-derived dendritic cells. PLoS One. 2014 Aug 29;9(8):e106533. doi: 10.1371/journal.pone.0106533
- House RV, Thomas PT, Bhargava HN. Comparison of the hallucinogenic indole alkaloids ibogaine and harmaline for potential immunomodulatory activity. Pharmacology. 1995 Jun;51(1):56-65. doi: 10.1159/000139317
- Checkley SA, Murray RM, Oon MC, Rodnight R, Birley JL. A longitudinal study of urinary excretion of N,N,-dimethyltryptamine in psychotic patients. Br J Psychiatry. 1980 Sep;137:236-9. doi: 10.1192/bjp.137.3.236
- Rosenberg D.E., Isbell H., Miner E.J. Comparison of a placebo, N,N-dimethyltryptamine, and 6-hydroxy-N-dimethyltryptamine in man. Psychopharmacology (Berl.) 1963;4:39–42. doi.org/10.1007/BF00429362
- Rosenberg D.E., Isbell H., Miner E.J., Logan C.R. The effect of N,N-dimethyltryptamine in human subjects tolerant to lysergic acid diethylamide. Psychopharmacology (Berl.) 1964;5:217–227. doi.org/10.1007/BF00413244
- Bitsios P, Szabadi E, Bradshaw CM. Comparison of the effects of venlafaxine, paroxetine and desipramine on the pupillary light reflex in man. Psychopharmacology (Berl). 1999 Apr;143(3):286-92. doi: 10.1007/s002130050949
- Hartley TR, Lovallo WR, Whitsett TL. Cardiovascular effects of caffeine in men and women. Am J Cardiol. 2004 Apr 15;93(8):1022-6. doi: 10.1016/j.amjcard.2003.12.057
- Pitol DL, Siéssere S, Dos Santos RG, Rosa ML, Hallak JE, Scalize PH, Pereira BF, Iyomasa MM, Semprini M, Riba J, Regalo SC. Ayahuasca Alters Structural Parameters of the Rat Aorta. J Cardiovasc Pharmacol. 2015 Jul;66(1):58-62. doi: 10.1097/FJC.0000000000000243
- Gillin JC, Kaplan J, Stillman R, Wyatt RJ. The psychedelic model of schizophrenia: the case of N,N-dimethyltryptamine. Am J Psychiatry. 1976 Feb;133(2):203-8. doi: 10.1176/ajp.133.2.203
- Anderson BT, Labate BC, Meyer M, Tupper KW, Barbosa PC, Grob CS, Dawson A, McKenna D. Statement on ayahuasca. Int J Drug Policy. 2012 May;23(3):173-5. doi: 10.1016/j.drugpo.2012.02.007
- Paterson NE, Darby WC, Sandhu PS. N,N-Dimethyltryptamine-Induced Psychosis. Clin Neuropharmacol. 2015 Jul-Aug;38(4):141-3. doi: 10.1097/WNF.0000000000000078
- Warren JM, Dham-Nayyar P, Alexander J. Recreational use of naturally occurring dimethyltryptamine–contributing to psychosis? Aust N Z J Psychiatry. 2013 Apr;47(4):398-9. doi: 10.1177/0004867412462749
- Szmulewicz AG, Valerio MP, Smith JM. Switch to mania after ayahuasca consumption in a man with bipolar disorder: a case report. Int J Bipolar Disord. 2015 Feb 24;3:4. doi: 10.1186/s40345-014-0020-y
- Strassman R. Subjective effects of DMT and the development of the hallucinogen rating scale Newsletter Multidisciplinary Assoc. for Psychedelic Studies, . 1992;3(2
- Bresnick T., Levin R. Phenomenal qualities of ayahuasca ingestion and its relation to fringe consciousness and personality. Consciousness Studies. 2006;13(9):5–24.
- Shanon B. Altered temporality. Consciousness Studies. 2001;8(1):35–58.
- Riba J, Rodríguez-Fornells A, Urbano G, Morte A, Antonijoan R, Montero M, Callaway JC, Barbanoj MJ. Subjective effects and tolerability of the South American psychoactive beverage Ayahuasca in healthy volunteers. Psychopharmacology (Berl). 2001 Feb;154(1):85-95. doi: 10.1007/s002130000606
- Beyer S.V. Singing to the plants: A guide to Mestizo shamanism in the upper Amazon. Albuquerque: University of New Mexico Press; 2009.
- Riba J, Rodríguez-Fornells A, Barbanoj MJ. Effects of ayahuasca on sensory and sensorimotor gating in humans as measured by P50 suppression and prepulse inhibition of the startle reflex, respectively. Psychopharmacology (Berl). 2002 Dec;165(1):18-28. doi: 10.1007/s00213-002-1237-5
- Metzner R. Hallucinogenic drugs and plants in psychotherapy and shamanism. J Psychoactive Drugs. 1998 Oct-Dec;30(4):333-41. doi: 10.1080/02791072.1998.10399709
- A HYPOTHESIS OF THE MECHANISMS UNDERLYING VISUAL DISTORTIONS CAUSED BY PSYCHEDELIC DRUGS. https://www.erowid.org/archive/rhodium/pharmacology/visualdistortions.html
- Shanon B. The antipodes of the mind: charting the phenomenology of the ayahuasca experience. Oxford, UK: Oxford University Press; 2007.
