What is MCH
MCH is short for mean corpuscular hemoglobin is also called mean cell hemoglobin. MCH (mean corpuscular hemoglobin) is a calculation of the average amount of hemoglobin inside a single red blood cell. MCH blood test shows hemoglobin amount of red blood cells in circulation. Normal levels of MCH (mean corpuscular hemoglobin) are between 27 and 31 picograms (pg) of hemoglobin per red blood cell. To calculate MCH, the hemoglobin is divided by red blood cell, yielding an average amount of hemoglobin per red blood cell. In contrast to MCH (mean corpuscular hemoglobin), MCHC (mean corpuscular hemoglobin concentration) indicates the amount of hemoglobin per unit volume and MCHC correlates the hemoglobin content with the volume of the cell. It is expressed as g/dl of red blood cells or as a percentage value. The normal values for MCHC (mean corpuscular hemoglobin concentration) are 34 ± 2 g/dl.
Figure 1. Mean corpuscular hemoglobin formula
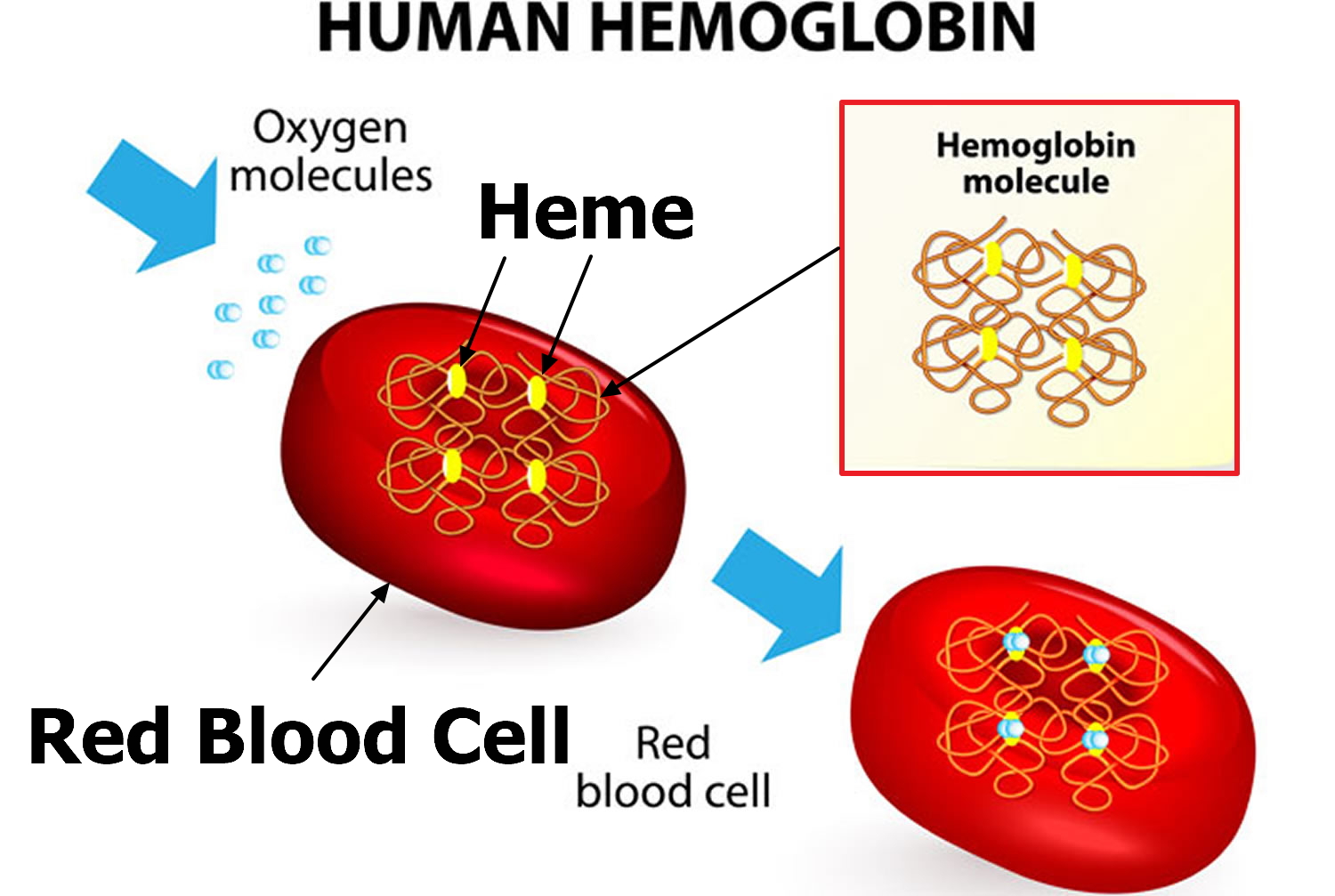
Hemoglobin is the iron-containing protein found in all red blood cells (RBCs) that gives the cells their characteristic red color. Hemoglobin enables red blood cells to bind to oxygen in the lungs and carry it to tissues and organs throughout the body. Hemoglobin also helps transport a small portion of carbon dioxide, a product of cell metabolism, from tissues and organs to the lungs, where it is exhaled.
The MCH blood test is done as a component of a blood test called a Complete Blood Count (CBC), which evaluates the composition of the blood, checking hematocrit, white blood cells (WBC) and platelets as well as hemoglobin and red blood cells (red blood cell). It is usually ordered to get an overview of general health. MCH is not measured directly, but calculated based on the hemoglobin value (Hb), which is the total measure of hemoglobin in the blood, and the red blood cell, which is the number of red blood cells in the blood.
A Complete Blood Count (CBC) is commonly included in bloodwork ordered by your doctor during an annual checkup. Also included in this standard test bundling is a comprehensive metabolic panel, urinalysis, hemoglobin A1c, and lipid panel.
The hemoglobin test measures the amount of hemoglobin in a person’s sample of blood. A hemoglobin level can be performed alone or with a hematocrit (Hct), a test that measures the proportion of blood that is made up of red blood cells, to quickly evaluate an individual’s red blood cells. Red blood cells, which make up about 40% (ranging 37-49%) of the blood’s volume, are produced in the bone marrow and are released into the bloodstream when they are, or nearly are, mature. The typical lifespan of an red blood cell is 120 days, and the bone marrow must continually produce new red blood cells to replace those that age and degrade or are lost through bleeding.
Several diseases and conditions can affect red blood cells and consequently the level of hemoglobin in the blood. In general, the hemoglobin level and hematocrit rise when the number of red blood cells increases. The hemoglobin level and hematocrit fall to less than normal when there is a drop in production of red blood cells by the bone marrow, an increase in the destruction of red blood cells, or if blood is lost due to bleeding. A drop in the red blood cell count, hemoglobin and hematocrit can result in anemia, a condition in which tissues and organs in the body do not get enough oxygen, causing fatigue and weakness. If too many red blood cells are produced, polycythemia results and the blood can become thickened, causing sluggish blood flow and related problems.
Abnormally high or low levels of MCH, as determined by blood testing, can be an indication of a number of problems within the body, ranging from nutrient deficiencies to chronic diseases.
Several diseases and conditions can affect red blood cells and consequently the level of hemoglobin in the blood. In general, the hemoglobin level and hematocrit rise when the number of red blood cells increases. The hemoglobin level and hematocrit fall to less than normal when there is a drop in production of red blood cells by the bone marrow, an increase in the destruction of red blood cells, or if blood is lost due to bleeding. A drop in the red blood cell count, hemoglobin and hematocrit can result in anemia, a condition in which tissues and organs in the body do not get enough oxygen, causing fatigue and weakness. If too many red blood cells are produced, polycythemia (erythrocytosis) results and the blood can become thickened, causing sluggish blood flow and related problems.
The best way to evaluate your MCH result is to look the all parameters of complete blood count (CBC) together and diagnose the anemia correctly. Complete blood count laboratory test is a simple and quick test but with it’s lots of sub parameters provides a lot of information about your general health condition and also diseases.
MCH levels
MCH normal range
Normal range of MCH for adults is 27 to 31 picograms/red blood cell (pg/cell)
This range may change according to the laboratory and the analyzer that is used.
- 1-3 days old baby: 31-37 pg
- 1 week-1 month old baby: 28-40 pg
- 3-6 months baby: 25-35 pg
- 6 months- 2 years old baby: 23-31 pg
- 2-6 years old child: 24-30 pg
- 6-12 years old child: 25-33 pg
- 12-18 years old teen: 25-35 pg
MCH low
MCH levels below 27 pg (hypochromia) are considered abnormally low. Common causes of Low MCH results include blood loss, iron deficiency and microcytic anemia, which is a condition in which red blood cells are abnormally small, carrying less hemoglobin. Other potential causes of a low MCH test include hemoglobinopathy, which is a group of disorders that cause changes in the structure of hemoglobin, and iron-deficiency anemia.
