What are monocytes
Monocytes make up 5-10% of the total white blood cell count about 2×105/ml 1. Monocytes are circulating mononuclear phagocytic white blood cell of the innate immune system that ingest pathogens and cellular debris and present antigen to T cells 2. After circulating for several days in the bloodstream, monocytes usually undergo extravasation. Monocytes have the capacity to differentiate into macrophages when they extravasate from the vasculature 3. This differentiation can, in the presence of the right environmental cues, be re-directed instead to dendritic cells (DCs). Once a monocyte extravasates into tissue, a differentiation program toward macrophages or dendritic cells (DCs) begins, and the cell should no longer be referred to as a monocyte, but rather as a monocyte-derived cell 4. “Patrolling” monocytes constantly migrate along the endothelium in blood vessels serving as vascular innate immune system 5. Major macrophage functions include phagocytosis of tissue debris and foreign materials, steady state defense and tissue repair after injuries. When macrophages become dysfunctional, they may promote disease; for example, ineffective clearance of dead cells by macrophages exacerbates atherosclerosis. The cells are also nodes of information transfer and signal amplification, facilitated, for instance, by amplifying pro-or anti-inflammatory cytokines. In addition, macrophages are antigen presenting cells and express major histocompatibility antigens.
Monocytes help get rid of dead or damaged tissue and regulate your body’s immune response. Infections, cancer, autoimmune diseases and other conditions can cause an increased number of monocytes. A decreased number can be the result of toxins, chemotherapy and other causes.
Monocytes are similar to neutrophils, they move to an area of infection and engulf and destroy bacteria. Monocytes are associated more often with chronic rather than acute infections.
Current literature suggests that monocytes may mainly serve as precursors for potent antigen-presenting dendritic cells (DCs) in peripheral organs, and particularly so during inflammation. There they may acquire antigens from parenchymal cells or pathogens that they encounter, and they would process these along with foreign antigens they may have picked up much earlier in the bone marrow or blood for loading onto major histocompatibility complex (MHC) molecules. Though they migrate with these major histocompatibility complex (MHC)/peptide complexes to downstream lymph nodes and simultaneously elevate expression of other costimulatory molecules, it remains unknown whether they have a unique role in directly presenting antigen to T cells. Instead, they may share antigen, and likely intact major histocompatibility complex (MHC)/peptides, with other dendritic cells (DCs) that then also or alternatively interact with T cells.
Dendritic cells (DCs) are specialized antigen-presenting cells that can efficiently initiate immune responses 6, and there has been a long interest in the role of monocytes as potential precursors for these cells. A growing body of evidence indicates that many dendritic cell populations are not typically derived from monocytes 7. Nonetheless, monocytes do have a role as precursors for important antigen-presenting cells, including dendritic cells.
Monocytes can be specified into different subsets such as CD16high14− monocytes which produce high amounts of inflammatory cytokines such as tumor necrosis factor or a more regulatory CD16low14+ monocyte subset 8.
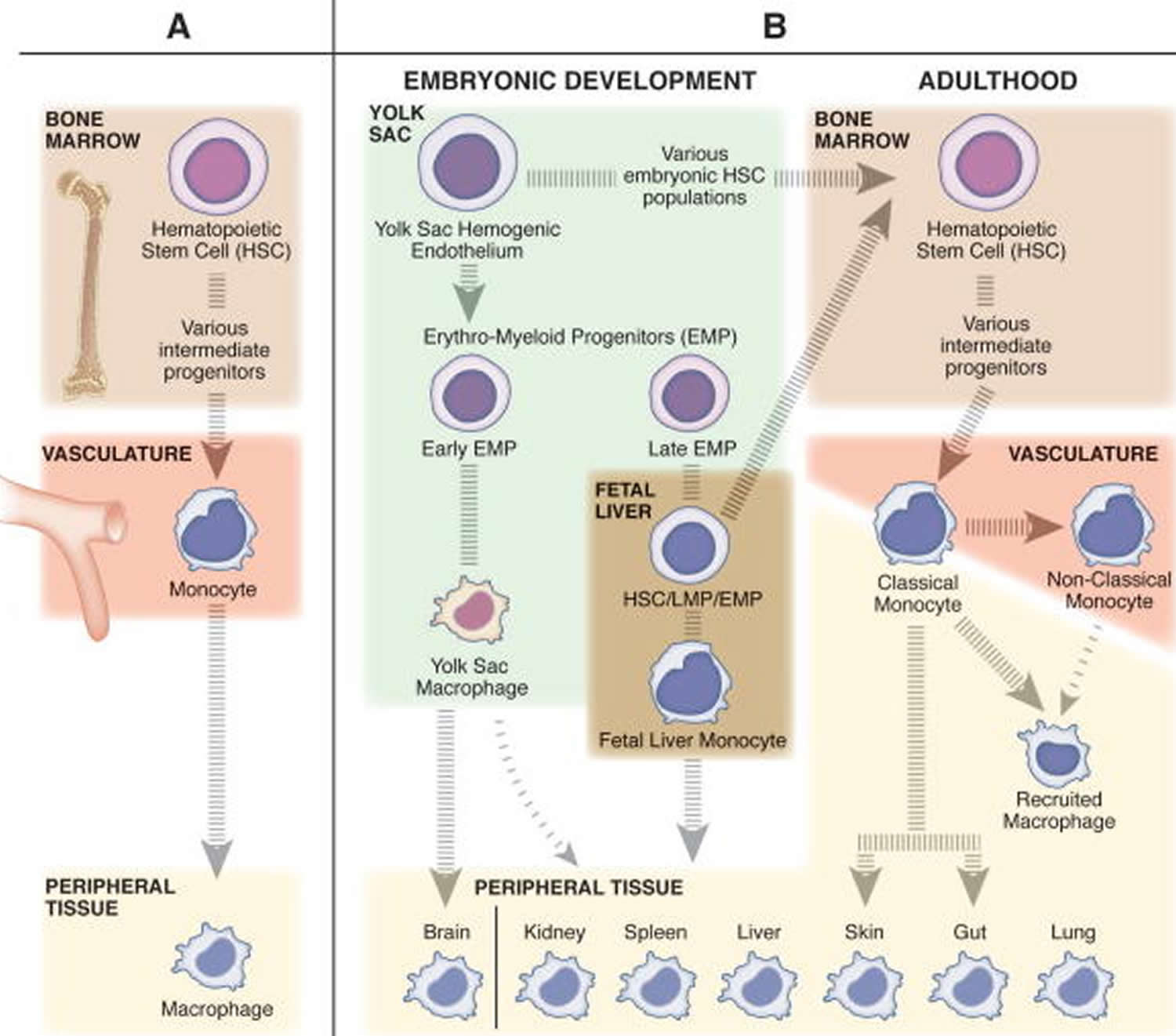
Figure 1. The diverse origins of monocytes and macrophages
Footnotes: (A) The classic belief that tissue resident macrophages are populated from monocytes through adult hematopoiesis. In the bone marrow, hematopoietic stem cells (HSCs) differentiate into monocytes through a series of intermediate progenitors, extravasate from the vasculature, and differentiate into macrophages to maintain the tissue resident niches. (B) The more contemporary view that tissue resident macrophages undergo self-renewal, and have a distinct embryonic origin sourced from populations of erythro-myeloid progenitors (EMPs) derived from the yolk sac hemogenic endothelium. The early population of EMPs differentiate into yolk sac macrophages and then migrate to the tissues and contribute to the tissue resident macrophage population. The late population of EMPs gives rise to fetal liver monocytes through an unknown intermediate embryonic progenitor cell type (maybe EMP, HSC, or lympho-myeloid progenitor (LMP)). Fetal liver monocytes then go on to seed the tissue resident macrophage populations for all peripheral tissues other than the brain. Only the skin and gut resident macrophage populations are recognized to be replenished in homeostatic conditions by bone marrow derived cells, specifically classical monocytes. Classical monocytes (inflammatory monocytes) patrol the peripheral tissues in homeostatic conditions, while nonclassical (anti-inflammatory monocytes) surveil the vasculature. In inflammatory conditions, classical monocytes, and to a lesser extent nonclassical monocytes, can leave the vasculature and differentiate into recruited macrophages to transiently regulate the immune response.