- Cloninger CR. A systematic method for clinical description and classification of personality variants. A proposal. Arch Gen Psychiatry. 1987 Jun;44(6):573-88. doi: 10.1001/archpsyc.1987.01800180093014
- Fábregas JM, González D, Fondevila S, Cutchet M, Fernández X, Barbosa PC, Alcázar-Córcoles MÁ, Barbanoj MJ, Riba J, Bouso JC. Assessment of addiction severity among ritual users of ayahuasca. Drug Alcohol Depend. 2010 Oct 1;111(3):257-61. doi: 10.1016/j.drugalcdep.2010.03.024
- Harris R, Gurel L. A study of ayahuasca use in North America. J Psychoactive Drugs. 2012 Jul-Aug;44(3):209-15. doi: 10.1080/02791072.2012.703100
- Barbosa PC, Cazorla IM, Giglio JS, Strassman R. A six-month prospective evaluation of personality traits, psychiatric symptoms and quality of life in ayahuasca-naïve subjects. J Psychoactive Drugs. 2009 Sep;41(3):205-12. doi: 10.1080/02791072.2009.10400530
- Barbosa PC, Strassman RJ, da Silveira DX, Areco K, Hoy R, Pommy J, Thoma R, Bogenschutz M. Psychological and neuropsychological assessment of regular hoasca users. Compr Psychiatry. 2016 Nov;71:95-105. doi: 10.1016/j.comppsych.2016.09.003
- Frecska E, Móré CE, Vargha A, Luna LE. Enhancement of creative expression and entoptic phenomena as after-effects of repeated ayahuasca ceremonies. J Psychoactive Drugs. 2012 Jul-Aug;44(3):191-9. doi: 10.1080/02791072.2012.703099
- Soler J, Elices M, Franquesa A, Barker S, Friedlander P, Feilding A, Pascual JC, Riba J. Exploring the therapeutic potential of Ayahuasca: acute intake increases mindfulness-related capacities. Psychopharmacology (Berl). 2016 Mar;233(5):823-9. doi: 10.1007/s00213-015-4162-0
- Bouso JC, González D, Fondevila S, Cutchet M, Fernández X, Ribeiro Barbosa PC, Alcázar-Córcoles MÁ, Araújo WS, Barbanoj MJ, Fábregas JM, Riba J. Personality, psychopathology, life attitudes and neuropsychological performance among ritual users of Ayahuasca: a longitudinal study. PLoS One. 2012;7(8):e42421. doi: 10.1371/journal.pone.0042421
- Kuypers KP, Riba J, de la Fuente Revenga M, Barker S, Theunissen EL, Ramaekers JG. Ayahuasca enhances creative divergent thinking while decreasing conventional convergent thinking. Psychopharmacology (Berl). 2016 Sep;233(18):3395-403. doi: 10.1007/s00213-016-4377-8
- Burton J.C. Psychological variables predicting intensity and contents of consciousness during ayahuasca experiences: A pilot study. 2010.
- Winkelman M. Drug tourism or spiritual healing? Ayahuasca seekers in Amazonia. J Psychoactive Drugs. 2005 Jun;37(2):209-18. doi: 10.1080/02791072.2005.10399803
- Loizaga-Velder A, Verres R. Therapeutic effects of ritual ayahuasca use in the treatment of substance dependence–qualitative results. J Psychoactive Drugs. 2014 Jan-Mar;46(1):63-72. doi: 10.1080/02791072.2013.873157
- Cavnar C. The effects of ayahuasca ritual participation on gay and lesbian identity. J Psychoactive Drugs. 2014 Jul-Aug;46(3):252-60. doi: 10.1080/02791072.2014.920117
- Dos Santos RG, Osório FL, Crippa JA, Hallak JE. Antidepressive and anxiolytic effects of ayahuasca: a systematic literature review of animal and human studies. Braz J Psychiatry. 2016 Mar;38(1):65-72. doi:10.1590/1516-4446-2015-1701
- Mackey KM, Anderson JK, Williams BE, et al. Evidence Brief: Psychedelic Medications for Mental Health and Substance Use Disorders. Washington (DC): Department of Veterans Affairs (US); 2022 Oct. Available from: https://www.ncbi.nlm.nih.gov/books/NBK586533
- Cakic V, Potkonyak J, Marshall A. Dimethyltryptamine (DMT): subjective effects and patterns of use among Australian recreational users. Drug Alcohol Depend. 2010 Sep 1;111(1-2):30-7. https://doi.org/10.1016/j.drugalcdep.2010.03.015
- Cardenas A.V., Gomez A.P. Urban use of yaje (ayahuasca) in Colombia. Adicciones. 2004;16:323–334.
- Fiedler L., Jungaberle H., Verres R. Motives for the consumption of psychoactive substances demonstrated in the example of the use of ayahuasca in the Santo Daime community. Zeitschr Fur Medizin Psychol. 2011;20:137–144.
- Santos RG, Landeira-Fernandez J, Strassman RJ, Motta V, Cruz AP. Effects of ayahuasca on psychometric measures of anxiety, panic-like and hopelessness in Santo Daime members. J Ethnopharmacol. 2007 Jul 25;112(3):507-13. doi: 10.1016/j.jep.2007.04.012
- Barbosa PC, Mizumoto S, Bogenschutz MP, Strassman RJ. Health status of ayahuasca users. Drug Test Anal. 2012 Jul-Aug;4(7-8):601-9. doi: 10.1002/dta.1383
- Palhano-Fontes F, Alchieri JC, Oliveira JPM, Soares BL, Hallak JEC, Galvão-Coelho N, et al. The therapeutic potentials of ayahuasca in the treatment of depression. In: Labate BC, Cavnar C, editors. The therapeutic use of ayahuasca. Berlin: Springer-Verlag; 2014. pp. p. 23–39.