MCH high
MCH levels over 31 pg (hyperchromia) are generally considered abnormally high. The most common reason for high MCH is macrocytic anemia or megaloblastic anemia, which is a blood disorder in which the body fails to produce enough red blood cells. In macrocytic anemia, red blood cells that are produced are larger than usual, each carrying more hemoglobin than normal-sized cells would. This condition can be caused by deficient levels of vitamin B-12 or folic acid in the body; nutrients found in foods like fish, liver, green leafy vegetables and fortified cereals. These contribute to efficient red blood cell production and may be lacking in your diet, you may not be able to absorb them or there may be other reasons your body cannot process them. Other possible causes of megaloblastic anemia, hypothyroidism, liver disease, and primary bone marrow dysplasias (including myelodysplasia and myeloproliferative disorders) are some of the more common causes.
Symptoms of macrocytic anemia can include:
- Unexplained fatigue
- Heart palpitations
- Pallor
- Heart complications. Since this can interfere with heart function, early diagnosis of high MCH (macrocytic) anemia is important.
- Other possible reasons for a high MCH test include several other forms of anemia, thyroid dysfunction, chemotherapy, certain infections, over use of estrogen-containing medications, some forms of leukemia and hereditary spherocytosis; a condition that causes a shortage of red blood cells.
What are red blood cells
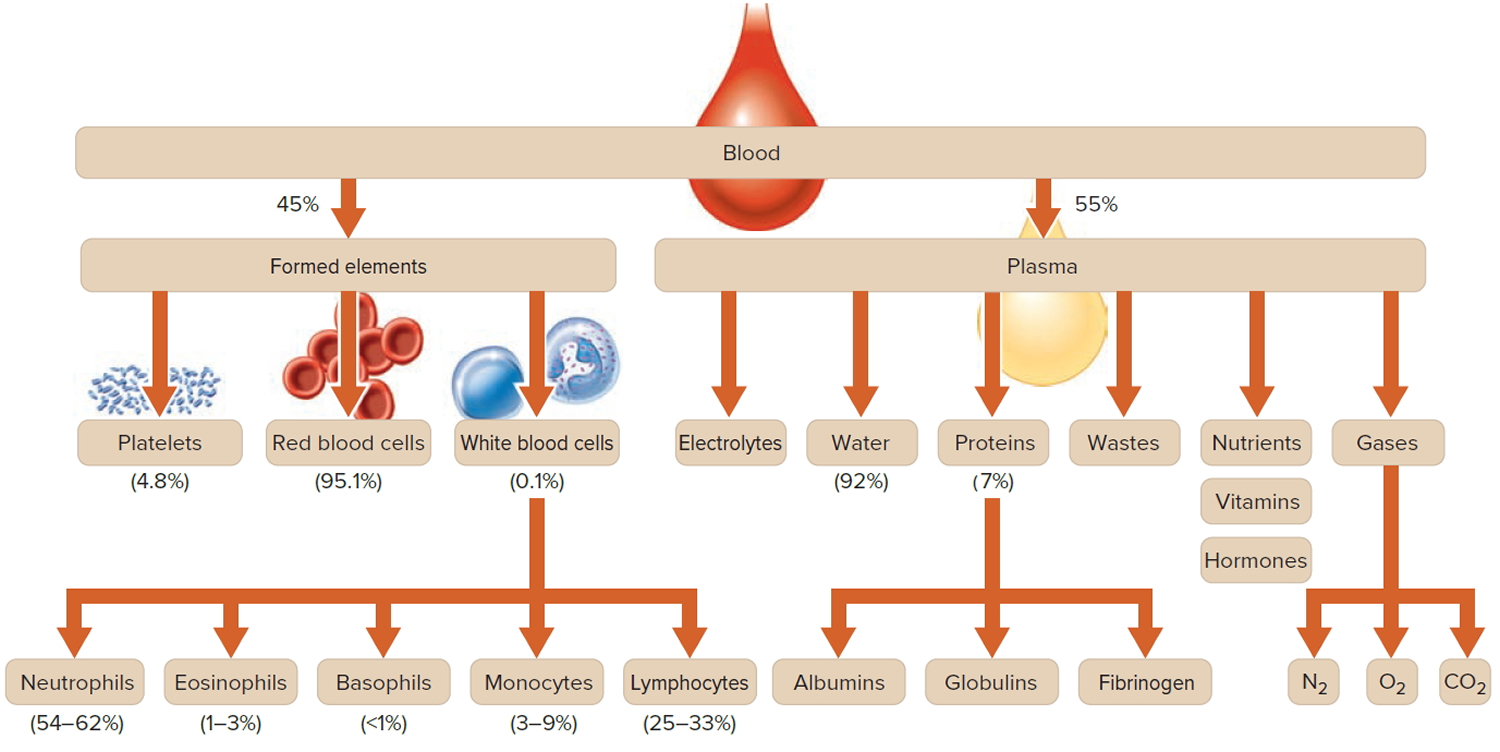
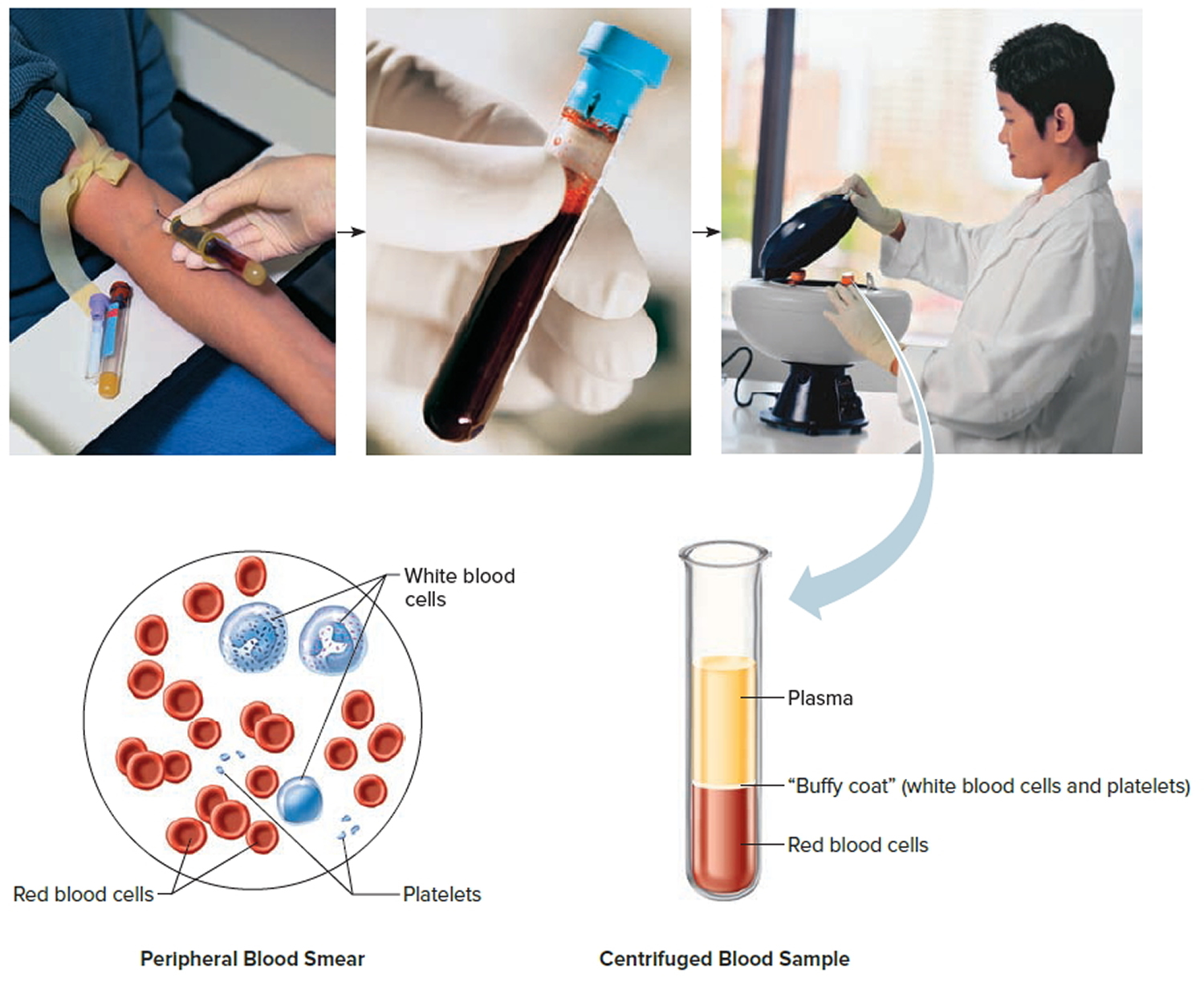
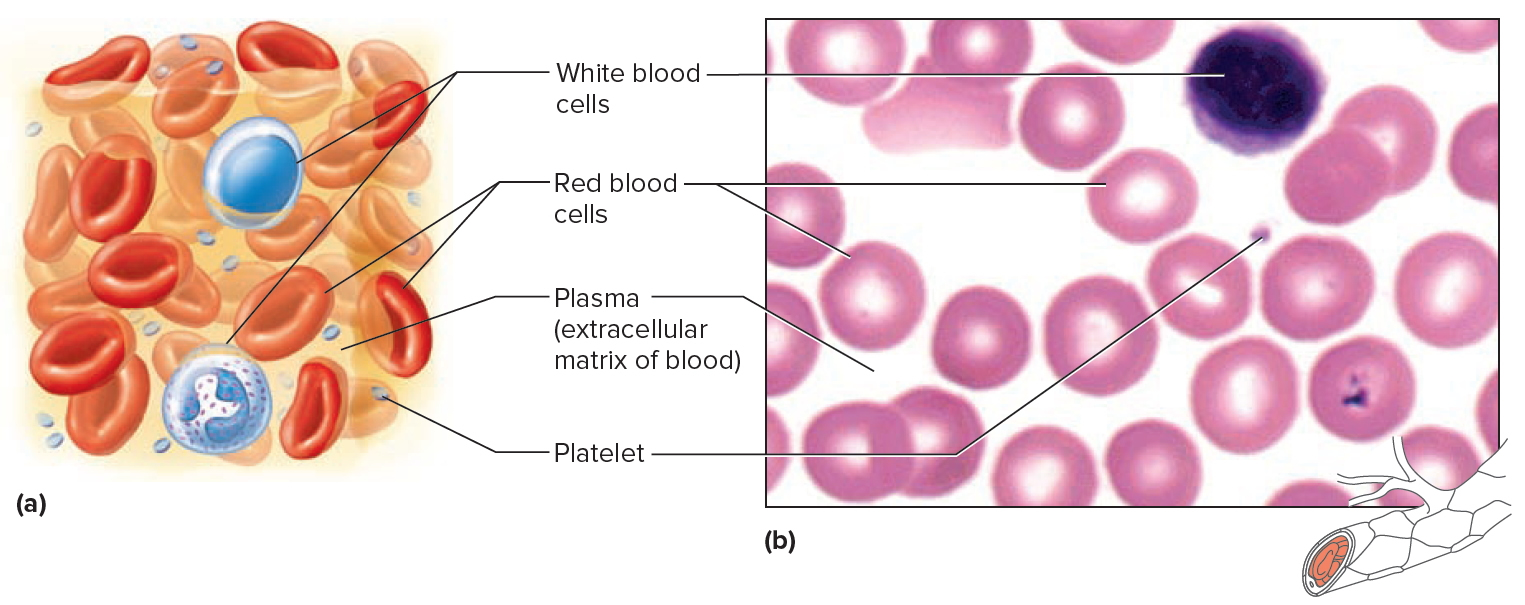
Blood transports a variety of materials between interior body cells and those that exchange substances with the external environment. In this way, blood helps maintain stable internal environmental conditions. Blood is composed of formed elements suspended in a fluid extracellular matrix called blood plasma. The “formed elements” include red blood cells, white blood cells, and cell fragments called platelets. Most blood cells form in red marrow within the hollow parts of certain long bones.
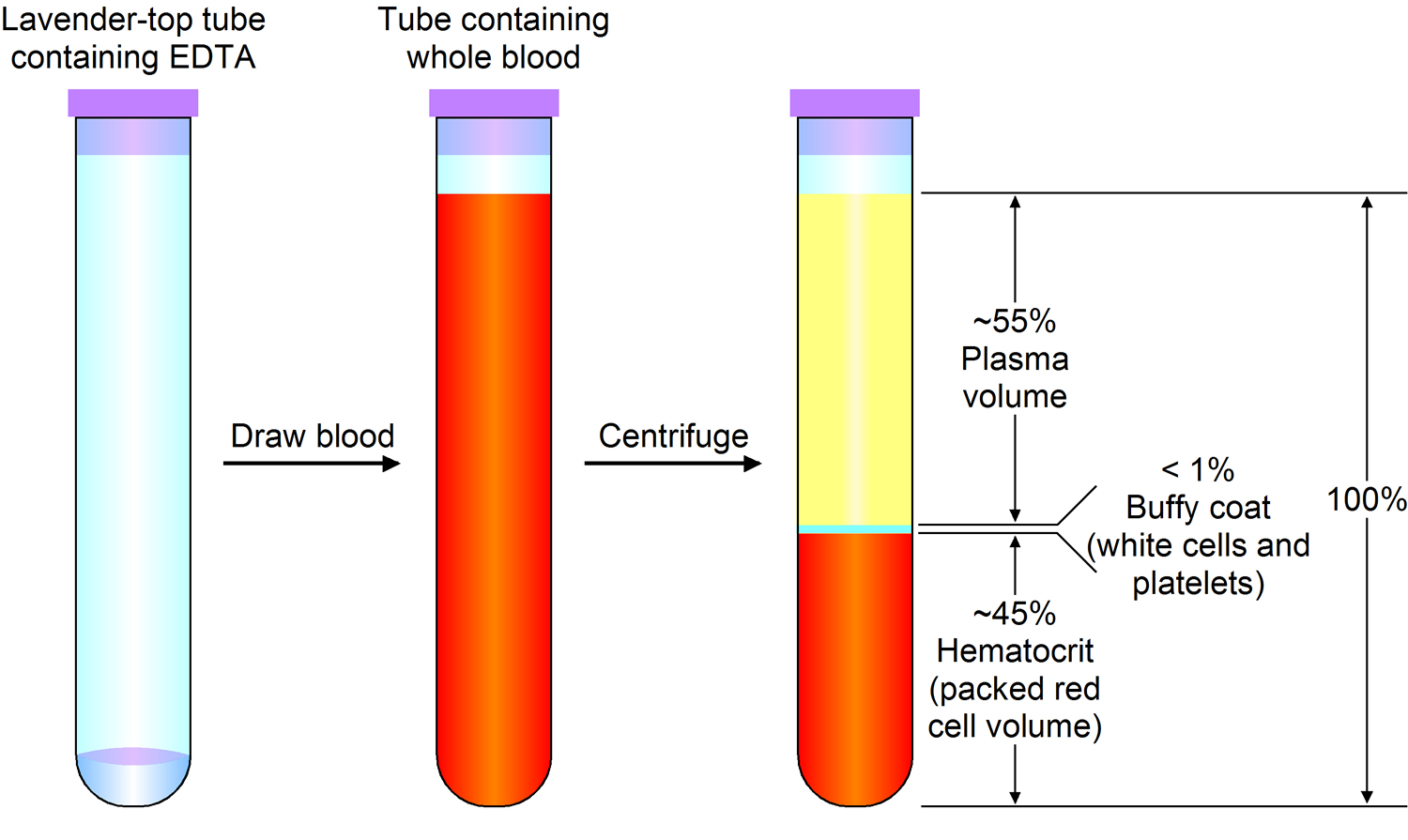
Most blood samples are roughly 37% to 49% red blood cells by volume – adult females is 38–46% (average = 42%) and for adult males, it is 40–54% (average = 47). This percentage is called the hematocrit. The white blood cells and platelets account for less than 1% of blood volume. The remaining blood sample, about 55%, is the plasma, a clear, straw-colored liquid. Blood plasma is a complex mixture of water, gases, amino acids, proteins, carbohydrates, lipids, vitamins, hormones, electrolytes, and cellular wastes (see Figure 1).
Blood volume varies with body size, percent adipose tissue, and changes in fluid and electrolyte concentrations. An average-size adult has a blood volume of about 5 liters (5.3 quarts), 4–5 liters in a female and 5–6 liters in a male.
Red blood cell (also called erythrocyte) is biconcave disc without a nucleus. This biconcave shape is an adaptation for transporting the gases oxygen and carbon dioxide. It increases the surface area through which oxygen and carbon dioxide can diffuse into and out of the cell. The characteristic shape of a red blood cell also places the cell membrane closer to oxygen-carrying hemoglobin molecules in the cell reducing the distance for diffusion.
Each red blood cell is about one-third hemoglobin by volume. This protein imparts the color of blood. When hemoglobin binds oxygen, the resulting oxyhemoglobin is bright red, and when oxygen is released, the resulting deoxyhemoglobin is darker.
Prolonged oxygen deficiency (hypoxia) causes cyanosis, in which the skin and mucous membranes appear bluish due to an abnormally high blood concentration of deoxyhemoglobin in the superficial blood vessels. Exposure to low temperature may also result in cyanosis by constricting superficial blood vessels. This response to environmental change slows skin blood flow. As a result, more oxygen than usual is removed from the blood flowing through the vessels, increasing the concentration of deoxyhemoglobin.
Note: Blood is a complex mixture of formed elements in a liquid extracellular matrix, called blood plasma. Note that water and proteins account for 99% of the blood plasma.
Figure 1. Blood composition
Note: Blood consists of a liquid portion called plasma and a solid portion (the formed elements) that includes red blood cells, white blood cells, and platelets. When blood components are separated by centrifugation, the white blood cells and platelets form a thin layer, called the “buffy coat,” between the plasma and the red blood cells, which accounts for about 1% of the total blood volume. Blood cells and platelets can be seen under a light microscope when a blood sample is smeared onto a glass slide.
Blood Cell Formation
The process of blood cell formation, called hematopoiesis, begins in the yolk sac, which lies outside the human embryo. Later in the fetal development, red blood cells are manufactured (erythropoiesis) in the liver and spleen, and still later they form in bone marrow. After birth, these cells are produced in the red bone marrow.
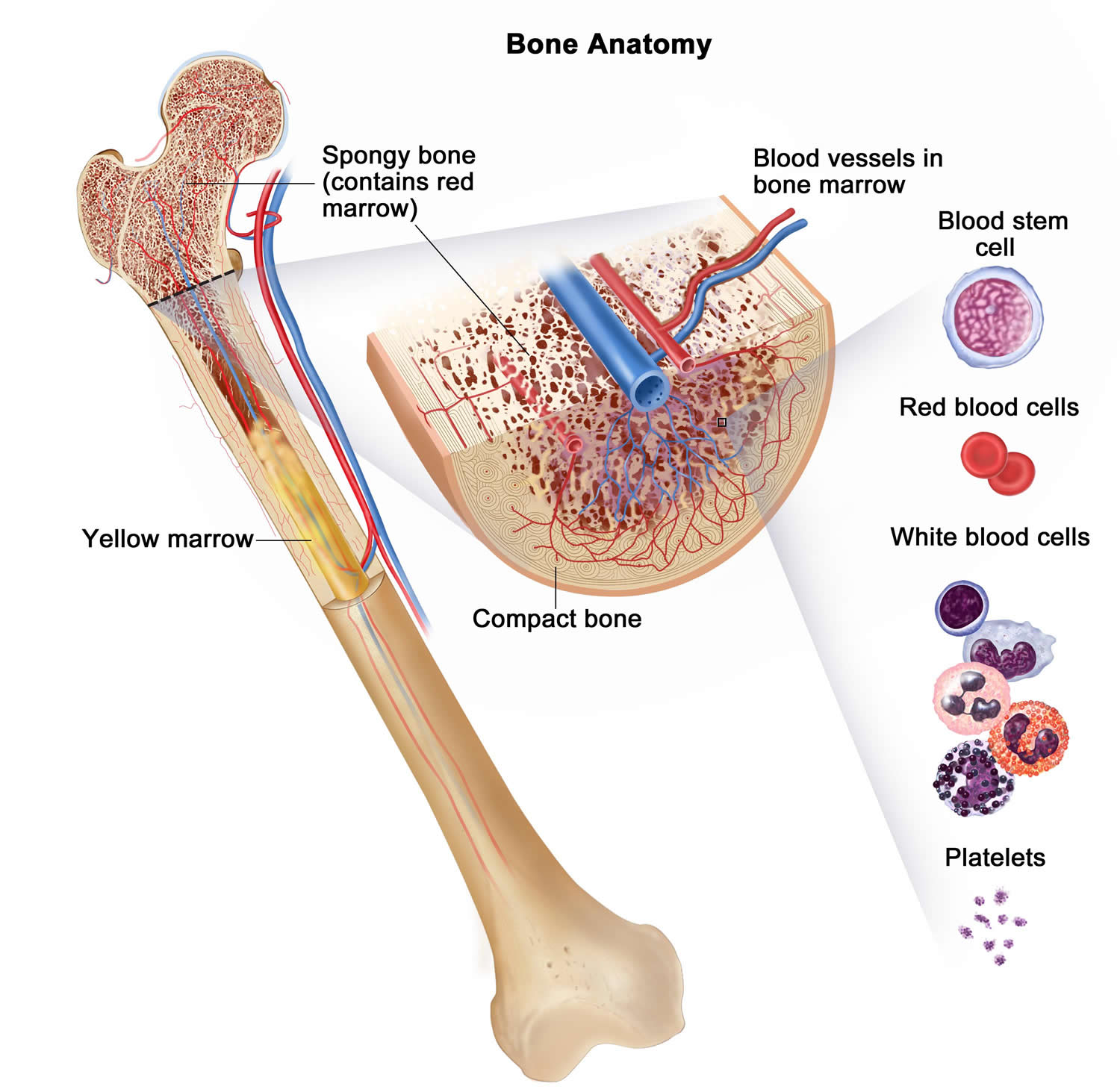
Bone marrow is a soft, netlike mass of connective tissue within the medullary cavities of long bones, in the irregular spaces of spongy bone, and in the larger central canals of compact bone tissue. It is of two kinds: red and yellow. Red bone marrow functions in the formation of red blood cells (erythrocytes), white blood cells (leukocytes), and blood platelets. The color comes from the oxygen-carrying pigment hemoglobin in the red blood cells.