[Source 9]The two subsets of monocytes do not arise from distinct lineages but instead the Ly-6Clo monocyte subset is a product of the Ly-6Chi monocyte subset 10. However, this conversion is not obligatory. Ly-6Chi monocytes often extravasate from the bloodstream before they convert to Ly-6Clo monocytes, and their differentiation pathway in tissues does not necessarily recapitulate the development of Ly-6Clo monocytes in the bloodstream. Befitting the definition for monocytes, there is evidence that both human monocyte subsets readily differentiate to macrophages 11. In mice, Ly6Chi monocytes contribute to macrophage pools in vivo during inflammation, such as in atherosclerosis 12 or other inflammatory settings 13, and Ly-6Clo monocytes have also been proposed to contribute to macrophage populations 14. When the antigen-presenting capacity of the monocyte subsets is tested through mixed lymphocyte reaction in vitro, human CD16+ monocytes 15 and their mouse counterparts 16 respectively show enhanced capacity for driving allogeneic T cell proliferation, compared with the other subset of monocytes.
Figure 2. Monocyte subsets
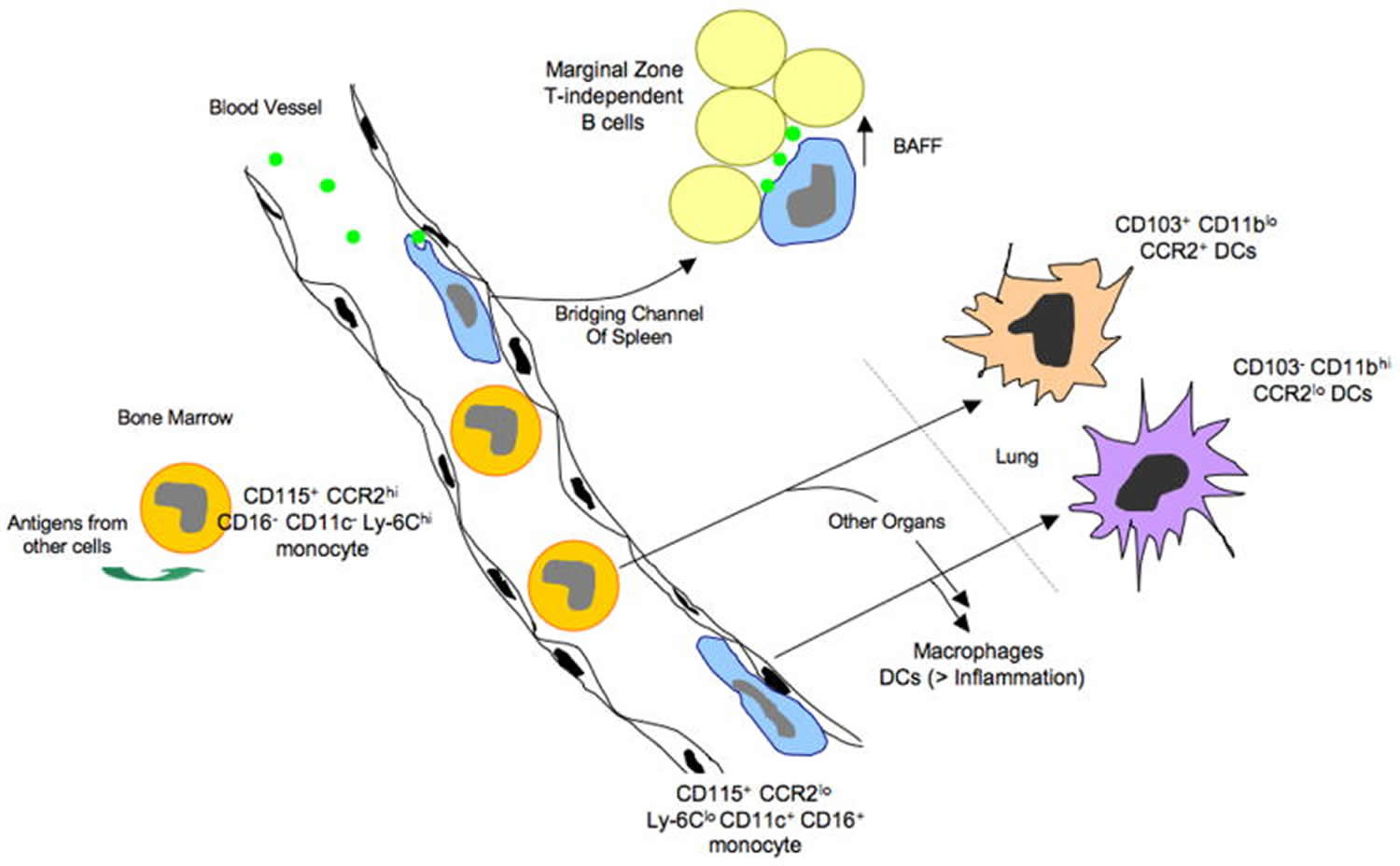 Footnotes: Monocyte subsets, access to antigen, and possible relationships to dendritic cells (DCs) and antigen-presenting cells that drive T-independent antibody production from innate B cells: model from the mouse. Monocytes emerge from the bone marrow as a CCR2hi Ly-6Chi population. In the bone marrow, they can take up antigens from at least some other neighboring cells, and these antigens can be presented later in the life-cycle of the monocyte. Once mobilized to the bloodstream, some CCR2hi monocytes convert to a subset of monocytes that expresses CD16 and CD11c and has low levels of CCR2 and Ly-6C. These monocytes crawl along the endothelial surfaces of the vessel wall and are able to preferentially acquire particulates that enter the bloodstream, possibly due to their marginated positioning that would give them an advantage in capturing particles. CCR2loLy-6Clo CD11c+ monocytes fit the description of DCs that have been reported to promote T-independent antibody responses by picking up particulate antigens in the blood and presenting them to B cells in the bridging channels of the spleen. Support of T-dependent immunity by monocyte-derived cells occurs in part when they differentiate into dendritic cells, a step that is facilitated by inflammatory signals. Both subsets of monocytes have the ability to become dendritic cells (DCs), in addition to macrophages, in various body organs. In the lung, the two subsets of monocytes respectively give rise to dendritic cells (DCs) with distinct characteristics.
[Source 4]
Footnotes: Monocyte subsets, access to antigen, and possible relationships to dendritic cells (DCs) and antigen-presenting cells that drive T-independent antibody production from innate B cells: model from the mouse. Monocytes emerge from the bone marrow as a CCR2hi Ly-6Chi population. In the bone marrow, they can take up antigens from at least some other neighboring cells, and these antigens can be presented later in the life-cycle of the monocyte. Once mobilized to the bloodstream, some CCR2hi monocytes convert to a subset of monocytes that expresses CD16 and CD11c and has low levels of CCR2 and Ly-6C. These monocytes crawl along the endothelial surfaces of the vessel wall and are able to preferentially acquire particulates that enter the bloodstream, possibly due to their marginated positioning that would give them an advantage in capturing particles. CCR2loLy-6Clo CD11c+ monocytes fit the description of DCs that have been reported to promote T-independent antibody responses by picking up particulate antigens in the blood and presenting them to B cells in the bridging channels of the spleen. Support of T-dependent immunity by monocyte-derived cells occurs in part when they differentiate into dendritic cells, a step that is facilitated by inflammatory signals. Both subsets of monocytes have the ability to become dendritic cells (DCs), in addition to macrophages, in various body organs. In the lung, the two subsets of monocytes respectively give rise to dendritic cells (DCs) with distinct characteristics.
[Source 4]Though it is known that CD16+ monocytes drive expansion of T cells in a mixed lymphocyte reaction better than CD16− monocytes 17, the role of monocytes per se as antigen-presenting cells is rarely considered. Typically, their role as antigen-presenting cells is mainly confined to discussions and studies that attempt to determine if they serve as precursors for particular populations of macrophages or dendritic cells (DCs). Recently, however, we have found evidence that antigens that monocytes encounter before they extravasate into tissues are sufficiently retained by monocytes such that those antigens can become available for presentation long after they were acquired 18. At least in experimental settings, monocytes took up exogenous cell-associated antigens in the bone marrow (Figure 2) and were able to present peptides derived from these antigens on MHC II molecules much later in their life cycle. This feature of retaining antigen for presentation later, after appropriate maturation, is a characteristic that has been ascribed mainly to dendritic cells (DCs) and is thought to distinguish them from macrophages 19, which have little capacity to retain antigens for delayed presentation. Monocytes may be less proteolytic than macrophages 20, allowing for retention rather than degradation of antigens.