- Osório Fde L, Sanches RF, Macedo LR, Santos RG, Maia-de-Oliveira JP, Wichert-Ana L, Araujo DB, Riba J, Crippa JA, Hallak JE. Antidepressant effects of a single dose of ayahuasca in patients with recurrent depression: a preliminary report. Braz J Psychiatry. 2015 Jan-Mar;37(1):13-20. doi: 10.1590/1516-4446-2014-1496
- Thomas G, Lucas P, Capler NR, Tupper KW, Martin G. Ayahuasca-assisted therapy for addiction: results from a preliminary observational study in Canada. Curr Drug Abuse Rev. 2013 Mar;6(1):30-42. doi: 10.2174/15733998113099990003
- Santos B.W.L., de Oliveira R.C., Sonsin-Oliveira J., Fagg C.W., Barbosa J.B.F., Caldas E.D. Biodiversity of β-carboline profile of banisteriopsis caapi and ayahuasca, a plant and a brew with neuropharmacological potential. Plants. 2020;9:870. doi: 10.3390/plants9070870
- Nižnanský Ľ, Nižnanská Ž, Kuruc R, Szórádová A, Šikuta J, Zummerová A. Ayahuasca as a Decoction Applied to Human: Analytical Methods, Pharmacology and Potential Toxic Effects. J Clin Med. 2022 Feb 21;11(4):1147. doi: 10.3390/jcm11041147
- dos Santos R.G., Osório F.L., Crippa J.A.S., Hallak J.E.C. Antidepressive and anxiolytic effects of ayahuasca: A systematic literature review of animal and human studies. Braz. J. Psychiatry. 2016;38:65–72. doi: 10.1590/1516-4446-2015-1701
- Domínguez-Clavé E., Soler J., Elices M., Pascual J.C., Álvarez E., de la Fuente Revenga M., Friedlander P., Feilding A., Riba J. Ayahuasca: Pharmacology, neuroscience and therapeutic potential. Brain Res. Bull. 2016;126:89–101. doi: 10.1016/j.brainresbull.2016.03.002
- Dos Santos R.G., Balthazar F.M., Bouso J.C., Hallak J.E.C. The current state of research on ayahuasca: A systematic review of human studies assessing psychiatric symptoms, neuropsychological functioning, and neuroimaging. J. Psychopharmacol. 2016;30:1230–1247. doi: 10.1177/0269881116652578
- de Menezes Galvão A.C., de Almeida R.N., Erick A., dos Santos Silva E.A., Freire F.A.M., Palhano-Fontes F., Onias H., Arcoverde E., Maia-de-Oliveira J.P., de Araújo D.B., et al. Cortisol modulation by ayahuasca in patients with treatment resistant depression and healthy controls. Front. Psychiatry. 2018;9:185. doi: 10.3389/fpsyt.2018.00185
- dos Santos R.G., Hallak J.E.C. Effects of the Natural β-Carboline Alkaloid Harmine, a Main Constituent of Ayahuasca, in Memory and in the Hippocampus: A Systematic Literature Review of Preclinical Studies. J. Psychoact. Drugs. 2017;49:1–10. doi: 10.1080/02791072.2016.1260189
- Galvão-Coelho N.L., de Menezes Galvão A.C., de Almeida R.N., Palhano-Fontes F., Campos Braga I., Lobão Soares B., Maia-de-Oliveira J.P., Perkins D., Sarris J., de Araujo D.B. Changes in inflammatory biomarkers are related to the antidepressant effects of Ayahuasca. J. Psychopharmacol. 2020;34:1125–1133. doi: 10.1177/0269881120936486
- Barbanoj M.J., Riba J., Clos S., Giménez S. Daytime Ayahuasca administration modulates REM and slow-wave sleep in healthy volunteers. Psychopharmacology. 2007;196:315–326. doi: 10.1007/s00213-007-0963-0
- Palhano-Fontes F., Barreto D., Onias H., Andrade K.C., Novaes M.M., Pessoa J.A., Mota-Rolim S.A., Osório F.L., Sanches R., Dos Santos R.G., et al. Rapid antidepressant effects of the psychedelic ayahuasca in treatment-resistant depression: A randomized placebo-controlled trial. Psychol. Med. 2019;49:655–663. doi: 10.1017/S0033291718001356
- Uthaug M.V., van Oorsouw K., Kuypers K.P.C., van Boxtel M., Broers N.J., Mason N.L., Toennes S.W., Riba J., Ramaekers J.G. Sub-acute and long-term effects of ayahuasca on affect and cognitive thinking style and their association with ego dissolution. Psychopharmacology. 2018;235:2979–2989. doi: 10.1007/s00213-018-4988-3
- Frecska E., Bokor P., Winkelman M. The therapeutic potentials of ayahuasca: Possible effects against various diseases of civilization. Front. Pharmacol. 2016;7:35. doi: 10.3389/fphar.2016.00035
- Liester M.B., Prickett J.I. Hypotheses regarding the mechanisms of ayahuasca in the treatment of addictions. J. Psychoactive Drugs. 2012;44:200–208. doi: 10.1080/02791072.2012.704590
- Fábregas J.M., González D., Fondevila S., Cutchet M., Fernández X., Barbosa P.C.R., Alcázar-Córcoles M.Á., Barbanoj M.J., Riba J., Bouso J.C. Assessment of addiction severity among ritual users of ayahuasca. Drug Alcohol. Depend. 2010;111:257–261. doi: 10.1016/j.drugalcdep.2010.03.024
- Barbosa P.C.R., Mizumoto S., Bogenschutz M.P., Strassman R.J. Health status of ayahuasca users. Drug Test. Anal. 2012;4:601–609. doi: 10.1002/dta.1383
- Schenberg E.E., Alexandre M., Filev R., Cravo M., Muthukumaraswamy S.D. Acute Biphasic Effects of Ayahuasca. PLoS ONE. 2015;10:e0137202. doi: 10.1371/journal.pone.0137202