In an infant, red marrow occupies the cavities of most bones. As a person ages, yellow bone marrow, which stores fat, replaces much of the red marrow. Yellow marrow is not active in blood cell production. In an adult, red marrow is primarily found in the spongy bone of the skull, ribs, breastbone (sternum), collarbones (clavicles), backbones (vertebrae), and hip bones. If the supply of blood cells is deficient, some yellow marrow may become red marrow, which then reverts to yellow marrow when the deficiency is corrected.
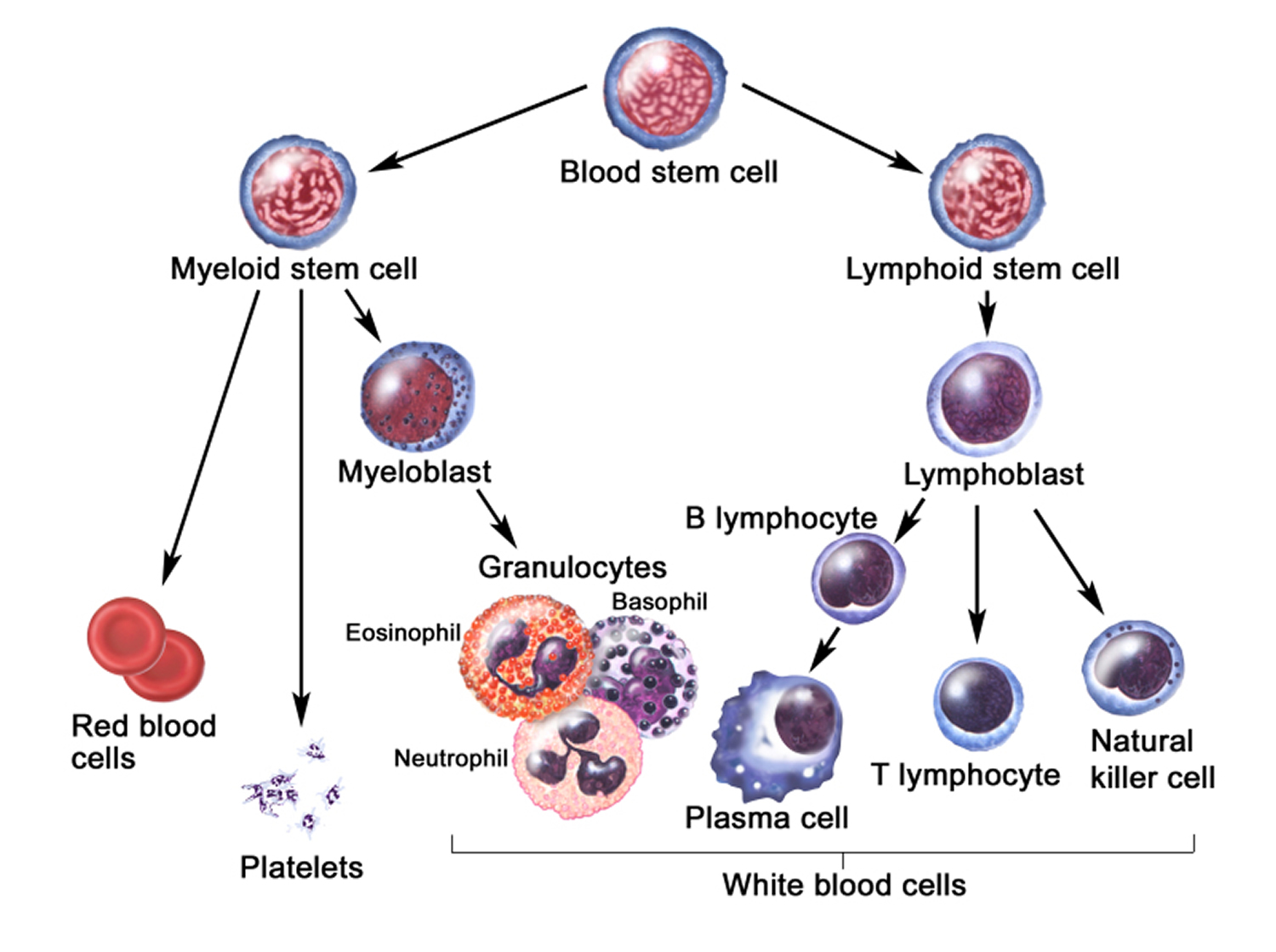
Figure 3 illustrates the stages in the formation of red blood cells from hematopoietic stem cells (blood-forming cells), which are also called hemocytoblasts.
Red blood cells have nuclei during their early stages of development but lose their nuclei as the cells mature. Losing the nuclei provides more space for hemoglobin. Because mature red blood cells do not have nuclei, they cannot divide. They use none of the oxygen they carry because they do not have mitochondria. Mature red blood cells produce ATP through glycolysis only.
The average life span of a red blood cell is 120 days. Many of these cells are removed from the circulation each day, and yet the number of cells in the circulating blood remains relatively stable. This observation suggests a homeostatic control of the rate of red blood cell production.
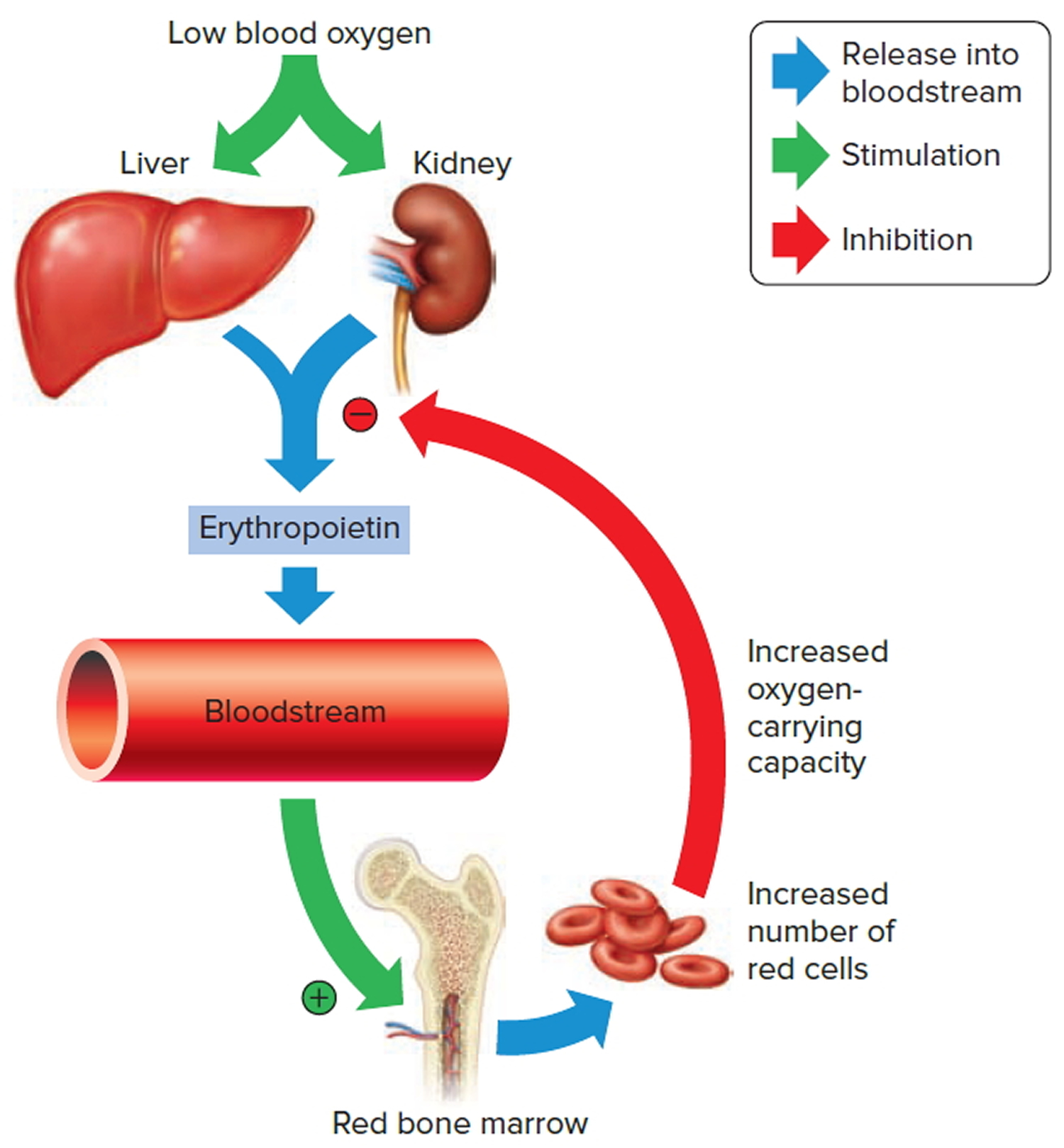
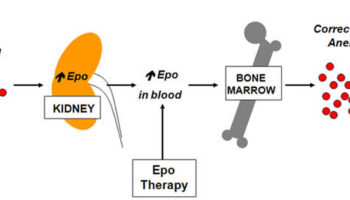
The hormone erythropoietin (EPO) controls the rate of red blood cell formation through negative feedback. The kidneys, and to a lesser extent the liver, release erythropoietin in response to prolonged oxygen deficiency (Figure 6). At high altitudes, for example, where the amount of oxygen in the air is reduced, the blood oxygen level initially decreases. This drop in the blood oxygen level triggers the release of erythropoietin, which travels via the blood to the red bone marrow and stimulates red blood cell production.
After a few days of exposure to high altitudes, many newly formed red blood cells appear in the circulating blood. The increased rate of production continues until the number of erythrocytes in the circulation is sufficient to supply tissues with oxygen. When the availability of oxygen returns to normal, erythropoietin release decreases, and the rate of red blood cell production returns to normal as well. An excessive increase in red blood cells is called polycythemia. This condition increases blood viscosity, slowing blood flow and impairing circulation.
Figure 2. Bone marrow anatomy
Anatomy of the bone. The bone is made up of compact bone, spongy bone, and bone marrow. Compact bone makes up the outer layer of the bone. Spongy bone is found mostly at the ends of bones and contains red marrow. Bone marrow is found in the center of most bones and has many blood vessels. There are two types of bone marrow: red and yellow. Red marrow contains blood stem cells that can become red blood cells, white blood cells, or platelets. Yellow marrow is made mostly of fat.
Dietary Factors Affecting Red Blood Cell Production
Availability of B-complex vitamins—vitamin B12 and folic acid—significantly influences red blood cell production. Because these vitamins are required for DNA synthesis, they are necessary for the growth and division of cells. Cell division is frequent in blood-forming (hematopoietic) tissue, so this tissue is especially vulnerable to a deficiency of either of these vitamins.
Hemoglobin synthesis and normal red blood cell production also require iron. The small intestine absorbs iron slowly from food. The body reuses much of the iron released by the decomposition of hemoglobin from damaged red blood cells. Nonetheless, insufficient dietary iron can reduce hemoglobin synthesis.
A deficiency of red blood cells or a reduction in the amount of hemoglobin they contain results in a condition called anemia. This reduces the oxygen-carrying capacity of the blood, and the affected person may appear pale and lack energy. A pregnant woman may have a normal number of red blood cells, but she develops a relative anemia because her plasma volume increases due to fluid retention. This shows up as a decreased hematocrit.
In contrast to anemia, the inherited disorder called hemochromatosis results in the absorption of iron in the small intestine at ten times the normal rate. Iron builds up in organs, to toxic levels. Treatment is periodic blood removal, as often as every week.
Figure 3. Blood cell development. A blood stem cell goes through several steps to become a red blood cell, platelet, or white blood cell
Figure 4. Blood cells
Note: Blood tissue consists of red blood cells, white blood cells, and platelets suspended in plasma. (a) Idealized representation of a sample of blood. (b) Micrograph of a sample of blood (1,000x).
Figure 5. Red blood cells
What is the function of red blood cells
Red blood cells function is to transports oxygen and carbon dioxide. to body tissues by blood flow via circulatory system. Red blood cells take oxygen from the lungs and release it into the cells or tissues. Lipids and proteins make up the cell membrane of red blood cells. Hemoglobin, an iron containing biomolecule, is the rich component of the cytoplasm of red blood cells mainly responsible for the oxygen binding and red color of the erythrocytes (see Figure 6).
Figure 6. Red blood cell formation
Note: Low blood oxygen causes the kidneys and to a lesser degree, the liver to release erythropoietin. Erythropoietin stimulates target cells in the red bone marrow to increase the production of red blood cells, which carry oxygen to tissues.