If monocytes are able to capture antigen normally within the bone marrow or during their transit through blood, they may be critical vehicles for the supply of antigen to other organ systems. For example, it has been argued that antigen-presenting cells that transit through blood can supply tissue-restricted antigens to the thymus 21, possibly filling in gaps of self-antigen that may be poorly expressed by thymic epithelium, even though the latter possess elegant mechanisms to express a wide-range of tissue-restricted antigens 22. Studies using mice that are surgically joined and therefore share a common circulatory system (parabiosis) indicate that antigen-presenting cells from the blood enter the thymus on an ongoing basis 21. Monocytes that differentiate into dendritic cells (DCs) may provide a source of antigen to be presented to thymocytes within the thymus, and if so, the capacity of monocytes to capture antigen from other cells prior to extravasation from blood would extend the range of antigens that they might present after differentiating into thymic dendritic cells (DCs).
Dendritic cells (DCs) are the most potent antigen-presenting cells in the immune system for promoting primary activation of naïve T cells 23, so if monocytes are significant precursors for dendritic cells (DCs), then they would be central to antigen presentation that drives primary T-dependent immune responses. However, several recent publications collectively agree that monocytes do not serve as precursors for the major splenic CD11chi dendritic cell populations in the steady state 24, including in particular the classical CD8α+ and CD8α− dendritic cell subsets described in mice 25. One study indicated that Ly-6Clo monocytes might make a minor contribution to splenic dendritic cells (DCs) in the steady state 24, but this is unlikely since elegant parabiosis studies are inconsistent with this possibility 26. It is unclear whether these studies would have included in their analysis the CD11clo cells that Kearney et al. 27 indicated contributed to antigen presentation to B cells during T-independent responses. In contrast, dendritic cells (DCs) in peripheral organs like lung and intestine do appear to arise from monocytes on an ongoing basis, raising the interesting and unexpected possibility that the origin of dendritic cells (DCs) in nonlymphoid tissues is different than in spleen. Thus, at least some dendritic cells (DCs) in lymph nodes—those that come into the lymph node from upstream lymphatics—may very well derive from monocytes.
The phenotype of Ly-6Clo monocytes is more akin to dendritic cells (DCs) than that of Ly-6Chi monocytes and we have found that the human counterparts of Ly-6Clo monocytes possessed migratory behavior in vitro reminiscent of dendritic cells (DCs) 28. Thus, scientists have wondered whether the two subsets of monocytes have differential capacity to become dendritic cells (DCs) or macrophages. Since both Ly-6Clo and Ly-6Chi monocyte subsets migrate into the lung on a continuous basis 14, tracking the fate of the two monocyte subsets in the lung provides a good model to compare their differentiation potentials.
What are white blood cells?
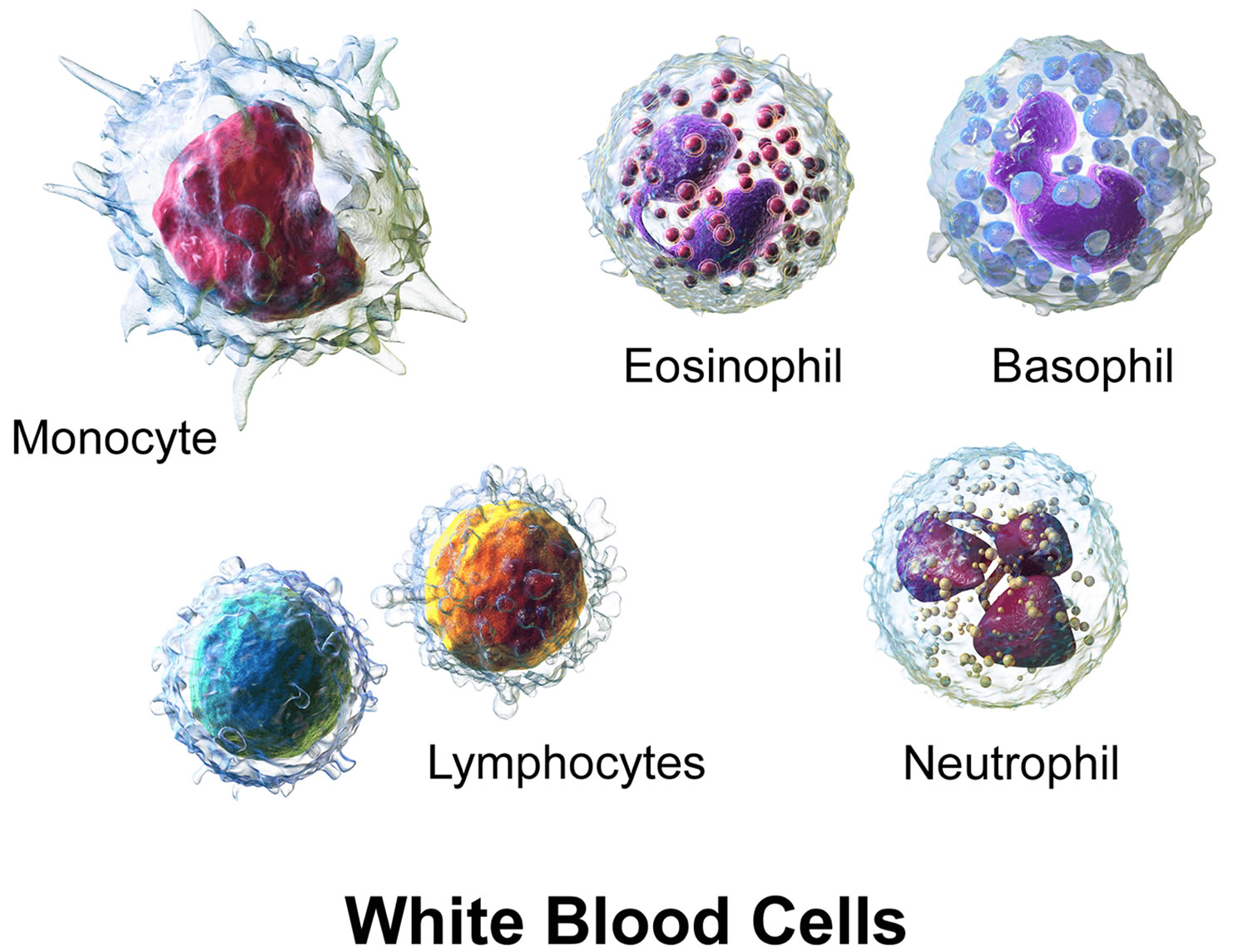
White blood cells (WBCs), also called leukocytes, are cells that circulate in the blood and the lymphatic system that help protect the body against infections. They are an important part of the body’s immune system and also have a role in inflammation, allergic responses, and protection against cancer. A white blood cell differential totals the number of each of the different types of white blood cells in a person’s sample of blood.
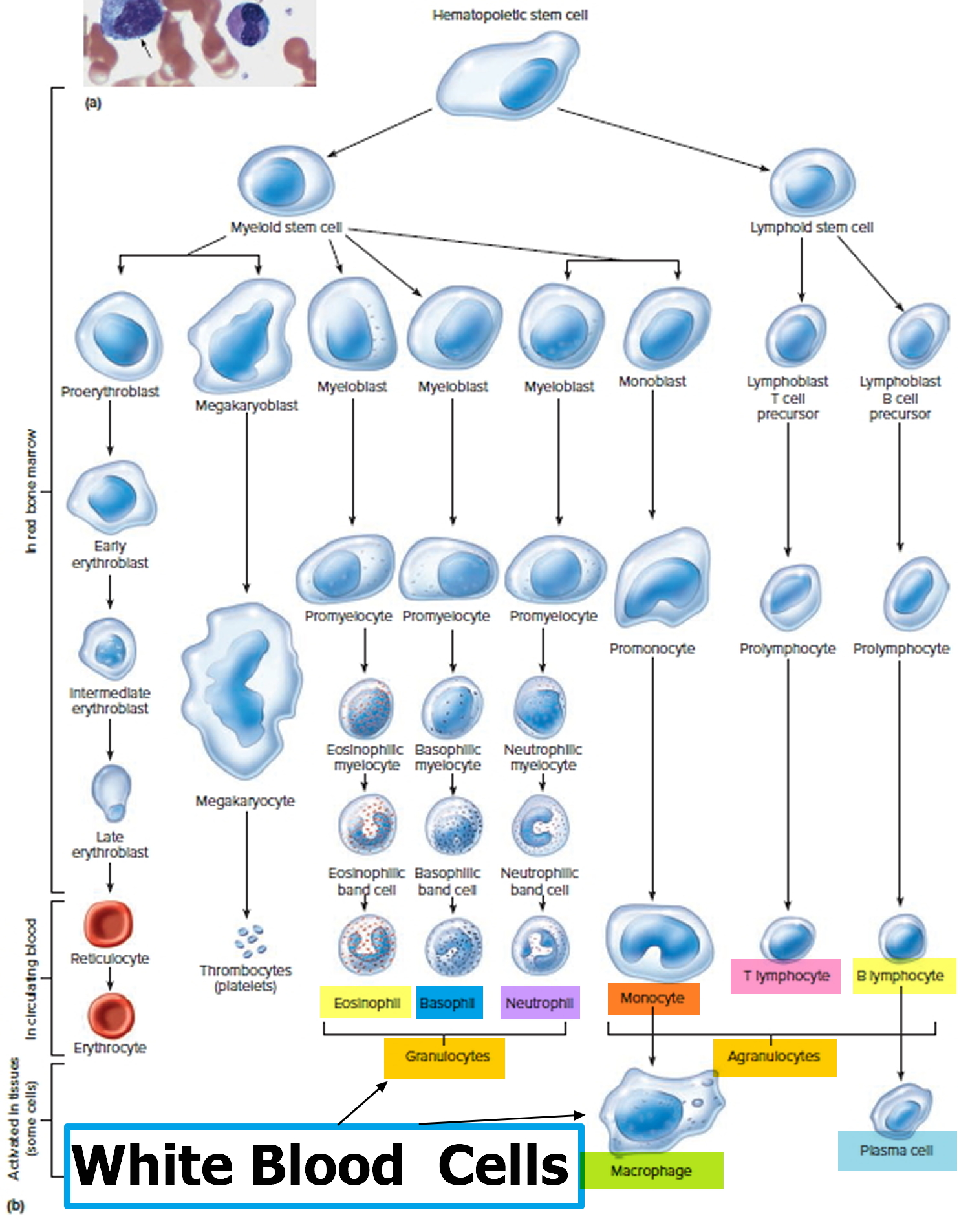
There are five types of white blood cells, each with different functions. The differential reveals if the cells are present in normal proportion to one another, if the number of one cell type is increased or decreased, or if abnormal and/or immature cells are present. This information is helpful in diagnosing specific types of illnesses that affect the immune system and the bone marrow.