- Sanches R.F., De Lima Osório F., Santos R.G.D., Macedo L.R.H., Maia-De-Oliveira J.P., Wichert-Ana L., De Araujo D.B., Riba J., Crippa J.A.S., Hallak J.E.C. Antidepressant effects of a single dose of ayahuasca in patients with recurrent depression a SPECT study. J. Clin. Psychopharmacol. 2016;36:77–81. doi: 10.1097/JCP.0000000000000436
- Ona G., Kohek M., Massaguer T., Gomariz A., Jiménez D.F., Dos Santos R.G., Hallak J.E.C., Alcázar-Córcoles M.Á., Bouso J.C. Ayahuasca and Public Health: Health Status, Psychosocial Well-Being, Lifestyle, and Coping Strategies in a Large Sample of Ritual Ayahuasca Users. J. Psychoact. Drugs. 2019;51:135–145. doi: 10.1080/02791072.2019.1567961
- De Almeida R.N., Galvão A.C.D.M., Da Silva F.S., Silva E.A., Palhano-Fontes F., Maia-De-Oliveira J.P., De Araújo L.-S.B., Lobão-Soares B., Galvão-Coelho N.L. Modulation of Serum Brain-Derived Neurotrophic Factor by a Single Dose of Ayahuasca: Observation From a Randomized Controlled Trial. Front. Psychol. 2019;10:1234. doi: 10.3389/fpsyg.2019.01234
- Hamill J., Hallak J., Dursun S.M., Baker G. Ayahuasca: Psychological and Physiologic Effects, Pharmacology and Potential Uses in Addiction and Mental Illness. Curr. Neuropharmacol. 2018;17:108–128. doi: 10.2174/1570159X16666180125095902
- Morgenstern J, Langenbucher J, Labouvie EW. The generalizability of the dependence syndrome across substances: an examination of some properties of the proposed DSM-IV dependence criteria. Addiction. 1994 Sep;89(9):1105-13. doi: 10.1111/j.1360-0443.1994.tb02787.x
- Liester MB, Prickett JI. Hypotheses regarding the mechanisms of ayahuasca in the treatment of addictions. J Psychoactive Drugs. 2012 Jul-Aug;44(3):200-8. doi: 10.1080/02791072.2012.704590
- Stahl S.M. Stahl’s essential psychopharmacology: Neuroscientific basic and practical applications. Cambridge, UK: Cambridge University Press; 2008.
- Aricioglu-Kartal F, Kayir H, Tayfun Uzbay I. Effects of harman and harmine on naloxone-precipitated withdrawal syndrome in morphine-dependent rats. Life Sci. 2003 Sep 19;73(18):2363-71. doi: 10.1016/s0024-3205(03)00647-7
- Miralles A, Esteban S, Sastre-Coll A, Moranta D, Asensio VJ, García-Sevilla JA. High-affinity binding of beta-carbolines to imidazoline I2B receptors and MAO-A in rat tissues: norharman blocks the effect of morphine withdrawal on DOPA/noradrenaline synthesis in the brain. Eur J Pharmacol. 2005 Aug 22;518(2-3):234-42. doi: 10.1016/j.ejphar.2005.06.023
- Glick SD, Kuehne ME, Raucci J, Wilson TE, Larson D, Keller RW Jr, Carlson JN. Effects of iboga alkaloids on morphine and cocaine self-administration in rats: relationship to tremorigenic effects and to effects on dopamine release in nucleus accumbens and striatum. Brain Res. 1994 Sep 19;657(1-2):14-22. doi: 10.1016/0006-8993(94)90948-2
- Halpern J.H. In: Hallucinogens in the treatment of alcoholism and other addictions. Psychedelic Medicine: New Evidence for Hallucinogenic Substances as Treatments; Winkelman, M.J. Roberts T.B., editor. Vol. 2 Westport, CT: Praeger; 2007.
- Doering-Silveira E, Grob CS, de Rios MD, Lopez E, Alonso LK, Tacla C, Da Silveira DX. Report on psychoactive drug use among adolescents using ayahuasca within a religious context. J Psychoactive Drugs. 2005 Jun;37(2):141-4. doi: 10.1080/02791072.2005.10399794
- Oliveira-Lima AJ, Santos R, Hollais AW, Gerardi-Junior CA, Baldaia MA, Wuo-Silva R, Yokoyama TS, Costa JL, Malpezzi-Marinho EL, Ribeiro-Barbosa PC, Berro LF, Frussa-Filho R, Marinho EA. Effects of ayahuasca on the development of ethanol-induced behavioral sensitization and on a post-sensitization treatment in mice. Physiol Behav. 2015 Apr 1;142:28-36. doi: 10.1016/j.physbeh.2015.01.032
- Dos Santos RG, Osório FL, Crippa JA, Riba J, Zuardi AW, Hallak JE. Antidepressive, anxiolytic, and antiaddictive effects of ayahuasca, psilocybin and lysergic acid diethylamide (LSD): a systematic review of clinical trials published in the last 25 years. Ther Adv Psychopharmacol. 2016 Jun;6(3):193-213. doi: 10.1177/2045125316638008
- de Sousa GM, de Oliveira Tavares VD, de Menezes Galvão AC, de Almeida RN, Palhano-Fontes F, Lobão-Soares B, de Morais Freire FA, Nunes EA, Maia-de-Oliveira JP, Perkins D, Sarris J, de Araujo DB, Galvão-Coelho NL. Moderators of ayahuasca’s biological antidepressant action. Front Psychiatry. 2022 Dec 5;13:1033816. doi: 10.3389/fpsyt.2022.1033816
- Osório F.L., de Macedo L.R.H., de Sousa J.P.M., Pinto J., Quevedo J., Crippa J.A.d.S., Hallak J.C.E. The ethnopharmacology of ayahuasca; dos Santos, R.G., Ed.; Transworld Research Network: Kerala, India. 2011. The therapeutic potential of harmine and ayahuasca in depression: Evidence from exploratory animal and human studies. ; pp. 75–85.