Destruction of Red Blood Cells
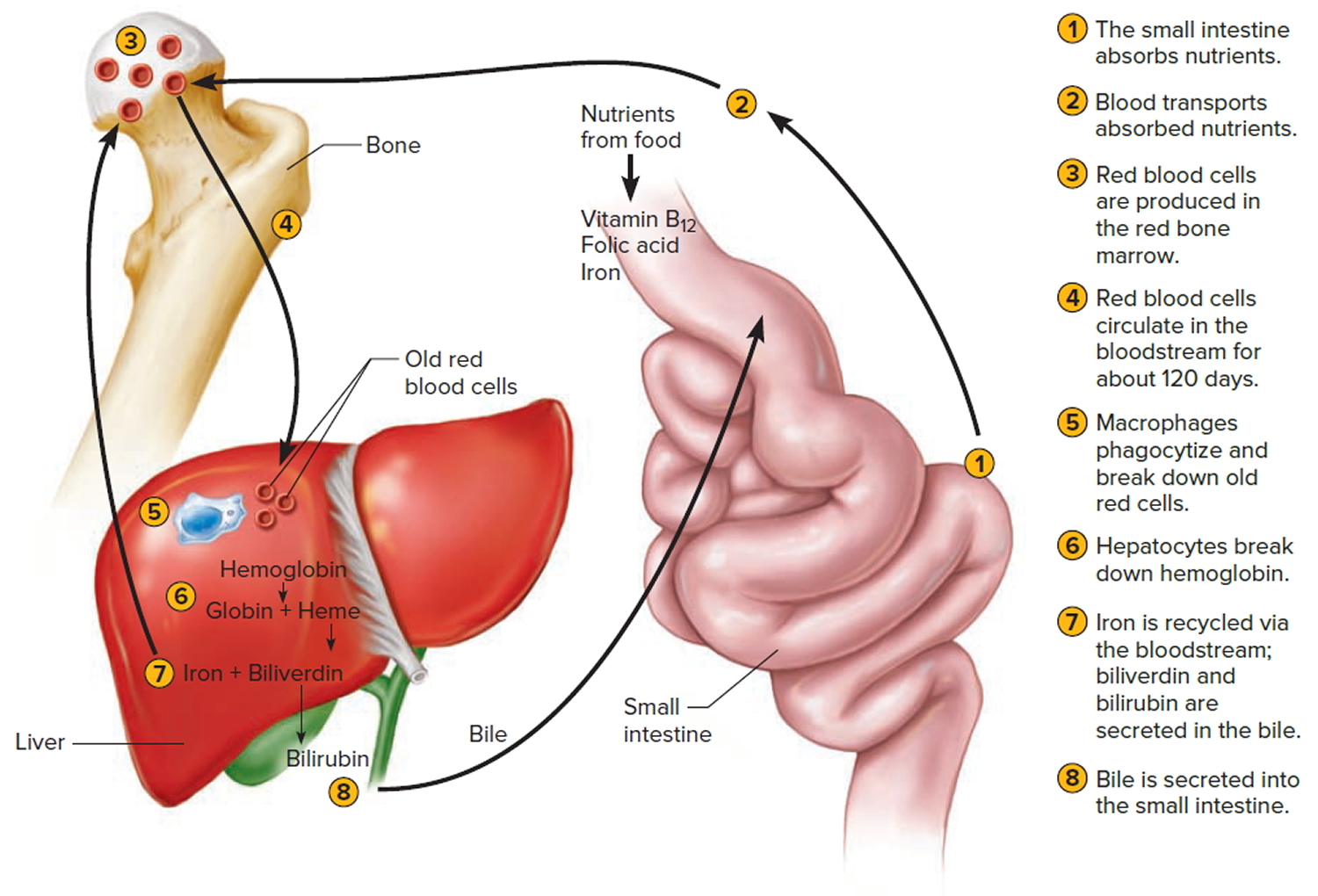
The average life span of red blood cells is about four months (120 days) after which it breaks down. Red blood cells are elastic and flexible, and they readily bend as they pass through small blood vessels. As the cells near the end of their four-month life span, however, they become more fragile. The cells may sustain damage simply passing through capillaries, particularly those in active muscles that must withstand strong forces. Macrophages phagocytize and destroy damaged red blood cells, primarily in the liver and spleen. Macrophages are large, phagocytic, wandering cells. During phagocytosis, the iron from the hemoglobin is retained in the liver and spleen cells and is again used in the formation of red blood cells in the body. About 2-10 million red blood cells are formed and destroyed each second in a normal person.
Hemoglobin molecules liberated from red blood cells break down into their four component polypeptide “globin” chains, each surrounding a heme group. The heme further decomposes into iron and a greenish pigment called biliverdin. The blood may transport the iron, combined with a protein, to the hematopoietic tissue in red bone marrow to be reused in synthesizing new hemoglobin. About 80% of the iron is stored in the liver in the form of an iron-protein complex. Biliverdin eventually is converted to an orange-yellow pigment called bilirubin. Biliverdin and bilirubin are secreted in the bile as bile pigments. Figure 7 summarizes the life cycle of a red blood cell.
In jaundice (yellow discoloration of the skin and the whites of the eyes), accumulation of bilirubin turns the skin and eyes yellowish. Newborns can develop physiologic jaundice a few days after birth. This condition may be the result of immature liver cells that ineffectively secrete bilirubin into the bile. Treatment includes exposure to fluorescent light, which breaks down bilirubin in the tissues, and feedings that promote bowel movements. In hospital nurseries, babies being treated for physiological jaundice lie under “bili lights,” clad only in diapers and protective goggles.
Figure 7. Red blood cell hemoglobin
Figure 8. Lifecycle of a red blood cell
What is anemia?
Anemia is a condition that occurs when the amount of hemoglobin in a person’s blood drops below normal. A decrease in hemoglobin is often associated with a decrease in the number of red blood cells (red blood cells) and hematocrit. Hemoglobin is contained within red blood cells and is necessary to transport and deliver oxygen from the lungs to the rest of the body. Without a sufficient supply of oxygen, many tissues and organs throughout the body can be adversely affected. People with anemia may experience fatigue and weakness and may lack energy.
Anemia is a fairly common condition, affecting both men and women of all ages, races, and ethnic groups. However, certain people have increased risk of developing anemia. These include people with diets poor in iron and vitamins, chronic diseases such as kidney disease, diabetes, cancer, inflammatory bowel disease, a family history of inherited anemia, chronic infections such as tuberculosis or HIV, and those who have had significant blood loss from injury or surgery. Anemia can be mild, moderate, or severe depending on how much the red blood cell count and/or hemoglobin levels are decreased.
In general, the main causes of anemia include:
- Impaired or decreased production of red blood cells by the bone marrow due to nutritional deficiency (e.g., iron deficiency, B vitamin deficiencies), bone marrow failure (e.g., aplastic anemia, myelodysplastic syndrome), or diseases that involve the bone marrow (e.g., infection, lymphoma, solid tumor)
- Loss of red blood cells due to bleeding or to increased destruction of red blood cells as in hemolytic anemia
Anemia may be acute or chronic. Chronic anemia may develop slowly over a period of time with long-term illnesses such as diabetes, chronic kidney disease, or cancer. In these situations, the anemia-related symptoms may not be apparent because the underlying disease masks its symptoms and/or the body adapts to anemia when it develops over a period of time. The presence of anemia in chronic conditions may often go undetected for a period of time and sometimes may only be discovered during tests or examinations for other conditions.
Anemia may also occur in acute episodes such as with substantial blood loss (extensive injury or invasive surgery) or with certain anemias in which a significant number of red blood cells are destroyed known as hemolytic anemia. Signs and symptoms may become apparent very quickly, and the cause can be determined from a combination of physical examination, medical history, and testing.
Types of anemia
Anemias can also be described based on the red blood cell size and concentration of hemoglobin in them. If cell size is much smaller than normal, it is known as microcytic anemia. If it is much bigger than normal (high MCV), then it is macrocytic anemia. Likewise, if the concentration of hemoglobin is much lower than normal, it is hypochromic anemia (low MCH); if the concentration is much higher than normal, the red blood cells are called hyperchromic (high MCH).
Within the two broad categories of general causes of anemia, there are several types with different specific causes. Some of the most common types are summarized in the table below. See the sections below to read more about each one.
| Type of Anemia | Description | Examples of Causes |
| Iron Deficiency | Deficiency of iron leads to decreased production of hemoglobin; low levels of hemoglobin in turn leads to production of smaller and hypochromic red blood cells (low MCV and low MCH). | Blood loss; diet low in iron; poor absorption of iron |
| Pernicious Anemia and B Vitamin Deficiency | Red blood cells do not develop as they normally would because of a lack of B vitamins (B12 and folate); leads to decreased production of red blood cells. | Lack of intrinsic factor (needed for B12 absorption); diet low in B vitamins; decreased absorption of B vitamins |
| Aplastic | Bone marrow is unable to produce enough blood cells; a life-threatening condition. | Cancer therapy, exposure to toxins, autoimmune disease, viral infections |
| Hemolytic | Red blood cells are destroyed faster than the bone marrow can replace them. | Inherited causes such as sickle cell disease and hereditary spherocytosis; other causes include transfusion of incompatible blood, autoimmune disease, certain drugs (penicillin) |
| Anemia of Chronic Diseases | Various conditions over the long term can cause decreased production of red blood cells. | Kidney disease, rheumatoid arthritis, diabetes, tuberculosis, HIV, Crohn disease, cancer, and others |
There are many other conditions that can, for various reasons, result in some level of anemia, such as:
Bleeding—significant bleeding resulting from, for example, trauma or surgery (acute) or from gastrointestinal bleeding (ulcers) occurring over time (chronic)
Leukemia (acute or chronic)
Lymphoma
Myelodysplastic syndrome
Multiple myeloma
Myeloproliferative neoplasms (e.g., myelofibrosis)
Infections (e.g., HIV)
Anemia signs and symptoms
Though different types of anemia have different causes, the signs and symptoms can be very similar. Mild or moderate forms of anemia may cause few, if any, symptoms. Some of the most common symptoms are:
- General feeling of tiredness or weakness (fatigue)
- Lack of energy
- Pale skin (pallor)
- Dizziness
- Headaches
Other signs and symptoms that may develop as the anemia becomes more severe include a feeling of cold or numbness in hands and/or feet, shortness of breath, fast or irregular heartbeat, and chest pain.
Tests for anemia
Several routine laboratory tests may be used to help diagnose anemia as well as help to determine the underlying cause. These are listed below. Depending on the results of these, the medical history of the person, and signs and symptoms, other tests may be done as follow up to help diagnose the cause of anemia and to help guide treatment. (Click on the links for the different types of anemia at the top of this page to read about these specific tests.)
Complete Blood Count (CBC)
Anemia may first be detected when a routine test that counts the number and relative proportion of each of the different types of cells in the blood stream, called a complete blood count (CBC), is done during a health exam or as part of testing for other conditions. A complete blood count (CBC) is often ordered as part of a yearly physical exam and helps to evaluate overall health and to screen for a wide variety of disorders.
With anemia, some of the components of the complete blood count (CBC) that may show abnormal results include:
- Red blood cell count—typically low
- Hemoglobin—low
- Hematocrit—low
- Red blood cell indices—these include mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH), and mean corpuscular hemoglobin concentration (MCHC). They give a healthcare practitioner information about the size of the red blood cells and the amount and concentration of hemoglobin in red blood cells present in someone’s blood at that moment. For example, the size and hemoglobin concentration of red blood cells can help with diagnosing anemia because those characteristics can vary for different kinds of anemia.
Blood Smear and Differential
If results of the CBC indicate anemia, it may be followed up with an examination of a blood smear or a differential, which counts white blood cells. The smear review can provide additional information, such as the shape of red blood cells and the presence of abnormal cells, which can help diagnose and classify anemia.
Reticulocyte Count
This test provides information on the number of relatively immature red blood cells in a person’s blood sample. When someone has anemia (low red blood cell count, hemoglobin, and hematocrit), the results of this test can help determine the cause and/or help classify the type of anemia. For example, for a person with anemia, an inappropriately low reticulocyte count often indicates decrease in red blood cell production in the bone marrow.
Results from these tests may give clues as to the cause. Several other tests may be run to help determine the cause of the anemia and to guide treatment. See the individual discussions of the different types of anemia for more on these.
High MCV and MCH
Macrocytosis, defined as a MCV (mean corpuscular volume) greater than 100 fL (femtoliter or 10−15 liter), occurs in approximately 3 percent of the general population 1. Debate persists about the upper limit of normal values of MCV (mean corpuscular volume). In a recent study, 7 percent of patients had a mean corpuscular volume greater than 96 fL, and 1.7 percent had a mean corpuscular volume greater than 100 fL 2. With the advent of automated complete blood cell (CBC) counters and the increased use of certain medications, elevations in the MCV (mean corpuscular volume) may be more commonly encountered.
Causes of high MCV and their prevalence in different populations are shown in Table 1 3. Although high MCV is associated with anemia, hypothyroidism is a more common cause in older persons than in other age groups. Results of a study in New York City indicated that medications for treating human immunodeficiency virus (HIV) infection have become a more prominent cause of high MCV (macrocytosis). Alcoholism is the cause in as many as 80 percent of patients in some populations 4. Bone marrow biopsy was only performed in these studies when another cause could not be determined, and resulted in diagnosis in approximately one fourth of these remaining patients 1. In recent years, an association has been found between Helicobacter pylori infection and vitamin B12 deficiency 5.
Table 1. Major causes of macrocytosis
| Etiology | Study population | |||
|---|---|---|---|---|
| Hospitalized patients in New York City (%) | Outpatients in Finland (%) | Finnish persons older than 75 years (%) | Finnish and American patients (%) | |
Alcohol | 26 | 65 | 15 | 36 |
B12 and/or folate deficiency | 6 | 9 | 28 | 21 |
Medications | 37* | 3 | 2 | 11 |
Hypothyroidism | — | 1 | 12 | 5 |
Bone marrow dysplasias | 6 | 1 | 5 | 5 |
Liver disease (nonalcoholic) | 6 | — | 2 | 6 |
Reticulocytosis | 8 | — | — | 7 |
Miscellaneous | 3 | 21 | 13 | 7 |
Not established | 7 | — | 22 | 12 |
Footnote: Causes listed from most to least common.
*— 13 percent from zidovudine (Retrovir).
The causes of high MCV (macrocytosis) can be broadly classified as megaloblastic and nonmegaloblastic (Table 2). Megaloblastic processes are characterized on the peripheral smear by macroovalocytes and hypersegmented neutrophils, which are absent in nonmegaloblastic macrocytic processes (Figure 9). Nonmegaloblastic processes have round macrocytes or macroreticulocytes (Figure 10). Because the mechanisms producing macrocytosis are not completely understood, the separation between megaloblastic and nonmegaloblastic causes is somewhat artificial. However, this concept remains useful for identifying the most predominant etiology for high MCV (macrocytosis). For example, whereas the effect of alcohol is thought to be primarily a nonmegaloblastic process, in chronic alcoholism there may be concomitant vitamin B12 or folate deficiency 6.