A differential may be performed in conjunction with a complete blood count (CBC), a test often used as a general health check, or it may be performed in follow-up to abnormal results on a complete blood count (CBC). Most often, a differential is performed on an automated blood analyzer but also may be performed manually by a trained laboratorian who examines a blood smear under a microscope. The values are typically reported as absolute numbers of cells but may be expressed as the relative percentages of the total number of white blood cells.
White blood cells develop from precursor cells produced in the bone marrow.
The five different types of white blood cells include:
- Granulocytes—these white blood cells have granules in their cytoplasm. The granules contain chemicals and other substances that are released as part of an immune response. The three types of granulocytes include:
- Neutrophils (neu) normally make up the largest number of circulating white blood cells. They move into an area of damaged or infected tissue, where they engulf and destroy bacteria or sometimes fungi.
- Eosinophils (eos) respond to infections caused by parasites, play a role in allergic reactions (hypersensitivities), and control the extent of immune responses and inflammation.
- Basophils (baso) usually make up the fewest number of circulating white blood cells and are thought to be involved in allergic reactions.
- Lymphocytes (lymphs) exist in both the blood and the lymphatic system. They are divided into three types, but the differential does not distinguish among them. All lymphocytes differentiate from common lymphoid progenitor cells in the bone marrow. The differential counts and reports all lymphocytes together. Separate specialized testing (like immunophenotyping) must be done to differentiate the three types:
- B lymphocytes (B cells) are antibody-producing cells that are essential for acquired, antigen-specific immune responses. Plasma cells are fully differentiated B-cells that produce antibodies, immune proteins that target and destroy bacteria, viruses and other “non-self” foreign antigens.
- T lymphocytes (T cells) finish maturing in the thymus and consist of a few different types. Some T cells help the body distinguish between “self” and “non-self” antigens. Others initiate and control the extent of an immune response, boosting it as needed and then slowing it as the condition resolves. Other types of T cells directly attack and neutralize virus-infected or cancerous cells.
- Natural killer cells (NK cells) directly attack and kill abnormal cells such as cancer cells or those infected with a virus.