- Farzin D, Mansouri N. Antidepressant-like effect of harmane and other beta-carbolines in the mouse forced swim test. Eur Neuropsychopharmacol. 2006 Jul;16(5):324-8. doi: 10.1016/j.euroneuro.2005.08.005
- Fortunato JJ, Réus GZ, Kirsch TR, Stringari RB, Stertz L, Kapczinski F, Pinto JP, Hallak JE, Zuardi AW, Crippa JA, Quevedo J. Acute harmine administration induces antidepressive-like effects and increases BDNF levels in the rat hippocampus. Prog Neuropsychopharmacol Biol Psychiatry. 2009 Nov 13;33(8):1425-30. doi: 10.1016/j.pnpbp.2009.07.021
- Fortunato JJ, Réus GZ, Kirsch TR, Stringari RB, Fries GR, Kapczinski F, Hallak JE, Zuardi AW, Crippa JA, Quevedo J. Chronic administration of harmine elicits antidepressant-like effects and increases BDNF levels in rat hippocampus. J Neural Transm (Vienna). 2010 Oct;117(10):1131-7. doi: 10.1007/s00702-010-0451-2
- Fortunato JJ, Réus GZ, Kirsch TR, Stringari RB, Fries GR, Kapczinski F, Hallak JE, Zuardi AW, Crippa JA, Quevedo J. Effects of beta-carboline harmine on behavioral and physiological parameters observed in the chronic mild stress model: further evidence of antidepressant properties. Brain Res Bull. 2010 Mar 16;81(4-5):491-6. doi: 10.1016/j.brainresbull.2009.09.008
- Samoylenko V, Rahman MM, Tekwani BL, Tripathi LM, Wang YH, Khan SI, Khan IA, Miller LS, Joshi VC, Muhammad I. Banisteriopsis caapi, a unique combination of MAO inhibitory and antioxidative constituents for the activities relevant to neurodegenerative disorders and Parkinson’s disease. J Ethnopharmacol. 2010 Feb 3;127(2):357-67. doi: 10.1016/j.jep.2009.10.030
- Urani A, Roman FJ, Phan VL, Su TP, Maurice T. The antidepressant-like effect induced by sigma(1)-receptor agonists and neuroactive steroids in mice submitted to the forced swimming test. J Pharmacol Exp Ther. 2001 Sep;298(3):1269-79.
- Wang J, Mack AL, Coop A, Matsumoto RR. Novel sigma (sigma) receptor agonists produce antidepressant-like effects in mice. Eur Neuropsychopharmacol. 2007 Nov;17(11):708-16. doi: 10.1016/j.euroneuro.2007.02.007
- Piletz JE, Zhu H, Ordway G, Stockmeier C, Dilly G, Reis D, Halaris A. Imidazoline receptor proteins are decreased in the hippocampus of individuals with major depression. Biol Psychiatry. 2000 Nov 1;48(9):910-9. doi: 10.1016/s0006-3223(00)00892-1
- Finn DP, Martí O, Harbuz MS, Vallès A, Belda X, Márquez C, Jessop DS, Lalies MD, Armario A, Nutt DJ, Hudson AL. Behavioral, neuroendocrine and neurochemical effects of the imidazoline I2 receptor selective ligand BU224 in naive rats and rats exposed to the stress of the forced swim test. Psychopharmacology (Berl). 2003 May;167(2):195-202. doi: 10.1007/s00213-003-1392-3
- Halaris A, Piletz JE. Relevance of imidazoline receptors and agmatine to psychiatry: a decade of progress. Ann N Y Acad Sci. 2003 Dec;1009:1-20. doi: 10.1196/annals.1304.001
- Paterson LM, Robinson ES, Nutt DJ, Hudson AL. In vivo estimation of imidazoline(2) binding site turnover. Ann N Y Acad Sci. 2003 Dec;1009:367-70. doi: 10.1196/annals.1304.049
- García-Sevilla JA, Escribá PV, Guimón J. Imidazoline receptors and human brain disorders. Ann N Y Acad Sci. 1999 Jun 21;881:392-409. doi: 10.1111/j.1749-6632.1999.tb09388.x
- Bouayed J, Rammal H, Soulimani R. Oxidative stress and anxiety: relationship and cellular pathways. Oxid Med Cell Longev. 2009 Apr-Jun;2(2):63-7. doi: 10.4161/oxim.2.2.7944
- Jacob MS, Presti DE. Endogenous psychoactive tryptamines reconsidered: an anxiolytic role for dimethyltryptamine. Med Hypotheses. 2005;64(5):930-7. doi: 10.1016/j.mehy.2004.11.005
- Sarris J, McIntyre E, Camfield DA. Plant-based medicines for anxiety disorders, part 2: a review of clinical studies with supporting preclinical evidence. CNS Drugs. 2013 Apr;27(4):301-19. doi: 10.1007/s40263-013-0059-9. Erratum in: CNS Drugs. 2013 Aug;27(8):675. Dosage error in article text.
- Graeff FG, Guimarães FS, De Andrade TG, Deakin JF. Role of 5-HT in stress, anxiety, and depression. Pharmacol Biochem Behav. 1996 May;54(1):129-41. doi: 10.1016/0091-3057(95)02135-3
- Fotiou E, Gearin AK. Purging and the body in the therapeutic use of ayahuasca. Soc Sci Med. 2019;239: 112532. doi: 10.1016/j.socscimed.2019.112532
- dos Santos R, Hallack J. Ayahuasca, an ancient substance with traditional and contemporary use in neuropsychiatry and neuroscience. Epilepsy & Behaviour. 2019.