Figure 9. Megaloblastic anemia
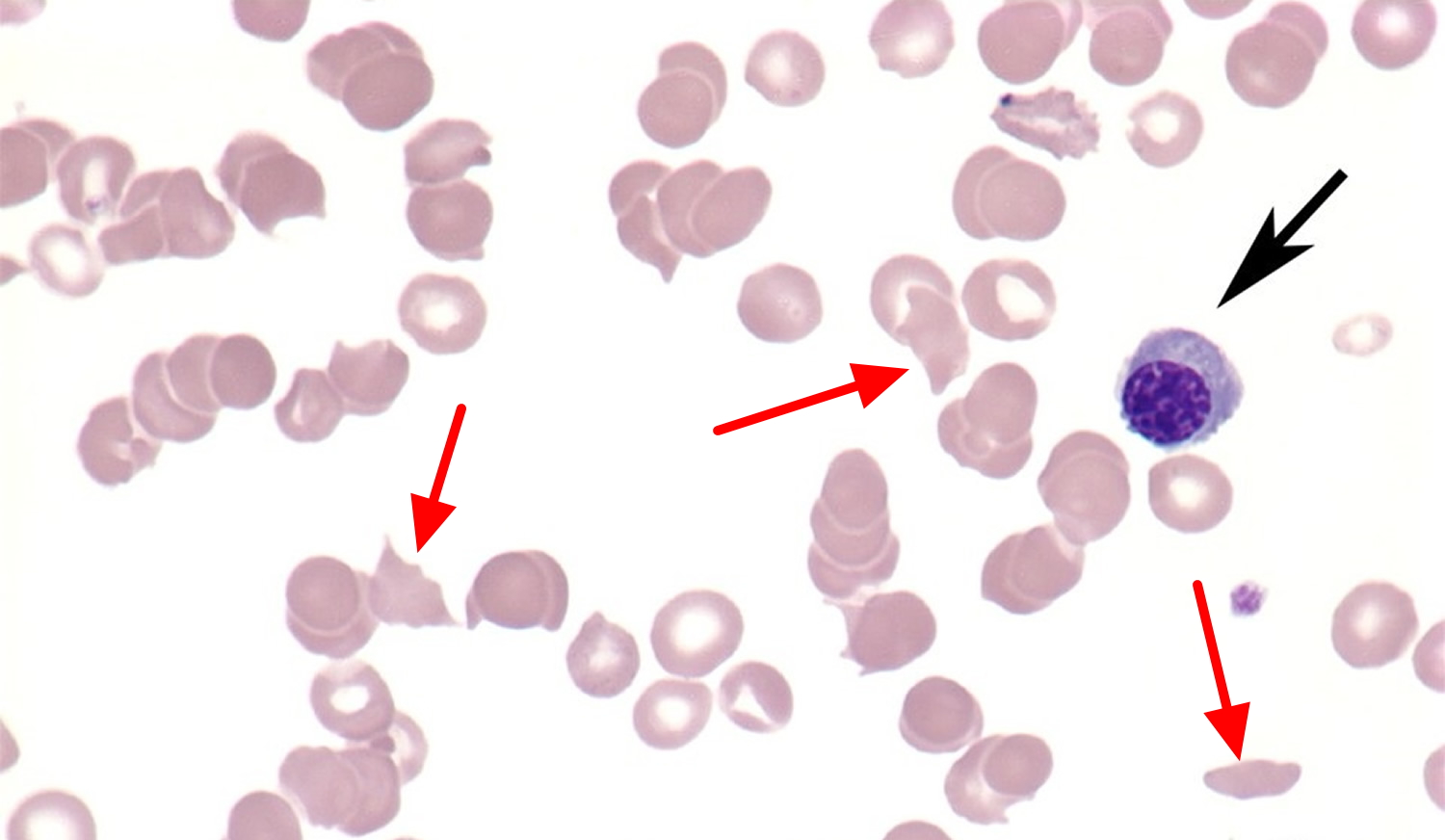
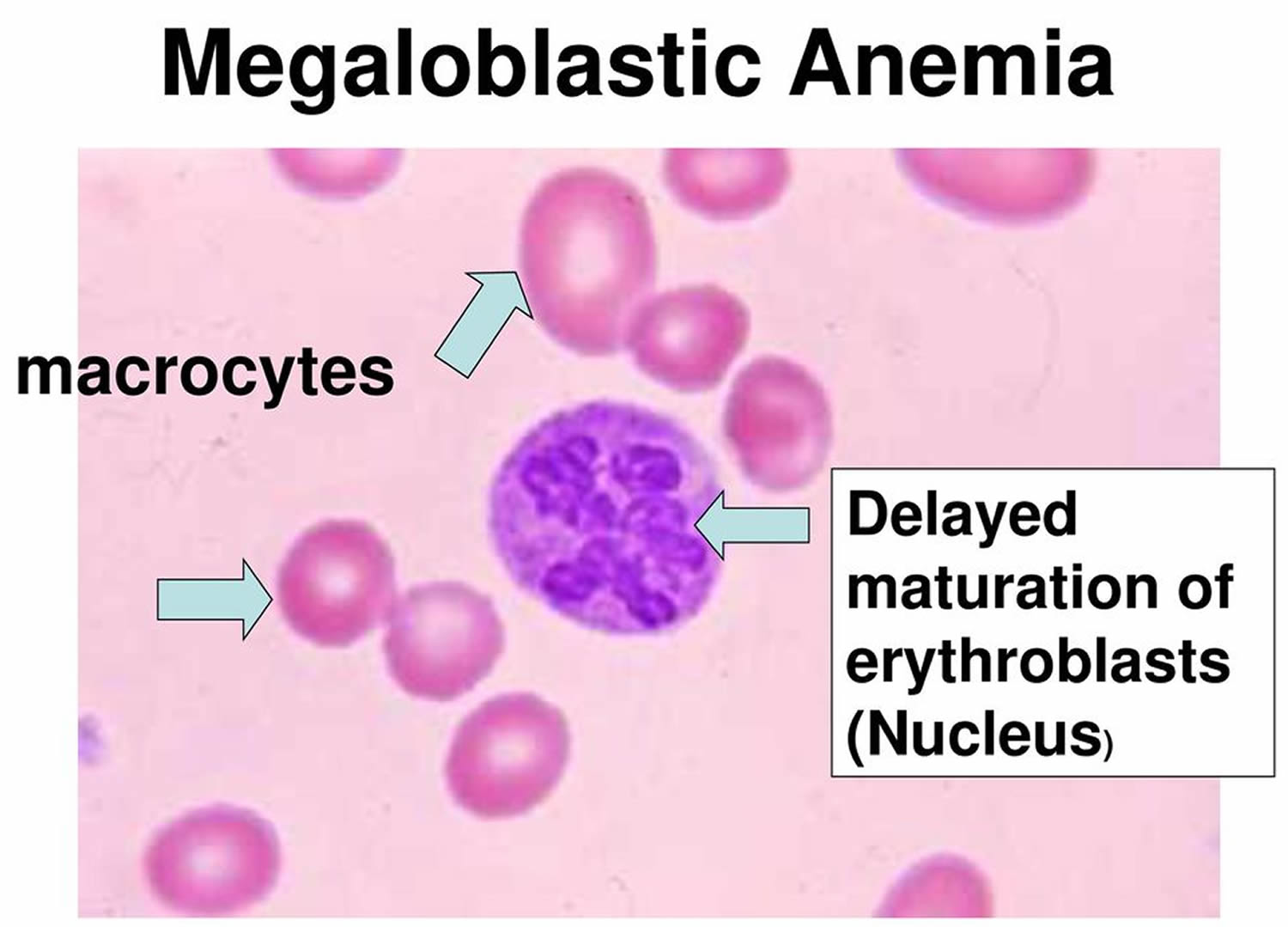 Figure 10. Microangiopathic hemolytic anemia (nonmegaloblastic), with polychromatophilic macrocytes (black arrow) and schistocytes (red arrows) typical of this particular disorder are also present. Schistocytes (red arrows) have an irregular shape and lack the area of central pallor usually seen in red blood cells (erythrocytes).
Figure 10. Microangiopathic hemolytic anemia (nonmegaloblastic), with polychromatophilic macrocytes (black arrow) and schistocytes (red arrows) typical of this particular disorder are also present. Schistocytes (red arrows) have an irregular shape and lack the area of central pallor usually seen in red blood cells (erythrocytes). Table 2. Differential diagnosis of high MCV (macrocytosis)
Megaloblastic (involving vitamin B12 and/or folate deficiencies) |
Atrophic gastritis |
Enteral malabsorption |
Human immunodeficiency virus treatments |
Anticonvulsants (some cause folate depletion) |
Primary bone marrow disorders |
Nitrous oxide abuse |
Inherited disorders |
Nonmegaloblastic |
Alcohol abuse |
Medication side effects (see Table 3) |
Myelodysplasia |
Hypothyroidism |
Liver disease |
Hemolysis |
Hemorrhage |
Chronic obstructive pulmonary disease |
Splenectomy |
False elevations |
Cold agglutinins |
Hyperglycemia |
Marked leukocytosis |
Footnote: Diagnoses listed from most to least common.
In megaloblastic processes, erythrogenic precursors are larger than mature red blood cells (red blood cells) because folate and vitamin B12 deficiencies result in defective RNA and DNA syntheses. Serum elevations in homocysteine and methylmalonic acid result from defective biochemical processes in folate and B12 deficiencies, and could be used to clarify the cause of megaloblastic anemia, although this is not yet standard clinical practice 7.
Nonmegaloblastic processes develop from multiple mechanisms and have not been fully outlined. Macrocytosis can occur when there is increased red blood cell production secondary to peripheral blood cell destruction (i.e., hemolysis) or loss (i.e., hemorrhage), leading to a reticulocytosis. Reticulocytes are incompletely processed red blood cells and, therefore, are slightly larger than the average red blood cell.
Table 3. Medications that may cause high MCV (macrocytosis)
Treatments for human immunodeficiency virus: reverse transcriptase inhibitors (e.g., stavudine [Zerit], lamivudine [Epivir], zidovudine [Retrovir]) |
Anticonvulsants (e.g., valproic acid [Depakote], phenytoin [Dilantin]) |
Folate antagonists (e.g., methotrexate) |
Chemotherapeutics (e.g., alkylating agents, pyrimidine, purine inhibitors) |
Trimethoprim/sulfamethoxazole (Bactrim, Septra) |
Biguanides (e.g., metformin [Glucophage]), cholestyramine (Questran) |
High MCV (Macrocytosis) Specific Causes
Vitamin B12 deficiency
Vitamin B12 is absorbed by the ileum when it is bound by intrinsic factor, which is produced by the parietal cells of the gastric mucosa. In pernicious anemia, the loss of parietal cells leads to insufficient absorption of vitamin B12, which then leads to vitamin B12 deficiency over time. Pernicious anemia is most commonly caused by auto-immune atrophic gastritis, in which autoantibodies are directed against parietal cells and intrinsic factor. Less commonly, pernicious anemia can be caused by nonautoimmune gastritis secondary to Helicobacter pylori (H. pylori) infections and Zollinger-Ellison syndrome.
Patients with vitamin B12 deficiency may describe paresthesias related to peripheral neuropathy, poor or strict vegan diet, lack of socioeconomic resources, bowel-related symptoms (including diarrhea), or a history of bowel surgery for weight loss. Findings on physical examination may include neurologic signs such as ataxia, decreased proprioception, and vibratory sensation. Patients may also have poor dentition or nonspecific oral stomatitis or glossitis.
Because pregnant women take folic acid routinely in prenatal vitamins, macrocytic anemia is much less common during pregnancy. Consider nitrous oxide abuse in at-risk populations, because nitrous oxide inactivates vitamin B12 through oxidation 8. Other uncommon causes include Diphyllobothrium latum (i.e., fish tapeworm) infection or inherited disorders of cobalamin metabolism, including Imerslund syndrome (a congenital vitamin B12 malabsorption associated with proteinuria) 9. Only 10 percent of persons with vitamin B12 deficiency are actually anemic 10.
The normal range for serum measures of vitamin B12 varies among laboratories. If the vitamin B12 level s borderline low (i.e., 100 to 400 pg per mL [74 to 295 pmol per L]), methylmalonic acid and homocysteine levels should be ordered and, if elevated, may provide evidence of B12 deficiency 7. The Schilling test (i.e., measuring enteral absorption of vitamin B12) is not widely available at this time.
Oral therapy appears to be as effective as intramuscular therapy for the treatment of vitamin B12 deficiency. Relapse of pernicious anemia occurs at a mean interval of 65 months after cessation of treatment. It is important for patients to adhere to long-term therapy because the deficiency will recur if treatment is stopped, unless a reversible cause is identified 11.
Folate deficiency
The history of folate deficiency may mimic the history of vitamin B12 deficiency in regard to poor nutritional intake or absorption. In addition, 35 percent of patients with alcoholism and macrocytic anemia are folate deficient, which can be caused by poor nutritional intake, malabsorption, hepatobiliary dysfunction, and possibly increased folate catabolism 12. Some medications that are used to treat seizure disorders, cancer, and autoimmune diseases can lead to folate deficiency. For example, methotrexate directly inhibits dihydrofolate reductase, which leads to a functional folate deficiency 13. Other medications that affect folate metabolism include 5-fluorouracil (Adrucil), hydroxyurea (Hydrea), pyrimethamine (Daraprim), trimethoprim/sulfamethoxazole (Bactrim, Septra), pentamidine (Pentam), and phenytoin (Dilantin) 14. Medications can also affect folate absorption, including metformin (Glucophage) and cholestyramine (Questran). Supplementing with folate may be necessary when treating a patient with such medications 15.
Serum folate levels are not useful because they fluctuate rapidly with dietary intake and are not cost effective 16. Red blood cell folate levels more accurately correlate with folate stores and should be performed if folate deficiency is suspected 17. In differentiating the cause of megaloblastic anemia, a methylmalonic acid level that is within normal range also points toward a diagnosis of folate deficiency, especially if the serum vitamin B12 level is within the normal range. Note that homocysteine levels will be elevated with vitamin B12 and folate deficiencies.
HIV medications
Treatment of HIV with reverse transcriptase inhibitors (e.g., stavudine [Zerit], lamivudine [Epivir], zidovudine [Retrovir]) will cause macrocytosis because they interfere with DNA production, which may lead to megaloblastic changes. Most patients with HIV who are being treated with reverse transcriptase inhibitors will display macrocytosis without anemia. This indicates medication compliance by the patient, and no treatment is necessary 18. Table 3 lists other medications that may cause macrocytosis 19.
Reticulocytosis (hemolysis or hemorrhage)
For the patient’s history, questions that are pertinent to evidence of blood loss, whether chronic or acute, can point to reticulocytosis. As part of the family history, physicians should ask about the presence of certain hematologic syndromes, including sickle cell disease, hereditary spherocytosis, and glucose-6-phosphate dehydrogenase deficiency (G6PD). In persons who do distance running, hemolysis from constant foot pounding has infrequently been shown to cause macrocytosis 20. The physical examination may reveal hepatosplenomegaly in hemolysis or other physical manifestations of blood loss (e.g., conjunctiva, mucosal pallor). In further diagnostic testing, the peripheral smear will reveal evidence of hemolyzed red blood cells (e.g., bite cells, helmet cells). Macrocytosis results from the marrow’s response to increased cell destruction or blood loss, with release of reticulocytes into the peripheral circulation. Treatment should be focused on the underlying cause of hemolysis or hemorrhage.
Alcohol abuse
The Michigan Alcoholism Screening test and obtaining γ-glutamyltransferase levels were found to be the two most sensitive tests for detecting alcohol abuse in patients with macrocytosis 4. Physical findings consistent with alcoholism include gynecomastia, caput medusae, and jaundice. Alcohol use more commonly causes macrocytosis through its toxic effect than through folate deficiency secondary to alcoholism. The mean corpuscular volume is generally less than 110 fL with chronic alcohol use. Abstinence from alcohol rapidly corrects the elevated mean corpuscular volume 6.
Bone marrow dysfunction
As noted above, myeloproliferative disorders (sometimes called refractory anemia) are a more common cause of macrocytosis and anemia among older persons than in younger populations. Although the peripheral smear may be suggestive, bone marrow biopsy is required to establish this diagnosis. Referral will likely be necessary for the work-up and management.