- Monocytes (mono), similar to neutrophils, move to an area of infection and engulf and destroy bacteria. They are associated more often with chronic rather than acute infections. They are also involved in tissue repair and other functions involving the immune system.
When there is an infection or an inflammatory process somewhere in the body, the bone marrow produces more white blood cells, releasing them into the blood. Depending on the cause of infection or inflammation, one particular type of white blood cell may be increased as opposed to other types. As the condition resolves, the production of that type of white blood cell subsides and the number drops to normal levels again.
In addition to infections and inflammation, there are a variety of conditions that can affect the production of white blood cells by the bone marrow or their survival in the blood, resulting in either increased or decreased numbers. The differential, along with the other components of the CBC, alerts the healthcare provider to possible health issues. Results are often interpreted in conjunction with additional tests such as a blood smear review, which can reveal the presence of abnormal and/or immature populations of white blood cells.
In a few serious diseases, some immature forms of the cells are released from the bone marrow into the circulation and may be detected by the white blood cell differential. This may occur with bacterial infection, leukemia, bone marrow involvement by solid tumor, myelodysplastic syndrome, or myeloproliferative neoplasms, for example. Some immature cells that may be detected include metamyelocytes, myelocytes, promyelocytes, and/or blasts.
If results indicate a problem, a wide variety of other tests may be performed in order to help determine the cause. A healthcare provider will typically consider an individual’s signs and symptoms, medical history, and results of a physical examination to decide what other tests may be necessary. For example, as needed, a bone marrow biopsy will be performed to evaluate the bone marrow status.
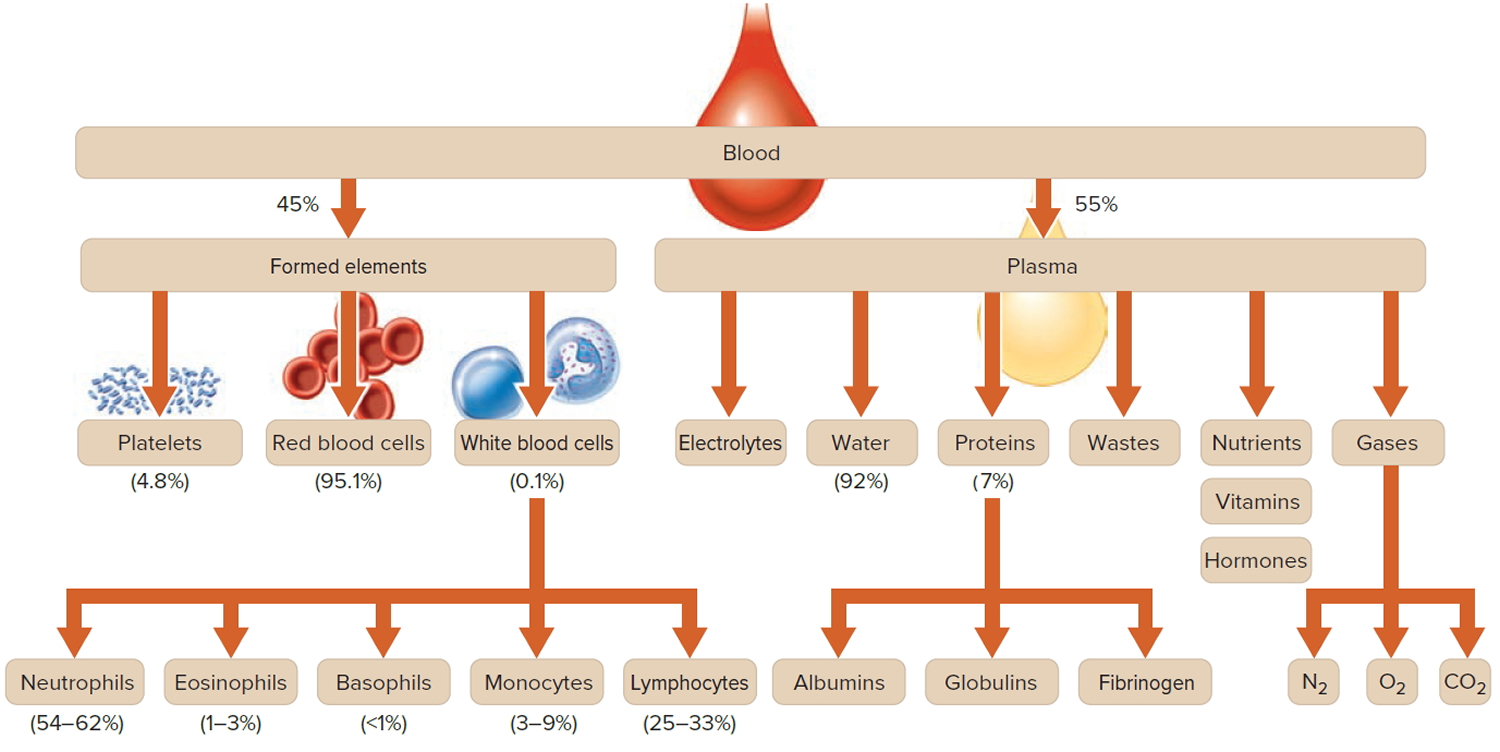
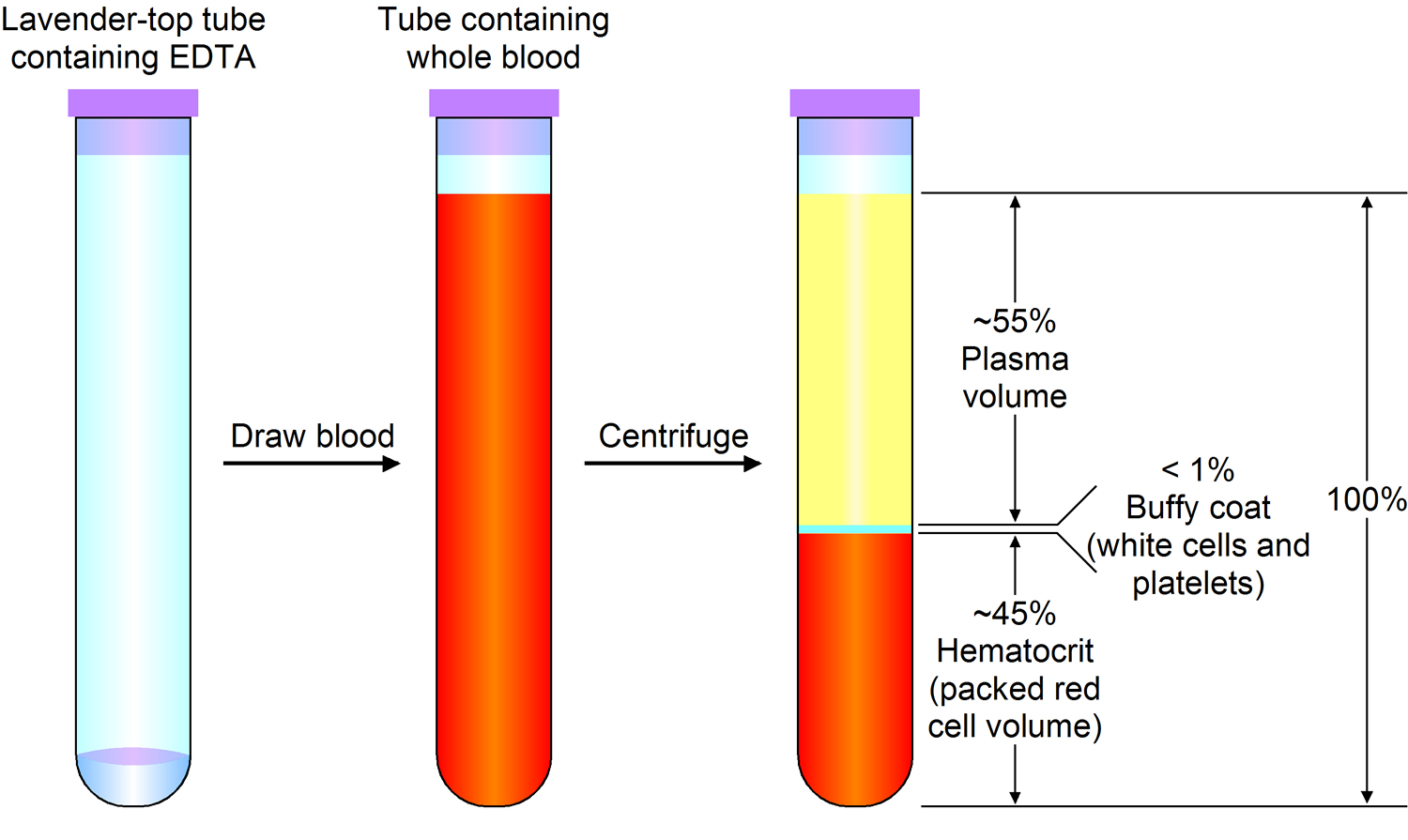
Figure 3. Blood composition
Note: Blood is a complex mixture of formed elements in a liquid extracellular matrix, called blood plasma. Note that water and proteins account for 99% of the blood plasma.
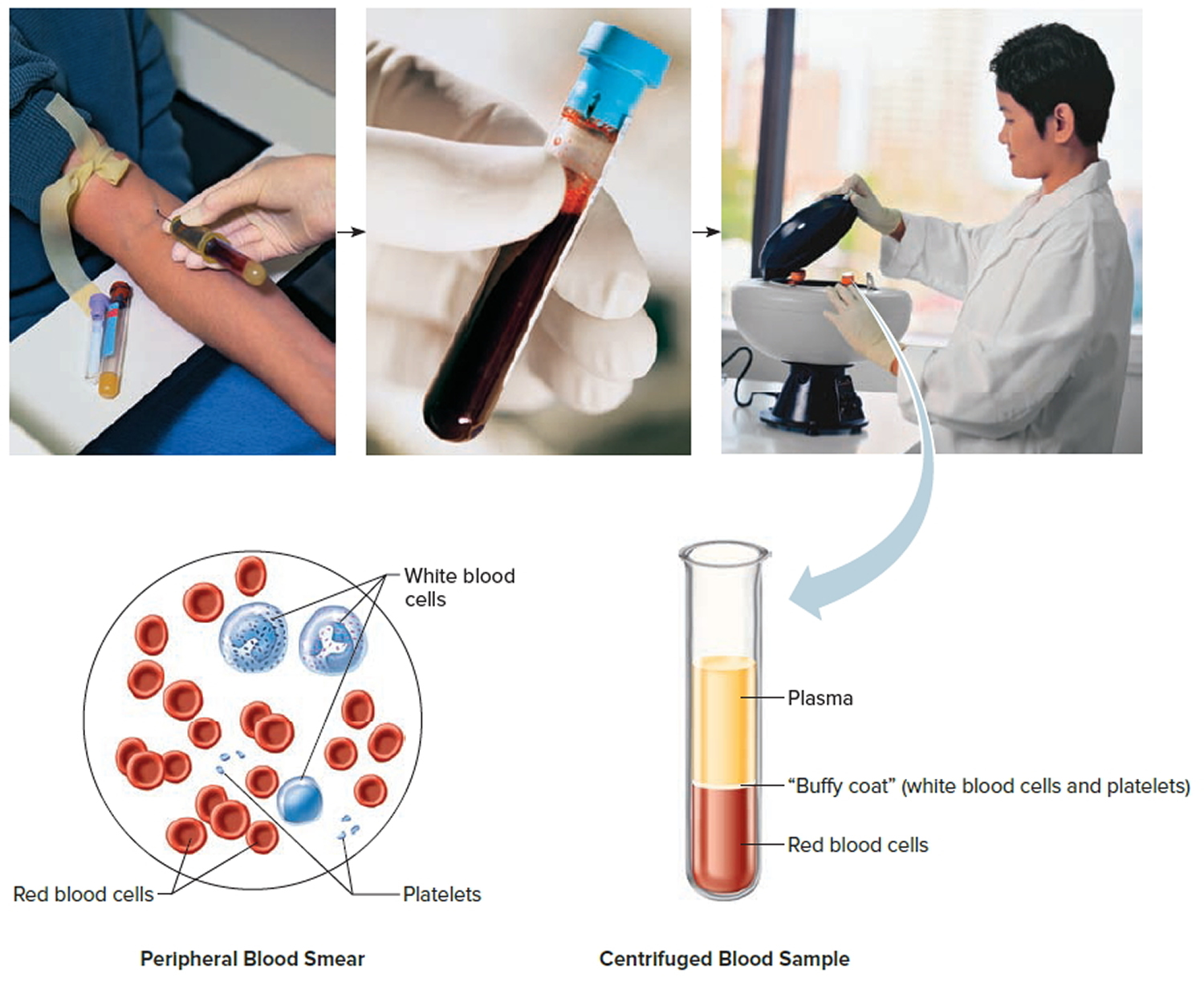
Note: Blood consists of a liquid portion called plasma and a solid portion (the formed elements) that includes red blood cells, white blood cells, and platelets. When blood components are separated by centrifugation, the white blood cells and platelets form a thin layer, called the “buffy coat,” between the plasma and the red blood cells, which accounts for about 1% of the total blood volume. Blood cells and platelets can be seen under a light microscope when a blood sample is smeared onto a glass slide.