- Bouso JC, dos Santos RG, Sánchez-Avilés C, Ona G, Grob SG, da Silveira DX, et al. 2021. Ayahuasca Technical Report. https://www.iceers.org/wp-content/uploads/2020/05/Ayahuasca_Technical_Report_ICEERS_2021_ENG.pdf
- Gómez-Sousa M, Jiménez-Garrido DF, Ona G, Dos Santos RG, Hallak JEC, Alcázar-Córcoles MÁ, et al. Acute Psychological Adverse Reactions in First-Time Ritual Ayahuasca Users: A Prospective Case Series. J Clin Psychopharmacol. 2021;41(2): 163–171. doi: 10.1097/JCP.0000000000001343
- Heise CW, Brooks DE. Ayahuasca Exposure: Descriptive Analysis of Calls to US Poison Control Centers from 2005 to 2015. J Med Toxicol. 2017;13(3): 245–248. doi: 10.1007/s13181-016-0593-1
- Durante Í, Dos Santos RG, Bouso JC, Hallak JE. Risk assessment of ayahuasca use in a religious context: self-reported risk factors and adverse effects. Braz J Psychiatry. 2021 Jul-Aug;43(4):362-369. doi: 10.1590/1516-4446-2020-0913
- Dos Santos RG, Bouso JC, Hallak JEC. Ayahuasca, dimethyltryptamine, and psychosis: a systematic review of human studies. Ther Adv Psychopharmacol. 2017;7(4): 141–157. doi: 10.1177/2045125316689030
- Bilhimer MH, Schult RF, Higgs KV, Wiegand TJ, Gorodetsky RM, Acquisto NM. Acute Intoxication following Dimethyltryptamine Ingestion. Case Rep Emerg Med. 2018;2018: 3452691. doi: 10.1155/2018/3452691
- Cerón Tapia HR, González Guzmán MA, Córdoba Ortiz SA. Ayahuasca-Induced Psychosis: A Case Report. Rev Colomb Psiquiatr (Engl Ed). 2022;51(3): 236–239. doi: 10.1016/j.rcpeng.2022.08.008
- Palma-Álvarez RF, Grau-López L, Ros-Cucurull E, Abad AC, Dualde J, Robles-Martínez M, et al. Psychosis induced by abuse of ayahuasca: a case report. Rev Colomb Psiquiatr (Engl Ed). 2021;50(1): 43–46. doi: 10.1016/j.rcp.2019.10.005
- Jiménez-Garrido DF, Gómez-Sousa M, Oña G, dos Santos RG, Hallak JEC, Alcázar-Córcoles MÁ, et al. Effects of ayahuasca on mental health and quality of life in naïve users: a longitudinal and cross-sectional study combination. Sci. Rep. 2020;10(1): 4075. doi: 10.1038/s41598-020-61169-x
- Riba J, Rodríguez-Fornells A, Urbano G, Morte A, Antonijoan R, Montero M, et al. Subjective effects and tolerability of the South American psychoactive beverage ayahuasca in healthy volunteers. Psychopharmacology. 2001;154(1): 85–95. doi: 10.1007/s002130000606
- Riba J, Valle M, Urbano G, Yritia M, Morte A, Barbanoj MJ. Human pharmacology of ayahuasca: subjective and cardiovascular effects, monoamine metabolite excretion, and pharmacokinetics. Journal of Pharmacology and Experimental Therapeutics. 2003;306(1): 73–83. doi: 10.1124/jpet.103.049882
- Riba J, Barbanoj MJ. Bringing ayahuasca to the clinical research laboratory. J Psychoactive Drugs. 2005;37(2): 219–30. doi: 10.1080/02791072.2005.10399804
- de Araujo DB, Ribeiro S, Cecchi GA, Carvalho FM, Sanchez TA, Pinto JP, et al. Seeing with the eyes shut: neural basis of enhanced imagery following ayahuasca ingestion. Human Brain Mapping. 2012;33(11): 2550–2560. doi: 10.1002/hbm.21381
- Rocha JM, Rossi GN, de Lima Osório F, Bouso JC, de Oliveira Silveira G, Yonamine M, et al. Effects of Ayahuasca on the Recognition of Facial Expressions of Emotions in Naive Healthy Volunteers: A Pilot, Proof-of-Concept, Randomized Controlled Trial. J Clin Psychopharmacol. 2021;41(3): 267–274. doi: 10.1097/JCP.0000000000001396
- Bouso JC, Andión Ó, Sarris JJ, Scheidegger M, Tófoli LF, Opaleye ES, Schubert V, Perkins D. Adverse effects of ayahuasca: Results from the Global Ayahuasca Survey. PLOS Glob Public Health. 2022 Nov 16;2(11):e0000438. doi: 10.1371/journal.pgph.0000438
- Perkins D, Schubert V, Simonová H, Tófoli LF, Bouso JC, Horák M, et al. Influence of Context and Setting on the Mental Health and Wellbeing Outcomes of Ayahuasca Drinkers: Results of a Large International Survey. Front Pharmacol. 2021;12: 623979. doi: 10.3389/fphar.2021.623979
- Bouso JC, González D, Fondevila S, Cutchet M, Fernández X, Ribeiro Barbosa PC, et al. Personality, psychopathology, life attitudes and neuropsychological performance among ritual users of Ayahuasca: a longitudinal study. PLoS One. 2012;7(8): e42421. doi: 10.1371/journal.pone.0042421