Other causes
Medications, hypothyroidism, liver and renal disease, and chronic obstructive pulmonary disease are associated with less dramatic elevations in the mean corpuscular volume. Nonalcoholic liver disease and hypothyroidism account for a substantial portion of macrocytosis. Patient history and physical examination should include a history of these medical diseases. Splenectomy may cause macrocytosis because cells are not processed as thoroughly when the spleen is absent. Down syndrome also may be associated with reticulocytosis or myeloproliferative disorders 21. Treatment, if needed, can be directed at the underlying disorder.
High MCV diagnosis
Once macrocytosis is identified, the history and physical examination help narrow the differential diagnosis. The presence of anemia, the degree of elevation of the mean corpuscular volume, and the patient’s overall health guide how aggressively the work-up progresses. At least some amount of investigation is warranted if the diagnosis is not readily apparent or if the patient is anemic (defined by the World Health Organization as a hemoglobin level less than 13 g per dL [130 g per L] in men and less than 12 g per dL [120 g per L] in women) 22.
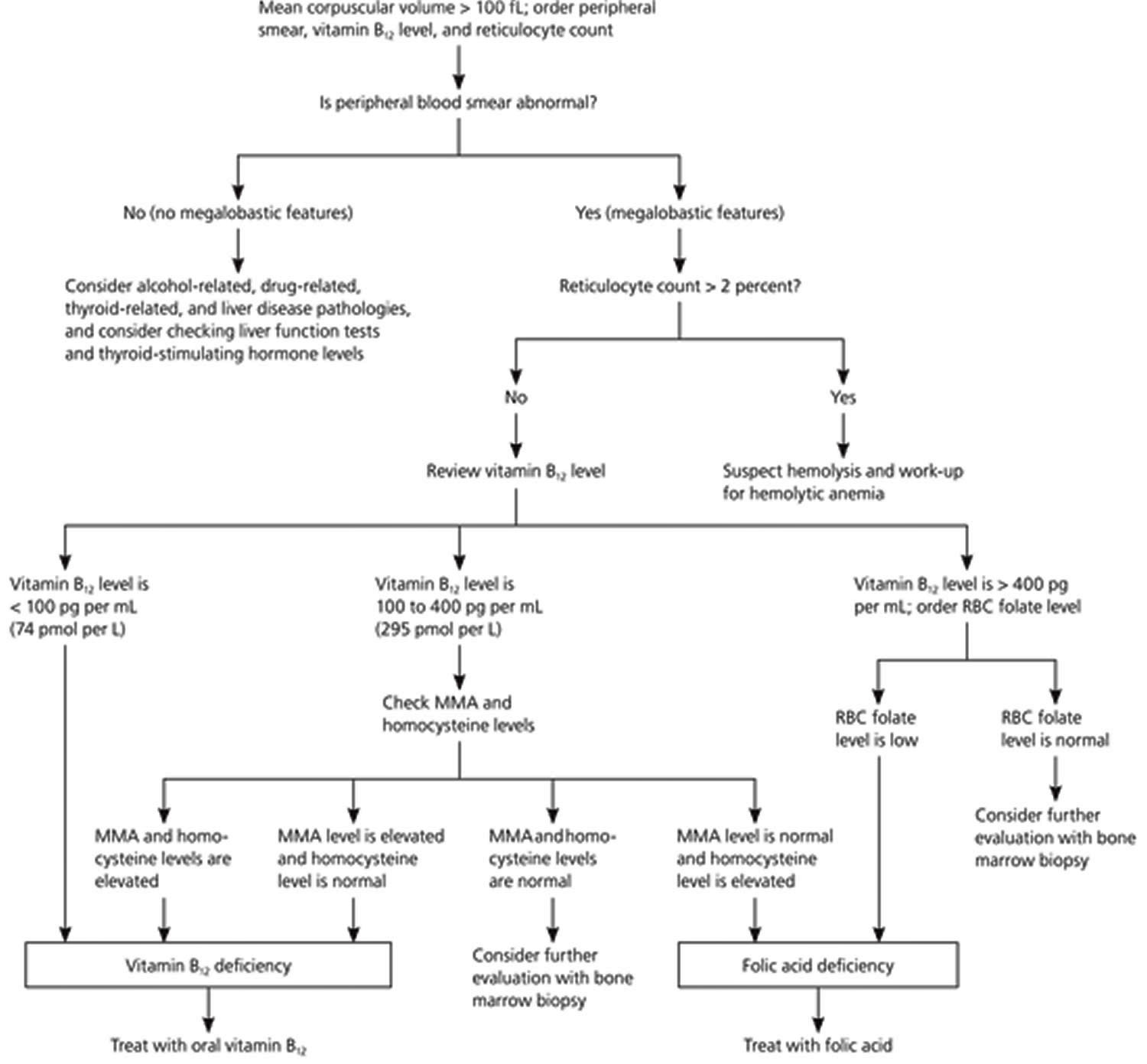
The algorithm outlined in Figure 11 suggests a work-up for macrocytosis. Physicians should begin by ordering a peripheral smear, a reticulocyte count, and a vitamin B12 serum level for all patients with macrocytosis. It may be necessary to specifically order a reticulocyte index in some laboratories, which assesses if there is an adequate bone marrow response. Hemorrhage or hemolysis is the most likely cause if the reticulocyte count is elevated, but anemia recovery also causes an elevation in the reticulocyte count. Measures of vitamin B12 are a useful part of the initial work-up, because if vitamin B12 deficiency is present but undiagnosed, folate repletion will correct the megaloblastic anemia, but not the possible neuropathic changes that occur with B12 deficiency 10.
Although uncommon, consider the possibility of spurious macrocytosis. This may be caused by cold agglutinins, hyperglycemia, or leukocytosis. Cold agglutinins cause the RBCs to clump, making them appear larger to the automatic counter 23. Hyperglycemic blood is more concentrated, and when it is diluted to measure the mean corpuscular volume, the cells swell more than usual, causing a false elevation 24. Increased turbidity of a sample with marked leukocytosis also can cause the machine to overestimate the cell size 25.
When the history and physical examination, peripheral smear, B12 level, and reticulocyte count have not lead to an obvious diagnosis, consider a comprehensive metabolic panel to look for liver and kidney disease, thyroid-stimulating hormone for thyroid disorders, and methylmalonic acid and homocysteine levels to assess for vitamin B12 deficiency, despite a normal vitamin B12 level. If the cause remains elusive, consider again whether the degree of anemia or the patient’s overall health warrants referral to a hematologist for bone marrow biopsy, or search for rarer causes, keeping in mind that the most extensive work-up will result in a diagnosis in approximately 90 percent of patients.
Figure 11. Evaluation of Macrocytic Anemia – RBC = red blood cell; MMA = methylmalonic acid
Low MCV and MCH
If MCH is found lower than 27 pg this means red blood cells hemoglobin is decreased too. Blood loss and iron deficiency are common causes of low MCH levels and this is called microcytic anemia (low MCV anemia). Red blood cell size is smaller than normal in microcytic anemia so red blood cells can’t carry enough hemoglobin and oxygen to the tissues.
Microcytosis (low MCV) is usually encountered incidentally when a complete blood count (CBC) is performed for various reasons. The condition is defined as a mean corpuscular volume of less than 80 μm³ (80 fL) in adults and is often associated with anemia 26. Normal mean corpuscular volume and hemoglobin levels vary during childhood, and normal hemoglobin levels can vary based on factors such as ethnicity, tobacco use, and altitude (Table 4) 27.
Table 4. Variations in Hemoglobin Level and Mean Red Blood Cell Volume (MCV)
| Age | Hemoglobin level in g per dL (g per L) | Mean corpuscular volume in μm3 (fL) | ||
|---|---|---|---|---|
| Mean* | Diagnostic of anemia | Mean | Diagnostic of microcytosis | |
3 to 6 months | 11.5 (115) | 9.5 (95) | 91 (91) | 74 (74) |
6 months to 2 years | 12.0 (120) | 10.5 (105) | 78 (78) | 70 (70) |
2 to 6 years | 12.5 (125) | 11.5 (115) | 81 (81) | 75 (75) |
6 to 12 years | 13.5 (135) | 11.5 (115) | 86 (86) | 77 (77) |
12 to 18 years (female) | 14.0 (140) | 12.0 (120) | 90 (90) | 78 (78) |
12 to 18 years (male) | 14.5 (145) | 13.0 (130) | 88 (88) | 78 |
20 to 59 years (white men) | — | 13.7 (137) | 90 | 80 (80) |
60 years and older (white men) | — | 13.2 (132) | 90 | 80 |
20 to 59 years (white women) | — | 12.2 (122) | 90 | 80 |
60 years and older (white women) | — | 12.2 (122) | 90 | 80 |
20 to 59 years (black men) | — | 12.9 (129) | 90 | 80 |
60 years and older (black men) | — | 12.7 (127) | 90 | 80 |
20 to 59 years (black women) | — | 11.5 (115) | 90 | 80 |
60 years and older (black women) | — | 11.5 (115) | 90 | 80 |
—No data available for some age groups.
[Source 28]Low MCV causes
In the United States, the most common cause of microcytosis is iron deficiency anemia. Other causes include anemia of chronic disease, lead toxicity, sideroblastic anemia, and thalassemia trait 29. Table 5 presents the differential diagnosis of microcytosis 30.
Table 5. Differential Diagnosis of Low MCV (Microcytosis)
Children and adolescents
Menstruating women
Men and nonmenstruating women
|
Footnote: Listed in descending order of frequency
[Source 28]Low MCV Diagnosis
History and Physical Examination
Once microcytosis is diagnosed, the history can sometimes provide clues to the underlying etiology. Important history information includes nutritional intake (especially whole milk intake in children) 27; pica or cravings for ice, which are symptoms of iron deficiency anemia; occupational or residential exposure to toxins, such as lead; family history of anemia or ethnicity suggestive of anemia due to an underlying hemoglobinopathy; and systemic symptoms of an underlying chronic infectious or inflammatory process 27. A review of gastrointestinal symptoms, including abdominal discomfort, hematochezia, and bright red rectal bleeding, is warranted in adults, and a menstrual history should be obtained in menstruating women. Finally, depending on the severity and acuity of the anemia, the patient may have varying levels of fatigue or dyspnea.
Most patients with microcytosis are asymptomatic, and physical examination findings are often limited. As the severity of anemia increases, physical findings may include a systolic murmur and pallor of the mucous membranes, nail beds, and palmar creases 31.
Laboratory Evaluation
Laboratory tests that may help in differentiating the cause of microcytosis include red blood cell distribution width using the complete blood count (CBC), serum iron levels, serum ferritin levels, total iron-binding capacity (TIBC), transferrin saturation, hemoglobin electrophoresis, and occasionally reticulocyte blood count and peripheral blood smears. Data do not support the routine use of other complete blood count (CBC) parameters in the evaluation of microcytosis 32. Table 6 provides a summary of laboratory results that suggest causes of microcytosis 26.
Table 6. Laboratory Tests in the Differential Diagnosis of Low MCV (Microcytosis)
| Test | Suggested diagnosis | |||
|---|---|---|---|---|
| Iron deficiency anemia | Thalassemia | Anemia of chronic disease | Sideroblastic anemia | |
Serum ferritin level | Decreased | Increased | Normal to increased | Normal to increased |
Red blood cell distribution width | Increased | Normal to increased | Normal | Increased |
Serum iron level | Decreased | Normal to increased | Normal to decreased | Normal to increased |
Total iron-binding capacity | Increased | Normal | Slightly decreased | Normal |
Transferrin saturation | Decreased | Normal to increased | Normal to slightly decreased | Normal to increased |
Red blood cell distribution width (RDW) measures the variation in red blood cell size and is often increased in persons with iron deficiency, but normal in those with anemia of chronic disease 26. However, this measurement is not sensitive or specific enough to differentiate iron deficiency and beta-thalassemia trait 33. The red blood cell count can help differentiate the two causes because it is often in the high to normal range with beta-thalassemia trait 34.
Serum iron levels are decreased in iron deficiency anemia and, to a lesser extent, in anemia of chronic disease. Serum iron levels can have diurnal variations with higher concentrations later in the day. Transient increases in serum iron levels may occur with meat ingestion or iron supplementation, but do not represent an increase in true iron stores 35.
Ferritin and transferrin saturation levels and total iron-binding capacity (TIBC) are useful in differentiating iron deficiency anemia and anemia of chronic disease when the serum iron level is decreased. Total iron-binding capacity (TIBC) refers to the ability of unsaturated transferrin, the transport protein for iron, to bind to iron. This measure is usually increased in iron deficiency, decreased in anemia of chronic disease, and normal in the less severe thalassemias. Similar to iron, total iron-binding capacity (TIBC) is affected by diurnal variations.
Transferrin saturation is a percentage calculated as serum iron concentration/TIBC × 100. Levels indicate the number of free binding sites on transferrin. A value less than 16 percent is often indicative of iron deficiency anemia 36.
Ferritin is a complex of iron and the binding protein apoferritin 36. Ferritin reflects true iron stores and is not susceptible to the short-term variations that occur with serum iron levels and total iron-binding capacity (TIBC). However, ferritin is also an acute phase reactant and can be elevated with liver disease, malignancy, and chronic renal disease.18 Iron deficiency anemia is likely if the ferritin level is less than 15 ng per mL (15 mcg per L) in an otherwise healthy person, or less than 50 ng mL (50 mcg per L) in a person with an underlying source of chronic inflammation. Iron deficiency can usually be excluded when the ferritin level is greater than 100 ng per mL (100 mcg per L) 37.