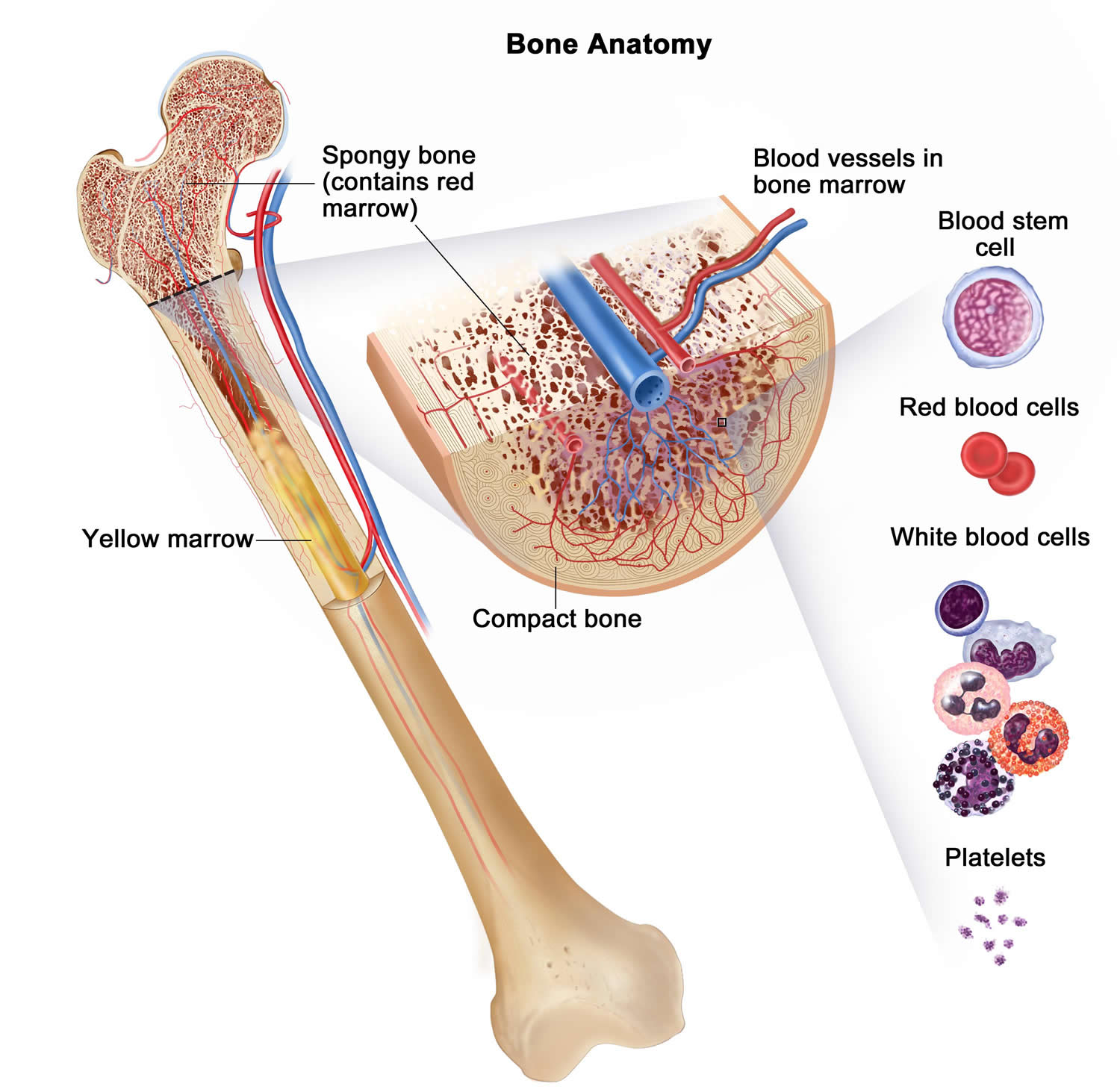
Figure 4. Bone marrow anatomy
Anatomy of the bone. The bone is made up of compact bone, spongy bone, and bone marrow. Compact bone makes up the outer layer of the bone. Spongy bone is found mostly at the ends of bones and contains red marrow. Bone marrow is found in the center of most bones and has many blood vessels. There are two types of bone marrow: red and yellow. Red marrow contains blood stem cells that can become red blood cells, white blood cells, or platelets. Yellow marrow is made mostly of fat.
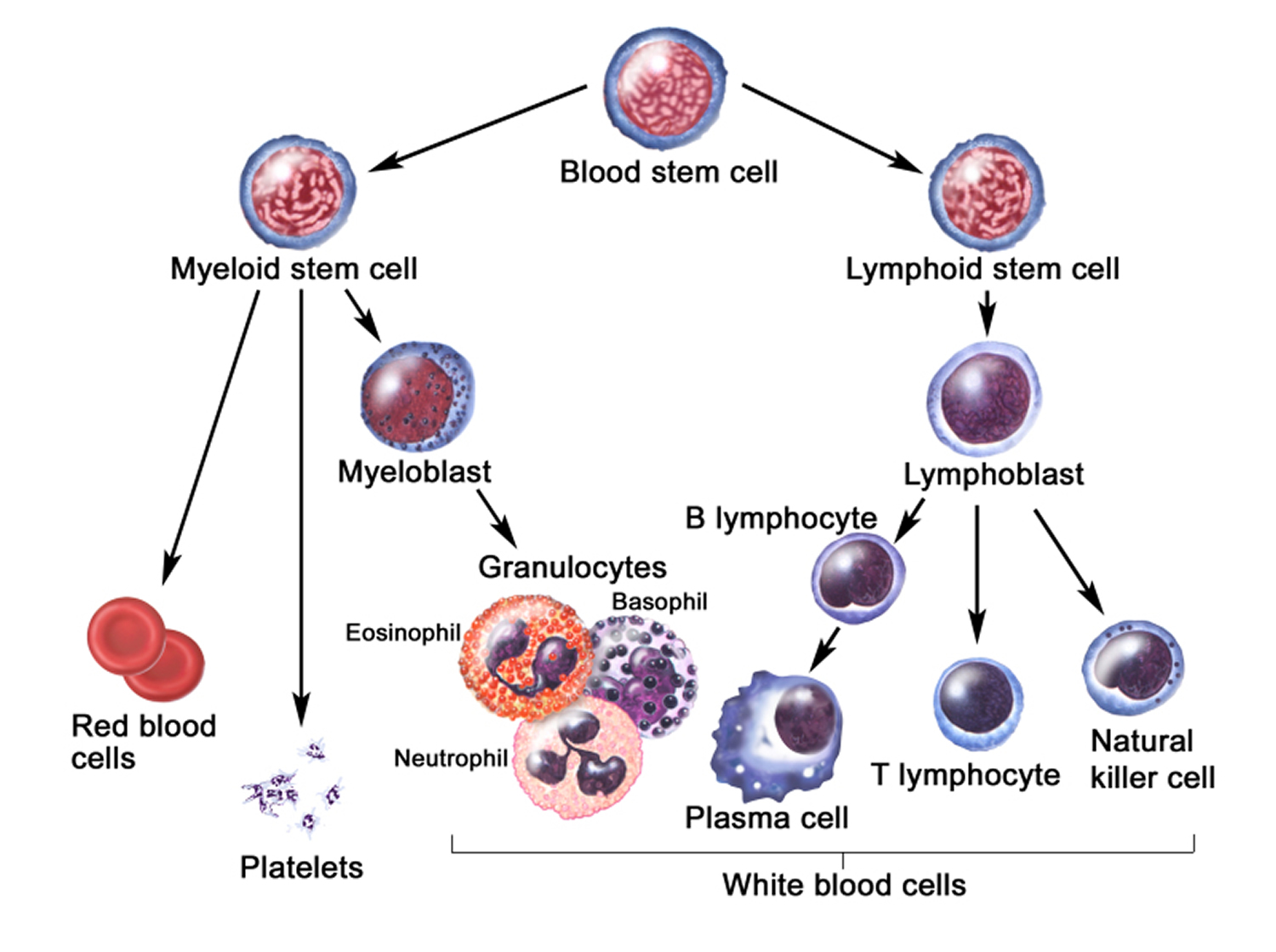
Figure 5. White blood cells development. A blood stem cell goes through several steps to become a red blood cell, platelet, or white blood cell
Figure 6. White blood cells development
Figure 7. White blood cells
What does it mean when your monocytes are high?