- Barbosa PC, Strassman RJ, da Silveira DX, Areco K, Hoy R, Pommy J, et al. Psychological and neuropsychological assessment of regular hoasca users. Compr. Psychiatry. 2016; 71:95–105. doi: 10.1016/j.comppsych.2016.09.003
- da Silveira DX, Grob CS, Dobkin de Rios M, Lopez E, Alonso LK, Tacla C, et al. Ayahuasca in adolescence: a preliminary psychiatric assessment. J. Psychoactive Drugs. 2005;37(2): 129–133. doi: 10.1080/02791072.2005.10399792
- Doering-Silveira E, Lopez E, Grob CS, Dobkin de Rios M, Alonso LK, Tacla C, et al. Ayahuasca in adolescence: a neuropsychological assessment. J. Psychoactive Drugs. 2005;37(2): 123–128. doi: 10.1080/02791072.2005.10399791
- Ona G, Kohek M, Massaguer T, Gomariz A, Jiménez DF, Dos Santos RG, et al. Ayahuasca and Public Health: Health Status, Psychosocial Well-Being, Lifestyle, and Coping Strategies in a Large Sample of Ritual Ayahuasca Users. J Psychoactive Drugs. 2019;51(2): 135–145. doi: 10.1080/02791072.2019.1567961
- Kohek M, Ona G, van Elk M, Dos Santos RG, Hallak JEC, Alcázar-Córcoles MÁ, et al. Ayahuasca and Public Health II: Health Status in a Large Sample of Ayahuasca-Ceremony Participants in the Netherlands. J Psychoactive Drugs. 2022. May 28. doi: 10.1080/02791072.2022.2077155
- Callaway JC, Grob CS. Ayahuasca preparations and serotonin reuptake inhibitors: a potential combination for severe adverse interactions. J Psychoactive Drugs. 1998 Oct-Dec;30(4):367-9. doi: 10.1080/02791072.1998.10399712
- Balíková M. Collective poisoning with hallucinogenous herbal tea. Forensic Sci Int. 2002 Aug 14;128(1-2):50-2. doi: 10.1016/s0379-0738(02)00162-7
- Zhao T, He YQ, Wang J, Ding KM, Wang CH, Wang ZT. Inhibition of human cytochrome P450 enzymes 3A4 and 2D6 by β-carboline alkaloids, harmine derivatives. Phytother Res. 2011 Nov;25(11):1671-7. doi: 10.1002/ptr.3458
- Strassman R.J., Qualls C.R., Berg L.M. Differential tolerance to biological and subjective effects of four closely spaced doses of N,N-dimethyltryptamine in humans. Biol. Psychiatry. 1996;39:784–795. doi: 10.1016/0006-3223(95)00200-6
- Bouso J.C., Fabregas J.M., Antonijoan R.M., Rodriguez-Fornells A., Riba J. Acute effects of ayahuasca on neuropsychological performance: Differences in executive function between experienced and occasional users. Psychopharmacology. 2013;230:415–424. doi: 10.1007/s00213-013-3167-9
- Strassman R.J., Qualls C.R. Dose-response study of N,N-dimethyltryptamine in humans. I. Neuroendocrine, autonomic, and cardiovascular effects. Arch. Gen. Psychiatry. 1994;51:85–97. doi: 10.1001/archpsyc.1994.03950020009001
- Dos Santos R.G., Grasa E., Valle M., Ballester M.R., Bouso J.C., Nomdedeu J.F., Homs R., Barbanoj M.J., Riba J. Pharmacology of ayahuasca administered in two repeated doses. Psychopharmacology. 2012;219:1039–1053. doi: 10.1007/s00213-011-2434-x
- Cole J.M., Pieper W.A. The effects of N,N-dimethyltryptamine on operant behavior in squirrel monkeys. Psychopharmacologia. 1973;29:107–112. doi: 10.1007/BF00422642
- Gillin JC, Cannon E, Magyar R, Schwartz M, Wyatt RJ. Failure of N,N-dimethyltryptamine to evoke tolerance in cats. Biol Psychiatry. 1973 Dec;7(3):213-20.
- Rosenberg D.E., Isbell H., Miner E.J., Logan C.R. The effect of N,N-dimethyltryptamine in human subjects tolerant to lysergic acid diethylamide. Psychopharmacologia. 1964;5:217–227. doi: 10.1007/BF00413244
- Balestrieri A., Fontanari D. Acquired and crossed tolerance to mescaline, LSD-25, and BOL-148. AMA Arch. Gen. Psychiatry. 1959;1:279–282. doi: 10.1001/archpsyc.1959.03590030063008
- Appel J.B., Freedman D.X. Tolerance and cross-tolerance among psychotomimetic drugs. Psychopharmacologia. 1968;13:267–274. doi: 10.1007/BF00401404
- Fantegrossi WE, Woods JH, Winger G. Transient reinforcing effects of phenylisopropylamine and indolealkylamine hallucinogens in rhesus monkeys. Behav Pharmacol. 2004 Mar;15(2):149-57. doi: 10.1097/00008877-200403000-00007
- Cole JM, Pieper WA. The effects of N,N-dimethyltryptamine on operant behavior in squirrel monkeys. Psychopharmacologia. 1973 Mar 16;29(2):107-12. doi: 10.1007/BF00422642
- Kovacic B, Domino EF. Tolerance and limited cross-tolerance to the effects of N, N-dimethyltryptamine (DMT) and lysergic acid diethylamide-25 (LSD) on food-rewarded bar pressing in the rat. J Pharmacol Exp Ther. 1976 Jun;197(3):495-502.