Low MCV diagnostic algorithm
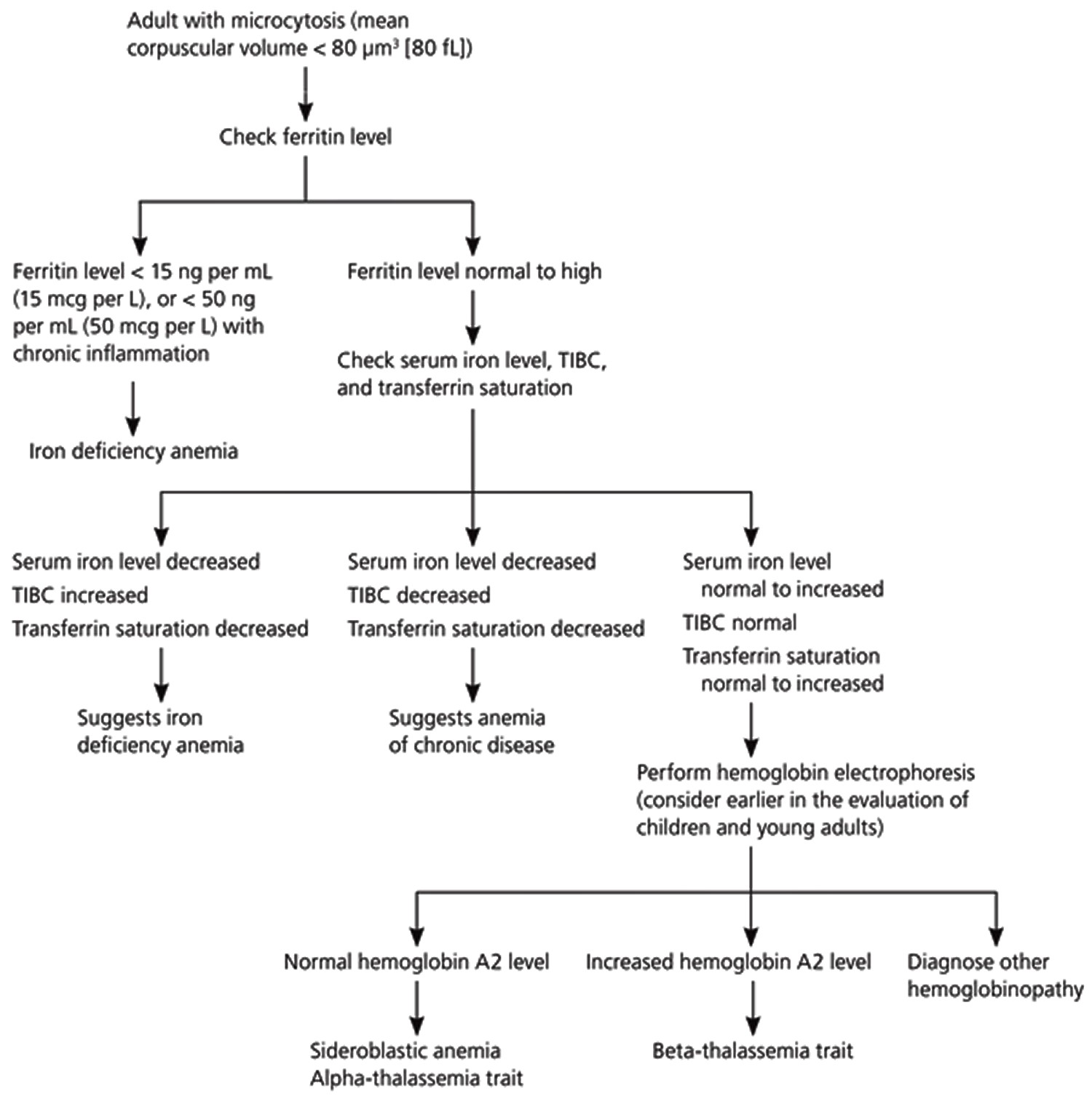
Figure 12 is a suggested algorithm for diagnosing the cause of microcytosis in adults 26. After confirmation of microcytosis on complete blood count, physicians should first order a serum ferritin level 37. If the ferritin level is consistent with iron deficiency anemia, identifying the underlying cause of the anemia is the priority. It is critical to exclude gastrointestinal malignancy in men and nonmenstruating women 38. Gastrointestinal sources of blood loss should also be considered in menstruating women and adolescent girls when the anemia is refractory to iron treatment or when gastrointestinal symptoms are present 39.
If the serum ferritin level is not consistent with iron deficiency anemia, the next stage of the evaluation should include measurement of serum iron level, total iron-binding capacity (TIBC), and transferrin saturation. Iron deficiency anemia is still probable if the serum iron level and transferrin saturation are decreased and total iron-binding capacity (TIBC) is increased. On the other hand, if the serum iron level is decreased and the total iron-binding capacity (TIBC) and transferrin saturation are decreased or normal, anemia of chronic disease is most likely 40.
If the diagnosis remains unclear, hemoglobin electrophoresis can identify beta-thalassemia trait and less common inherited causes of microcytosis. Hemoglobin electrophoresis may be considered earlier in the evaluation of children and young adults, in whom beta-thalassemia trait is more common 41. A bone marrow biopsy can help identify sideroblastic anemia (a group of disorders in which iron is deposited in bone marrow erythrocytes). A serum lead test can detect lead toxicity, and the Centers for Disease Control and Prevention provide guidelines on which high-risk groups of children to screen 42. Even in the presence of lead toxicity, microcytic anemia can be caused by coexisting iron deficiency anemia 43.
Figure 12. Diagnosing the cause of low MCV (microcytosis) algorithm
Low MCV (Microcytosis) Specific Causes
Iron deficiency anemia
Iron deficiency anemia is a common type of anemia and it has many causes. Iron deficiency anemia occurs when the absorption of iron through dietary intake does not match the needs of the body. The mismatch occurs from inadequate dietary intake or increased needs, which usually cause only mild anemia, or from blood loss or malabsorption, which can lead to more significant anemia. Young children, women of childbearing age, and pregnant women have the highest prevalence of the condition 36. Complications include developmental delays and behavior disturbances in children and preterm delivery in pregnant women 44. Heavy menstrual losses leading to significant anemia warrant additional evaluation for clotting disorders (e.g., von Willebrand disease).
Symptoms are related to the overall decrease in the number of red blood cells (red blood cells) and the level of hemoglobin. If the iron deficiency anemia is mild to moderate, there may be no signs or symptoms. In addition to the most common signs and symptoms, there are some that are more unique to iron deficiency and may appear as iron stores in the body are chronically depleted. These may include:
- Brittle or spoon-shaped nails
- Swollen or sore tongue
- Cracks or ulcers at the corners of the mouth
- Difficulty in swallowing
- Craving to eat unusual non-food substances such as ice or dirt (also known as “pica”)
Iron is an essential trace element and is necessary for the production of healthy red blood cells. It is one component of heme, a part of hemoglobin, which is the protein in red blood cells that binds to oxygen and allows red blood cells to transport oxygen throughout the body. If not enough iron is taken in compared to what the body needs, then iron stored in the body begins to be used up. As iron stores are depleted, the body makes fewer red blood cells with decreased amounts of hemoglobin in them, resulting in anemia.
Some of the causes of iron deficiency include:
- Chronic bleeding—if bleeding is excessive over a period of the time (chronic), the body’s stored iron is gradually depleted and, as a result, the body cannot produce enough hemoglobin and red blood cells. In women, iron deficiency may be due to heavy menstrual periods or bleeding fibroids. In older women and in men, the bleeding is usually from disease of the intestines such as ulcers and cancer.
- Dietary deficiency—iron deficiency may be due to a diet poor in iron. Meat, poultry, fish, and iron-fortified foods or dark leafy greens and certain beans are good sources of iron. Children and pregnant or nursing women especially need more iron due to increased requirements. In pregnant women, lack of iron can lead to low birth weight babies and premature delivery. Women who are pregnant or planning to become pregnant routinely take iron supplements to prevent these complications. Newborns who are nursing from deficient mothers tend to have iron deficiency anemia as well.
- Absorption problem—certain conditions affect the absorption of iron from food in the gastrointestinal (GI) tract and over time can result in anemia. These include celiac disease, Crohn disease, intestinal surgery such as gastric bypass, and reduced stomach acid from taking prescription medications.
Iron deficiency anemia laboratory tests
Initial blood tests typically include a complete blood count (CBC). Results may show:
- Hemoglobin (Hb)—may be normal early in the disease but will decrease as anemia worsens
- Red blood cell indices—early on, the red blood cells may be a normal size and color (normocytic, normochromic) but as the anemia progresses, the red blood cells become smaller (microcytic) and paler (hypochromic) than normal.
- Average size of red blood cells (mean corpuscular volume, MCV)—decreased
- Average amount of hemoglobin in red blood cells (mean corpuscular hemoglobin, MCH)—decreased
- Hemoglobin concentration (mean corpuscular hemoglobin concentration, MCHC)—decreased
- Increased variation in the size of red blood cells (red cell distribution width, RDW)
A blood smear may reveal red blood cells that are smaller and paler than normal as well as red blood cells that vary in size (anisocytosis) and shape (poikilocytosis).
If a healthcare provider suspects that someone’s anemia is due to iron deficiency, several follow-up tests may be run to confirm the iron deficiency. These may include:
- Serum iron—the level of iron in someone’s blood, which is usually decreased
- Ferritin—a protein used to store iron; the small quantity of ferritin that is released into the blood is a reflection of the amount of stored iron in the body and is usually low with iron deficiency anemia. It is considered to be the most specific test for identifying iron deficiency anemia, unless infection or inflammation are present.
- Transferrin and total iron-binding capacity (TIBC)—transferrin is a protein that binds to and carries iron through the blood; TIBC is a reflection of how much transferrin is available to bind to iron. In iron deficiency anemia, the transferrin level and TIBC are high.
- Reticulocyte count—reticulocytes are young, immature red blood cells; the number of reticulocytes in iron deficiency anemia is low because there is insufficient iron to produce new red blood cells.
- Soluble transferrin receptor (sTfR)—this test is primarily ordered to help distinguish between anemia that is caused by iron deficiency and anemia that is caused by inflammation or a chronic illness. It may be ordered as an alternative to ferritin when a person has a chronic illness and/or inflammation is present or suspected. It will be high in iron deficiency.
If the iron deficiency is thought to be due to chronic blood loss, such as GI (gastrointestinal) tract bleeding, then other tests and procedures may be performed. Laboratory tests that may be able to detect GI bleeding are the fecal occult blood test (FOBT) or fecal immunochemical test (FIT).
A test for Helicobacter pylori may detect a bacterium that can cause ulcers in the GI (gastrointestinal) tract that may be a cause of chronic bleeding. If any of these tests are positive or if it is strongly suspected that a GI (gastrointestinal) bleed exists, then procedures such as endoscopy or colonoscopy may be done to find the location of the bleeding so that it can be treated.
Iron deficiency anemia treatment
Treatment of iron deficiency typically involves iron supplements and/or a change in diet. Vitamin C also helps with iron absorption. However, if iron-deficiency is suspected to result from abnormal blood loss, further testing is often required to determine the reason for the bleeding. People with severe iron deficiency may require a transfusion of blood cells or iron therapy through intravenous (IV) or injections. When the underlying cause is found and treated, then the anemia usually resolves.
Thalassemia
Thalassemia is a group of inherited blood disorders that can be passed from parents to their children and affect the amount and type of hemoglobin the body produces.
Hemoglobin (Hb or Hgb) is a substance present in all red blood cells (RBCs). It is important for proper red blood cell function because it carries the oxygen that RBCs deliver around the body. One portion of hemoglobin called heme is the molecule with iron at the center. Another portion is made of up four protein chains called globins. Each of the four globin chains holds a heme group containing one iron atom. Depending on their structure, the globin chains are designated as alpha, beta, gamma, or delta.
Not all hemoglobin is the same. Different types of hemoglobin are classified according to the type of globin chains they contain. The type of globin chains present is important in hemoglobin’s ability to transport oxygen.
Normal hemoglobin types include:
- Hemoglobin A – this is the predominant type of Hb in adults (about 95-98%); Hb A contains two alpha (α) protein chains and two beta (ß) protein chains.
- Hb A2 – makes up about 2-3.5% of Hb found in adults; it has two alpha (α) and two delta (δ) protein chains.
- Hb F – makes up to 2% of Hb found in adults; it has two alpha (α) and two gamma (γ) protein chains. Hb F is the primary hemoglobin produced by a developing baby (fetus) during pregnancy. Its production usually falls to a low level within a year after birth.
People with thalassemia have one or more genetic mutations that they have inherited and that result in a decreased production of normal hemoglobin. When the body doesn’t make enough normal hemoglobin, red blood cells do not function properly and oxygen delivery suffers. This can lead to anemia with signs and symptoms that can range from mild to severe, depending on the type of thalassemia that a person has. Examples of signs and symptoms include weakness, fatigue, and pale skin (pallor). See the Classifications section for more about the signs, symptoms, and complications of the different types of thalassemia.
For hemoglobin, there are four genes in our DNA that code for the alpha globin chains and two genes (each) for the beta, delta, and gamma globin chains. Since everyone inherits a set of chromosomes from each parent, each person inherits two alpha globulin genes and one beta globulin gene from each parent.
With thalassemias, mutations in one or more of the globin genes cause a reduction in the amount of the particular globin chain produced. This can upset the balance of alpha to beta chains, resulting in unusual forms of hemoglobin or an increase in the amount of normally minor hemoglobin, such as Hb A2 or Hb F. The thalassemias are usually classified by the type of globin chain whose synthesis is decreased. For example, the most common alpha chain-related condition is called alpha thalassemia. The severity of this condition depends on the number of genes affected.
Beta-thalassemia is an autosomal recessive genetic condition in which the normal beta globin chains that make up hemoglobin are underproduced. Beta-thalassemia trait is the heterozygous form of the disease. Beta- thalassemia major (also known as Cooley anemia) is the homozygous form. Both are more common in black persons, and in persons of Southeast Asian, Greek, Italian, or Mediterranean descent 26.
Most patients with beta-thalassemia trait have mild anemia (hemoglobin level is rarely less than 9.3 g per dL [93 g per L]). In addition, the mean corpuscular volume can sometimes reach much lower levels than with iron deficiency anemia alone. Ultimately, the diagnosis of beta-thalassemia trait is made when hemoglobin electrophoresis shows a slight increase in hemoglobin A2 45. Coexisting iron deficiency anemia can lower hemoglobin A2 levels; therefore, iron deficiency anemia must be corrected before hemoglobin electrophoresis results can be appropriately evaluated 46.
Alpha-thalassemia is caused by an underproduction of alpha globin chains, and is most prevalent in persons of African or Southeast Asian descent. The production of alpha globin chains is controlled by four genes on two chromosomes. One gene deletion results in a silent carrier status with normal hematologic findings. Two gene deletions result in alpha-thalassemia trait, which usually leads to microcytosis without anemia. Hemoglobin electrophoresis is often normal in patients with silent carrier status or with alpha-thalassemia trait. The diagnosis is usually one of exclusion 47.
Thalassemia Tests and Diagnosis
Several laboratory tests may be used to help detect and diagnose thalassemia:
Complete blood count (CBC). The CBC is an evaluation of the cells in the blood. Among other things, the CBC determines the number of red blood cells present and how much hemoglobin is in them. It evaluates the size and shape of the red blood cells present, reported as the red cell indices. These include the mean corpuscular volume (MCV), a measurement of the size of the red blood cells. A low MCV is often the first indication of thalassemia. If the MCV is low and iron deficiency has been ruled out as a cause, thalassemia should be considered.
Blood smear (also called peripheral smear and manual differential). In this test, a trained laboratory professional examines a thin layer of blood that is treated with a special stain, on a slide, under a microscope. The number and type of white blood cells, red blood cells, and platelets are evaluated to see if they are normal and mature. With thalassemia, the red blood cells often appear smaller than normal (microcytic, low MCV). Red cells may also:
- Be paler than normal (hypochromic or low MCH)
- Vary in size and shape (anisocytosis and poikilocytosis)
- Be nucleated (normal, mature red blood cells do not have a nucleus)
- Have uneven hemoglobin distribution (producing “target cells” that look like a bull’s-eye under the microscope)
The greater the percentage of abnormal-looking red blood cells, the greater the likelihood of an underlying disorder and decreased ability of the red blood cells to cary oxygen.
Iron studies. These may include: iron, ferritin, unsaturated iron binding capacity (UIBC), total iron binding capacity (TIBC), and percent saturation of transferrin. These tests measure different aspects of the body’s iron storage and usage. The tests are ordered to help determine whether an iron deficiency is the cause of a person’s anemia. One or more of them may also be ordered to help monitor the degree of iron overload in an individual with thalassemia.