Increased monocytes count (> 1 × 109/L) also known as monocytosis can occurs in chronic infection such as tuberculosis, endocarditis, granulomatous disease, or in myeloproliferative disorders 29. Other disorders that can be associated with increased monocyte counts are the metabolic syndrome 30 such as obesity 31 and diabetes 32 and autoimmune disorders including rheumatoid arthritis 33. The underlying pathophysiology leading to monocytosis is not fully understood. Chemokines such as monocyte chemoattractant protein-1 and growth factors trigger monocyte recruitment and homeostasis 34. Smoking also leads to increased monocyte numbers 35.
Blood monocytosis enables eradication of infection and removal of cellular debris after injury and ischemia 36. Monocyte-derived macrophages may also support resolution of inflammation, if their phenotype permits. However, exaggerated monocytosis during inflammation likely harms tissues by limiting resolution of inflammation and propagating exaggerated immune activation, for instance in sepsis.
Monocytosis is associated with artherosclerosis and its consequences such as coronary artery disease, cerebrovascular disease, or kidney artery stenosis, for example, as a source of foam cells 37. Increased monocyte counts after acute myocardial infarction (heart attack) were associated with left ventricular dysfunction, left ventricular aneurysm, and other cardiac events 38. Another study showed similar effects to the nonrecovery of the left ventricular function after reperfused acute myocardial infarction (heart attack) 39. To this end, monocytosis has been identified as an independent risk factor for myocardial infarction or cerebral arterial disease 40. The level of the National Institutes of Health Stroke Scale on stroke patients correlates with the amount of monocytes 41.
Despite the profound knowledge in monocyte biology, surprisingly little is known about monocytosis in the clinical setting. In this large survey, the scientists identified peripheral blood monocytosis as a negative prognostic marker in the emergency setting 29. This is in line with a plethora of previous studies showing that activation of the innate immune system may be detrimentally associated with critical illness 42.
Monocytes are a major source of oxidative stress and thus can trigger organ damage under certain circumstances 43. Patients with monocytosis had more often respiratory symptoms and suffered from infection than individuals with normal monocyte counts. In part this might be related to the higher number of COPD (chronic obstructive pulmonary disease) patients in this group and indicates that smoking, triggers monocytosis. It can however be postulated that lung impairment, most likely due to infection, is a main stimulator of monocytosis. Fever, which was also associated with monocytosis in this study, further indicates that a potentially unspecific systemic inflammatory response is involved in monocytosis. Why neurologic diagnosis inversely correlated with monocytosis is unclear and surprising. Prior studies have shown an association between monocytes and cerebral vascular disease 44. Potentially, patrolling monocytes at the inner side of the vessel wall behave differently in blood–brain barrier than in the rest of the circulation.
In contrast, cardiovascular diagnoses were the strongest influence for the 30-day mortality in patients with monocytosis 29. This is in line with previous studies showing that monocytosis is also involved in the pathogenesis of atherosclerosis. Apart from the brain, scientists postulate that monocytosis is notably toxic to organs affected from atherosclerosis, for example, by increased extravasation or release of cytokines and oxidative stress 29. Monocytopenia was also associated with an increased 30-mortality in this survey 29, but this mainly affected hematological disorders and was not influenced by cardiovascular diagnoses. There was no negative effect of monocytosis on functional outcomes such as mobility or pain.
Monocytosis can be therapeutically regulated at different stages: monocyte production in the hematopoietic niche, release from the bone marrow and spleen, recruitment to sites of inflammation and/or macrophage polarization. Stem cell factor 45, IL-1β 46, IL-3, and G-MCSF 47 are candidate targets for reducing monocytosis following heart attack and in atherosclerosis. MCP-1, CCR2 and angiotensin-II receptor 1 may be blocked to reduce monocyte release from bone marrow and splenic niches. siRNA or small molecule inhibitors that target adhesion molecules such as VCAM-1, ICAMs, E-selectins, and chemokines such as MCP-1, CX3CL1, and CCL5 can reduce monocyte recruitment to inflammatory sites. Tissue healing and resolution of inflammation require balanced pro- and anti-inflammatory responses. Monocytes and macrophages secrete pro-inflammatory cytokines that can render atherosclerotic plaques vulnerable to rupture, promote heart failure and support insulin resistance in type II diabetic patients. On the other hand, alternatively activated macrophages stimulate tissue repair by encouraging collagen production, bolster angiogenesis and secrete anti-inflammatory cytokines that can increase insulin sensitivity in lean animals. This emerging complexity of the innate immune system implies that scientists must develop therapies that precisely target the right cells at the right time to maximize benefits and limit collateral damage.
Monocytes low
Monocytopenia (mean, 13.3 cells x 106/L; median, 14.5 cells x 106/L) is a form of leukocytopenia associated with a deficiency of monocytes 48. The major causes of this condition include use of myelotoxic drugs (chemotherapy), acute infectious stress, aplastic anemia, hairy cell leukemia and myeloid leukemia 49.
References- Differentiation of monocytes. Origin, nature, and fate of their azurophil granules. Nichols BA, Bainton DF, Farquhar MG. J Cell Biol. 1971 Aug; 50(2):498-515. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2108281/pdf/498.pdf
- Van Furth R. Phagocytic Cells: Development and Distribution of Mononuclear Phagocytes in Normal Steady State and Inflammation. In: Gallin JI, Goldstein IM, Snyderman R, editors. Inflammation: Basic Principles and Clinical Correlates. Raven Press, Ltd; 1988. pp. 281–295.
- Randolph GJ, Jakubzick C, Qu C. Antigen Presentation by Monocytes and Monocyte-derived Cells. Current opinion in immunology. 2008;20:52–60.
- Randolph GJ, Jakubzick C, Qu C. Antigen Presentation by Monocytes and Monocyte-derived Cells. Current opinion in immunology. 2008;20(1):52-60. doi:10.1016/j.coi.2007.10.010. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2408874/
- Monocyte-mediated defense against bacteria, fungi, and parasites. Lauvau G, Loke P, Hohl TM. Semin Immunol. 2015 Dec; 27(6):397-409.
- Cell biology of antigen processing in vitro and in vivo. Trombetta ES, Mellman I. Annu Rev Immunol. 2005; 23:975-1028.
- Monocytes give rise to mucosal, but not splenic, conventional dendritic cells. Varol C, Landsman L, Fogg DK, Greenshtein L, Gildor B, Margalit R, Kalchenko V, Geissmann F, Jung S. J Exp Med. 2007 Jan 22; 204(1):171-80.
- Monocyte-mediated defense against microbial pathogens. Serbina NV, Jia T, Hohl TM, Pamer EG. Annu Rev Immunol. 2008; 26:421-52.
- Corliss BA, Azimi MS, Munson J, Peirce SM, Murfee WL. Macrophages: An Inflammatory Link between Angiogenesis and Lymphangiogenesis. Microcirculation (New York, NY : 1994). 2016;23(2):95-121. doi:10.1111/micc.12259. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4744134/
- Varol C, Landsman L, Fogg DK, Greenshtein L, Gildor B, Margalit R, Kalchenko V, Geissmann F, Jung S. Monocytes give rise to mucosal, but not splenic, conventional dendritic cells. J Exp Med. 2007;204:171–180.
- Ancuta P, Autissier P, Wurcel A, Zaman T, Stone D, Gabuzda D. CD16+ monocyte-derived macrophages activate resting T cells for HIV infection by producing CCR3 and CCR4 ligands. J Immunol. 2006;176:5760–5771.
- Swirski FK, Libby P, Aikawa E, Alcaide P, Luscinskas FW, Weissleder R, Pittet MJ. Ly-6Chi monocytes dominate hypercholesterolemia-associated monocytosis and give rise to macrophages in atheromata. J Clin Invest. 2007;117:195–205.
- Arnold L, Henry A, Poron F, Baba-Amer Y, van Rooijen N, Plonquet A, Gherardi RK, Chazaud B. Inflammatory monocytes recruited after skeletal muscle injury switch into antiinflammatory macrophages to support myogenesis. J Exp Med. 2007;204:1057–1069.
- Landsman L, Varol C, Jung S. Distinct differentiation potential of blood monocyte subsets in the lung. J Immunol. 2007;178:2000–2007.
- Ziegler-Heitbrock L. The CD14+ CD16+ blood monocytes: their role in infection and inflammation. J Leukoc Biol. 2007;81:584–592.