- Airaksinen MM, Lecklin A, Saano V, Tuomisto L, Gynther J. Tremorigenic effect and inhibition of tryptamine and serotonin receptor binding by beta-carbolines. Pharmacol Toxicol. 1987 Jan;60(1):5-8. doi: 10.1111/j.1600-0773.1987.tb01711.x
- Bouso JC, Fábregas JM, Antonijoan RM, Rodríguez-Fornells A, Riba J. Acute effects of ayahuasca on neuropsychological performance: differences in executive function between experienced and occasional users. Psychopharmacology (Berl). 2013 Dec;230(3):415-24. doi: 10.1007/s00213-013-3167-9
- Pic-Taylor A, da Motta LG, de Morais JA, Junior WM, Santos Ade F, Campos LA, Mortari MR, von Zuben MV, Caldas ED. Behavioural and neurotoxic effects of ayahuasca infusion (Banisteriopsis caapi and Psychotria viridis) in female Wistar rat. Behav Processes. 2015 Sep;118:102-10. doi: 10.1016/j.beproc.2015.05.004
- Dos Santos RG. Toxicity of chronic ayahuasca administration to the pregnant rat: how relevant it is regarding the human, ritual use of ayahuasca? Birth Defects Res B Dev Reprod Toxicol. 2010 Dec;89(6):533-5; author reply 531-2.. doi: 10.1002/bdrb.20272
- Stuckey DE, Lawson R, Luna LE. EEG gamma coherence and other correlates of subjective reports during ayahuasca experiences. J Psychoactive Drugs. 2005 Jun;37(2):163-78. doi: 10.1080/02791072.2005.10399798
- Oliveira CD, Moreira CQ, de Sá LR, Spinosa Hde S, Yonamine M. Maternal and developmental toxicity of ayahuasca in Wistar rats. Birth Defects Res B Dev Reprod Toxicol. 2010 Jun;89(3):207-12. doi: 10.1002/bdrb.20244
- Gardner D, Riet-Correa F, Lemos D, Welch K, Pfister J, Panter K. Teratogenic effects of Mimosa tenuiflora in a rat model and possible role of N-methyl- and N,N-dimethyltryptamine. J Agric Food Chem. 2014 Jul 30;62(30):7398-401. doi: 10.1021/jf5005176
- Colaco C.S., Alves S.S., Nolli L.M., Pinheiro W.O., de Oliveira D.G.R., Santos B.W.L., Pic-Taylor A., Mortari M.R., Caldas E.D. Toxicity of ayahuasca after 28 days daily exposure and effects on monoamines and brain-derived neurotrophic factor (BDNF) in brain of Wistar rats. Metab. Brain Dis. 2020 doi: 10.1007/s11011-020-00547-w
- Pic-Taylor A., da Motta L.G., de Morais J.A., Junior W.M., Santos Ade F., Campos L.A., Mortari M.R., von Zuben M.V., Caldas E.D. Behavioural and neurotoxic effects of ayahuasca infusion (Banisteriopsis caapi and Psychotria viridis) in female Wistar rat. Behav. Process. 2015;118:102–110. doi: 10.1016/j.beproc.2015.05.004
- Halpern J.H., Sherwood A.R., Hudson J.I., Yurgelun-Todd D., Pope H.G., Jr. Psychological and cognitive effects of long-term peyote use among Native Americans. Biological. Psychiatry. 2005;58:624–631. doi: 10.1016/j.biopsych.2005.06.038
- Krebs T.S., Johansen P.O. Psychedelics and mental health: A population study. PLoS ONE. 2013;8:e63972. doi: 10.1371/journal.pone.0063972
- Mahmoudian M., Jalilpour H., Salehian P. Toxicity of Peganum harmala: Review and a case report. Iranian J. Pharmacol. Ther. 2002;1:1–4.
- Moshiri M, Etemad L, Javidi S, Alizadeh A. Peganum harmala intoxication, a case report. Avicenna J Phytomed. 2013 Summer;3(3):288-92. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4075715
- Yuruktumen A., Karaduman S., Bengi F., Fowler J. Syrian rue tea: A recipe for disaster. Clin. Toxicol. 2008;46:749–752. doi: 10.1080/15563650701323205
- Ben Salah N, Amamou M, Jerbi Z, Ben Salah F, Yacoub M. Un cas de surdosage en Peganum harmala L [A case of overdose with Peganum harmala L]. J Toxicol Clin Exp. 1986 Sep-Oct;6(5):319-22. French.
- Frison G., Favretto D., Zancanaro F., Fazzin G., Ferrara S.D. A case of beta-carboline alkaloid intoxication following ingestion of Peganum harmala seed extract. Forensic Sci. Int. 2008;179:e37–e43. doi: 10.1016/j.forsciint.2008.05.003
- dos Santos RG. Safety and side effects of ayahuasca in humans–an overview focusing on developmental toxicology. J Psychoactive Drugs. 2013 Jan-Mar;45(1):68-78. doi: 10.1080/02791072.2013.763564
- Kamel S.H., Ibrahim T.M., Hamza S.M. Effect of harmine and harmaline hydrochloride on pregnancy in white rats. Zent. Vet. A. 1971;18:230–233. doi: 10.1111/j.1439-0442.1971.tb00573.x
- Poulson E., Robson J.M. The effect of amine oxidase inhibitors on pregnancy. J. Endocrinol. 1963;27:147–155. doi: 10.1677/joe.0.0270147
- Oliveira C.D., Moreira C.Q., de Sa L.R., Spinosa Hde S., Yonamine M. Maternal and developmental toxicity of ayahuasca in Wistar rats. Birth. Defects. Res. B Dev. Reprod. Toxicol. 2010;89:207–212. doi: 10.1002/bdrb.20244