- Alpha thalassemia is sometimes confused with iron deficiency anemia because both disorders have smaller than usual (microcytic) red blood cells. If someone has thalassemia, his or her iron levels are not expected to be low. Iron therapy will not help people with alpha thalassemia and may lead to iron overload, which can cause organ damage over time.
- Erythrocyte porphyrin tests may be used to distinguish an unclear beta thalassemia minor diagnosis from iron deficiency or lead poisoning. Individuals with beta thalassemia will have normal porphyrin levels, but those with the latter conditions will have elevated porphyrin.
Hemoglobinopathy (Hb) evaluation (hemoglobin electrophoresis). This test assess the type and relative amounts of hemoglobin present in red blood cells. Hemoglobin A (Hb A), composed of both alpha and beta globin, is the type of hemoglobin that normally makes up 95% to 98% of hemoglobin in adults. Hemoglobin A2 (HbA2) is usually 2% to 3% of hemoglobin in adults, while hemoglobin F usually makes up less than 2%.
Beta thalassemia upsets the balance of beta and alpha hemoglobin chain formation and causes an increase in those minor hemoglobin components. So individuals with the beta thalassemia major usually have larger percentages of Hb F. Those with beta thalassemia minor usually have elevated fraction of Hb A2. Hb H is a less common form of hemoglobin that may be seen in some cases of alpha thalassemia. Hb S is the hemoglobin more common in people with sickle cell disease.
Hemoglobinopathy (Hb) evaluations are used for state-mandated newborn hemoglobin screening and prenatal screening when parents are at high risk for hemoglobin abnormalities.
DNA analysis. These tests are used to help confirm mutations in the alpha and beta globin-producing genes. DNA testing is not routinely done but can be used to help diagnose thalassemia and to determine carrier status, if indicated.
- For beta thalassemia, the hemoglobin beta gene, HBB, may be analyzed or sequenced to confirm the presence of thalassemia-causing mutations. Genetic tests may also be given for other HBB mutations such as Hb S mutation, which is associated with sickle cell disease. More than 250 mutations have been associated with beta thalassemia, though some cause no signs or symptoms. However, others decrease the amount of beta globin production and some prevent it completely. The presence of one of those mutations confirms a diagnosis of beta thalassemia.
- The primary molecular test available for alpha thalassemia detects common mutations (e.g., deletions) in the two alpha genes HBA1 and HBA2. Each person has two copies of each of these genes, called alleles, in their cells, one from their mother and one from their father. These alleles govern alpha globin production and if mutations lead to functional loss of one or more of alpha genes, alpha thalassemia occurs.
Since having relatives who carry mutations for thalassemia increases a person’s risk of carrying the same mutant gene, family studies may be done to evaluate carrier status and the types of mutations present in other family members if deemed necessary by a healthcare practitioner.
Genetic testing of amniotic fluid is used in the rare instances a fetus is at increased risk for thalassemia. This is especially important if both parents likely carry a mutation because that increases the risk that their child may inherit a combination of abnormal genes, causing a more severe form of thalassemia.
Thalassemia treatment
Most individuals with mild thalassemia traits require no treatment. They may want to consider genetic counseling, however, because they may pass the mutant gene on to their children.
People with hemoglobin H disease or beta thalassemia intermedia will experience variable amounts of anemia throughout their life. They can live relatively normal lives but will require regular monitoring and may occasionally need blood transfusion. Folic acid supplementation is often given, but iron supplementation is not recommended.
Those with beta thalassemia major will usually require regular blood transfusions, as frequently as every few weeks, and chelation therapy to remove iron throughout their life. These transfusions help maintain hemoglobin at a high enough level to provide oxygen to the body and prevent growth abnormalities and organ damage. Frequent transfusions, however, can raise body iron to toxic levels, resulting in deposits of iron in the liver, heart, and other organs. Regular iron chelation therapy is used to help decrease iron in the body.
Bone marrow transplant known as hematopoietic stem cell transplantation can also be used for treatment of beta thalassemia major.
Fetuses with alpha thalassemia major are usually miscarried, stillborn, or die shortly after birth. Experimental treatments, such as fetal blood transfusions and even fetal marrow transplant, have been successful in a very few cases in bringing a baby to term.
Anemia of chronic disease
Some chronic (long-term) illnesses can cause anemia. Anemia of chronic disease can be caused by chronic infections or inflammatory processes. Increased levels of cytokines cause a decrease in erythropoietin production, a decreased response to erythropoietin, and interference with iron metabolism 48. Although anemia of chronic disease is usually normocytic, about one fourth to one third of cases are mildly microcytic. The anemia is usually mild and not progressive. Additionally, although serum iron levels are decreased in anemia of chronic disease (similar to iron deficiency anemia), ferritin levels are increased because ferritin is an acute phase reactant 26.
Often, anemia caused by chronic diseases goes undetected until a routine test such as a complete blood count (CBC) reveals abnormal results. Several follow-up tests may be used to determine the underlying cause. There are many chronic conditions and diseases that can result in anemia. Some examples of these include:
- Kidney disease—red blood cells are produced by the bone marrow in response to a hormone called erythropoietin, made primarily by the kidneys. Chronic kidney disease can cause anemia resulting from too little production of this hormone; the anemia can be treated by giving erythropoietin injections.
- Anemia of chronic disease—whenever there are chronic diseases that stimulate the body’s inflammatory response, the ability of the bone marrow to respond to erythropoietin is decreased, mainly due to impairment in body iron regulation. For example, rheumatoid arthritis (a severe form of joint disease caused by the body attacking its own joints, called an autoimmune disease) can cause anemia by this mechanism. Other diseases that can produce anemia in the same way include chronic infections such as HIV or tuberculosis (TB).
Chronic disease anemia laboratory tests
A number of tests may be used as follow up to abnormal results of initial tests such as a CBC and blood smear to determine the underlying cause of chronic anemia. Some of these may include:
- Reticulocyte count—will typically be low
- Comprehensive metabolic panel—used to detect evidence of chronic disorders
Tests for anemia of chronic disease may include:
- Tests for inflammation such as C-Reactive Protein (CRP)
- Erythropoietin—is typically mildly increased
- Tests for infections such as HIV and TB
- Iron and transferrin (TIBC)—are typically both low
- Soluble transferrin receptor (sTfR)—is typically normal or low
Chronic disease anemia treatment
Treatment of anemia due to chronic conditions usually involves determining and/or resolving the underlying disease. Blood transfusions may be used to treat the condition in the short term.
References- Seppä K, Heinilä K, Sillanaukee P, Saarni M. Evaluation of macrocytosis by general practitioners. J Stud Alcohol. 1996;57(1):97–100.
- Rumsey SE, Hokin B, Magin PJ, Pond D. Macrocytosis—an Australian general practice perspective. Aust Fam Physician. 2007;36(7):571–572.
- Evaluation of Macrocytosis. Am Fam Physician. 2009 Feb 1;79(3):203-208. https://www.aafp.org/afp/2009/0201/p203.html
- Seppä K, Laippala P, Saarni M. Macrocytosis as a consequence of alcohol abuse among patients in general practice. Alcohol Clin Exp Res. 1991;15(5):871–876.
- Kaptan K, Beyan C, Ural AU, et al. Helicobacter pylori–is it a novel causative agent in Vitamin B12 deficiency? Arch Intern Med. 2000;160(9):1349–1353.
- Maruyama S, Hirayama C, Yamamoto S, et al. Red blood cell status in alcoholic and non-alcoholic liver disease. J Lab Clin Med. 2001;138(5):332–337.
- Savage DG, Lindenbaum J, Stabler SP, Allen RH. Sensitivity of serum methylmalonic acid and total homocysteine determinations for diagnosing cobalamin and folate deficiencies. Am J Med. 1994;96(3):239–246.
- Miller MA, Martinez V, McCarthy R, Patel MM. Nitrous oxide “whippit” abuse presenting as clinical B12 deficiency and ataxia. Am J Emerg Med. 2004;22(2):124.
- Zimran A, Hershko C. The changing pattern of megaloblastic anemia: megaloblastic anemia in Israel. Am J Clin Nutr. 1983;37(5):855–861.
- Chui CH, Lau FY, Wong R, et al. Vitamin B12 deficiency—need for a new guideline. Nutrition. 2001;17(11–12):917–920.
- Savage D, Lindenbaum J. Relapses after interruption of cyanocobalamin therapy in patients with pernicious anemia. Am J Med. 1983;74(5):765–772.
- Suh JR, Herbig AK, Stover PJ. New perspectives on folate catabolism. Annu Rev Nutr. 2001;21:255–282.
- Waxman S, Metz J, Herbert V. Defective DNA synthesis in human megaloblastic bone marrow: effects of homocysteine and methionine. J Clin Invest. 1969;48(2):284–289.
- Moran RG, Keyomarsi K. Biochemical rationale for the synergism of 5-fluorouracil and folinic acid. NCI Monogr. 1987;(5):159–163.
- Ortiz Z, Shea B, Suarez Almazor M, Moher D, Wells G, Tugwell P. Folic acid and folinic acid for reducing side effects in patients receiving methotrexate for rheumatoid arthritis. Cochrane Database Syst Rev. 2000;(2):CD000951.
- Robinson AR, Mladenovic J. Lack of clinical utility of folate levels in the evaluation of macrocytosis or anemia. Am J Med. 2001;110(2):88–90.
- Phekoo K, Williams Y, Schey SA, Andrews VE, Dudley JM, Hoff-brand AV. Folate assays: serum or red cell? J R Coll Physicians Lond. 1997;31(3):291–295.
- Geené D, Sudre P, Anwar D, Goehring C, Saaïdia A, Hirschel B. Causes of macrocytosis in HIV-infected patients not treated with zidovudine. Swiss HIV Cohort Study. J Infect. 2000;40(2):160–163.
- DRUGDEX System, PDR Electronic Library. Thompson Micromedex. Greenwood Village, Co.; 1974–2007.
- Dang CV. Runner’s anemia. JAMA. 2001;286(6):714–716.
- Wachtel TJ, Pueschel SM. Macrocytosis in Down Syndrome. Am J Ment Retard. 1991;95(4):417–420.
- World Health Organization. Nutritional Anaemias: Report of a WHO Scientific Group. Geneva: World Health Organization; 1968.
- Gloster ES, Strauss RA. More on spurious red blood cell parameters. Am J Clin Pathol. 1985;84(6):775–776.
- van Duijnhoven HL, Treskes M. Marked interference of hyperglycemia in measurements of mean (red) cell volume by Technicon H analyzers. Clin Chem. 1996;42(1):76–80.
- Francis DA, Francis JL, Roath OS. Improved assessment of haemoglobin and red cell indices in blood samples with high white cell counts. Med Lab Sci. 1985;42(3):285–286.
- Hematologic diseases. In: Wallach J. Interpretation of Diagnostic Tests. 8th ed. Boston, Mass.: Little Brown and Company; 2006:385–419.
- Richardson M. Microcytic anemia [published corrections appear in Pediatr Rev. 2007;28(7):275, Pediatr Rev. 2009;30(5):181, and Pediatr Rev. 2007;28(4):151]. Pediatr Rev. 2007;28(1):5–14.
- Evaluation of Microcytosis. Am Fam Physician. 2010 Nov 1;82(9):1117-1122. https://www.aafp.org/afp/2010/1101/p1117.html
- Moreno Chulilla JA, Romero Colás MS, Gutiérrez Martín M. Classification of anemia for gastroenterologists. World J Gastroenterol. 2009;15(37):4627–4637.
- Guralnik JM, Eisenstaedt RS, Ferrucci L, Klein HG, Woodman RC. Prevalence of anemia in persons 65 years and older in the United States: evidence for a high rate of unexplained anemia. Blood. 2004;104(8):2263–2268.
- Muhe L, Oljira B, Degefu H, Jaffar S, Weber MW. Evaluation of clinical pallor in the identification and treatment of children with moderate and severe anaemia [published correction appears in Trop Med Int Health. 2001;6(4):326]. Trop Med Int Health. 2000;5(11):805–810.
- AlFadhli SM, Al-Awadhi AM, AlKhaldi D. Validity assessment of nine discriminant functions used for the differentiation between iron deficiency anemia and thalassemia minor. J Trop Pediatr. 2007;53(2):93–97.
- Ntaios G, Chatzinikolaou A, Saouli Z, et al. Discrimination indices as screening tests for beta-thalassemic trait. Ann Hematol. 2007;86(7):487–491.
- Demir A, Yarali N, Fisgin T, Duru F, Kara A. Most reliable indices in differentiation between thalassemia trait and iron deficiency anemia. Pediatr Int. 2002;44(6):612–616.
- Brugnara C. Iron deficiency and erythropoiesis: new diagnostic approaches. Clin Chem. 2003;49(10):1573–1578.
- Recommendations to prevent and control iron deficiency in the United States. Centers for Disease Control and Prevention. MMWR Recomm Rep. 1998;47(RR-3):1–29.
- Knovich MA, Storey JA, Coffman LG, Torti SV, Torti FM. Ferritin for the clinician. Blood Rev. 2009;23(3):95–104.
- Hershko C, Skikne B. Pathogenesis and management of iron deficiency anemia: emerging role of celiac disease, helicobacter pylori, and autoimmune gastritis. Semin Hematol. 2009;46(4):339–350.
- Vannella L, Aloe Spiriti MA, Cozza G, et al. Benefit of concomitant gastrointestinal and gynaecological evaluation in premenopausal women with iron deficiency anaemia. Aliment Pharmacol Ther. 2008;28(4):422–430
- Thomas C, Thomas L. Anemia of chronic disease: pathophysiology and laboratory diagnosis. Lab Hematol. 2005;11(1):14–23.
- Jain S, Kamat D. Evaluation of microcytic anemia. Clin Pediatr (Phila). 2009;48(1):7–13.
- Centers for Disease Control and Prevention. Screening for lead at the domestic refugee medical examination. https://www.cdc.gov/immigrantrefugeehealth/pdf/lead.pdf
- Clark M, Royal J, Seeler R. Interaction of iron deficiency and lead and the hematologic findings in children with severe lead poisoning. Pediatrics. 1988;81(2):247–254.
- U.S. Preventive Services Task Force. Screening for iron deficiency anemia. https://www.uspreventiveservicestaskforce.org/Page/Document/UpdateSummaryFinal/iron-deficiency-anemia-in-pregnant-women-screening-and-supplementation
- Henderson S, Timbs A, McCarthy J, et al. Incidence of haemoglobinopathies in various populations-the impact of immigration. Clin Biochem. 2009;42(18):1745–1756.
- El-Agouza A, Abu Shahla A, Sirdah M. The effect of iron deficiency anaemia on the levels of haemoglobin subtypes: possible consequences for clinical diagnosis. Clin Lab Haemotol. 2002;24(5):285–289.
- Muncie HL Jr, Campbell J. Alpha and beta thalassemia. Am Fam Physician. 2009;80(4):339–344.
- Thomas C, Kirschbaum A, Boehm D, Thomas L. The diagnostic plot: a concept for identifying different states of iron deficiency and monitoring the response to epoetin therapy. Med Oncol. 2006;23(1):23–36.