- Qu C, Edwards EW, Tacke F, Angeli V, Llodra J, Sanchez-Schmitz G, Garin A, Haque NS, Peters W, van Rooijen N, et al. Role of CCR8 and other chemokine pathways in the migration of monocyte-derived dendritic cells to lymph nodes. J Exp Med. 2004;200:1231–1241
- Ziegler-Heitbrock L. The CD14+ CD16+ blood monocytes: their role in infection and inflammation. J Leukoc Biol. 2007;81:584–592
- Tacke F, Ginhoux F, Jakubzick C, van Rooijen N, Merad M, Randolph GJ. Immature monocytes acquire antigens from other cells in the bone marrow and present them to T cells after maturing in the periphery. J Exp Med. 2006;203:583–597
- Delamarre L, Pack M, Chang H, Mellman I, Trombetta ES. Differential lysosomal proteolysis in antigen-presenting cells determines antigen fate. Science. 2005;307:1630–1634.
- Schmid H, Sauerbrei R, Schwarz G, Weber E, Kalbacher H, Driessen C. Modulation of the endosomal and lysosomal distribution of cathepsins B, L and S in human monocytes/macrophages. Biol Chem. 2002;383:1277–1283
- Bonasio R, Scimone ML, Schaerli P, Grabie N, Lichtman AH, von Andrian UH. Clonal deletion of thymocytes by circulating dendritic cells homing to the thymus. Nat Immunol. 2006;7:1092–1100.
- Mathis D, Benoist C. Back to central tolerance. Immunity. 2004;20:509–516.
- Steinman RM, Hemmi H. Dendritic cells: translating innate to adaptive immunity. Curr Top Microbiol Immunol. 2006;311:17–58.
- Naik SH, Metcalf D, van Nieuwenhuijze A, Wicks I, Wu L, O’Keeffe M, Shortman K. Intrasplenic steady-state dendritic cell precursors that are distinct from monocytes. Nat Immunol. 2006;7:663–671.
- Shortman K, Liu YJ. Mouse and human dendritic cell subtypes. Nat Rev Immunol. 2002;2:151–161.
- Liu K, Waskow C, Liu X, Yao K, Hoh J, Nussenzweig M. Origin of dendritic cells in peripheral lymphoid organs of mice. Nat Immunol. 2007;8:578–583.
- Balazs M, Martin F, Zhou T, Kearney J. Blood dendritic cells interact with splenic marginal zone B cells to initiate T-independent immune responses. Immunity. 2002;17:341–352.
- Randolph GJ, Beaulieu S, Lebecque S, Steinman RM, Muller WA. Differentiation of monocytes into dendritic cells in a model of transendothelial trafficking. Science. 1998;282:480–483.
- Hensel M, Grädel L, Kutz A, et al. Peripheral monocytosis as a predictive factor for adverse outcome in the emergency department: Survey based on a register study. Rosenthal. KS, ed. Medicine. 2017;96(28):e7404. doi:10.1097/MD.0000000000007404. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5515748/
- Dutta P, Nahrendorf M. Regulation and consequences of monocytosis. Immunol Rev 2014;262:167–78.
- Adipose tissue macrophages promote myelopoiesis and monocytosis in obesity. Nagareddy PR, Kraakman M, Masters SL, Stirzaker RA, Gorman DJ, Grant RW, Dragoljevic D, Hong ES, Abdel-Latif A, Smyth SS, Choi SH, Korner J, Bornfeldt KE, Fisher EA, Dixit VD, Tall AR, Goldberg IJ, Murphy AJ. Cell Metab. 2014 May 6; 19(5):821-35.
- Hyperglycemia promotes myelopoiesis and impairs the resolution of atherosclerosis. Nagareddy PR, Murphy AJ, Stirzaker RA, Hu Y, Yu S, Miller RG, Ramkhelawon B, Distel E, Westerterp M, Huang LS, Schmidt AM, Orchard TJ, Fisher EA, Tall AR, Goldberg IJ. Cell Metab. 2013 May 7; 17(5):695-708.
- Klimek E, Mikolajczyk T, Sulicka J, et al. Blood monocyte subsets and selected cardiovascular risk markers in rheumatoid arthritis of short duration in relation to disease activity. Biomed Res Int 2014;2014:736853
- Deshmane SL, Kremlev S, Amini S, et al. Monocyte chemoattractant protein-1 (MCP-1): an overview. J Interf Cytokine Res 2009;29:313–26.
- Corre F, Lellouch J, Schwartz D. Smoking and leucocyte-counts. Lancet 1971;2:632–4.
- Dutta P, Nahrendorf M. Regulation and consequences of monocytosis. Immunological reviews. 2014;262(1):167-178. doi:10.1111/imr.12219. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4203415/
- Woollard KJ, Geissmann F. Monocytes in atherosclerosis: subsets and functions. Nat Rev Cardiol 2010;7:77–86.
- Maekawa Y, Anzai T, Yoshikawa T, et al. Prognostic significance of peripheral monocytosis after reperfused acute myocardial infarction:a possible role for left ventricular remodeling. J Am Coll Cardiol 2002;39:241–6.
- Hong YJ, Jeong MH, Ahn Y, et al. Relationship between peripheral monocytosis and nonrecovery of left ventricular function in patients. Circ J 2007;71:1219–24.
- Abrahão Afiune Neto, Antonio de Pádua Mansur SDA, Everly PSG, et al. Monocytosis is an independent risk marker for coronary artery disease. Arq Bras Cardiol 2006;86:240–4.
- Kaito M, Araya SI, Gondo Y, et al. Relevance of distinct monocyte subsets to clinical course of ischemic stroke patients. PLoS One 2013;8:e69409.
- Host innate immune responses to sepsis. Wiersinga WJ, Leopold SJ, Cranendonk DR, van der Poll T. Virulence. 2014 Jan 1; 5(1):36-44.
- Monocyte and macrophage heterogeneity in the heart. Nahrendorf M, Swirski FK. Circ Res. 2013 Jun 7; 112(12):1624-33.
- Relevance of distinct monocyte subsets to clinical course of ischemic stroke patients. Kaito M, Araya S, Gondo Y, Fujita M, Minato N, Nakanishi M, Matsui M. PLoS One. 2013; 8(8):e69409.
- Myocardial infarction accelerates atherosclerosis. Dutta P, Courties G, Wei Y, Leuschner F, Gorbatov R, Robbins CS, Iwamoto Y, Thompson B, Carlson AL, Heidt T, Majmudar MD, Lasitschka F, Etzrodt M, Waterman P, Waring MT, Chicoine AT, van der Laan AM, Niessen HW, Piek JJ, Rubin BB, Butany J, Stone JR, Katus HA, Murphy SA, Morrow DA, Sabatine MS, Vinegoni C, Moskowitz MA, Pittet MJ, Libby P, Lin CP, Swirski FK, Weissleder R, Nahrendorf M. Nature. 2012 Jul 19; 487(7407):325-9.
- Rapid monocyte kinetics in acute myocardial infarction are sustained by extramedullary monocytopoiesis. Leuschner F, Rauch PJ, Ueno T, Gorbatov R, Marinelli B, Lee WW, Dutta P, Wei Y, Robbins C, Iwamoto Y, Sena B, Chudnovskiy A, Panizzi P, Keliher E, Higgins JM, Libby P, Moskowitz MA, Pittet MJ, Swirski FK, Weissleder R, Nahrendorf M. J Exp Med. 2012 Jan 16; 209(1):123-37.
- Extramedullary hematopoiesis generates Ly-6C(high) monocytes that infiltrate atherosclerotic lesions. Robbins CS, Chudnovskiy A, Rauch PJ, Figueiredo JL, Iwamoto Y, Gorbatov R, Etzrodt M, Weber GF, Ueno T, van Rooijen N, Mulligan-Kehoe MJ, Libby P, Nahrendorf M, Pittet MJ, Weissleder R, Swirski FK. Circulation. 2012 Jan 17; 125(2):364-74.
- Nazir T, Taha N, Islam A, Abraham S, Mahmood A, Mustafa M. Monocytopenia; Induction by Vinorelbine, Cisplatin and Doxorubicin in Breast, Non-Small Cell Lung and Cervix Cancer Patients. International Journal of Health Sciences. 2016;10(4):542-547. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5085349/
- Which one is a risk factor for chemotherapy-induced febrile neutropenia in childhood solid tumors: early lymphopenia or monocytopenia? Oguz A, Karadeniz C, Ckitak EC, Cil V. Pediatr Hematol Oncol. 2006 Mar; 23(2):143-51