What is pseudotumor cerebri
Pseudotumor cerebri also known as idiopathic intracranial hypertension or benign intracranial hypertension, is a condition that affects the brain characterized by signs and symptoms of increased intracranial pressure in the absence of a space-occupying lesion 1. The name of pseudotumor cerebri literally means “false brain tumor.” Pseudotumor cerebri (idiopathic intracranial hypertension) is a disorder of unknown etiology that predominantly affects obese women of childbearing age 2. Pseudotumor cerebri is likely due to high pressure within the skull caused by the build-up of cerebrospinal fluid (CSF). Pseudotumor cerebri is most common in obese women between the ages of 20 and 50, but can also occur in children and about 5 percent of those affected are men.
The incidence of pseudotumor cerebri in the general population is thought to be about 1 per 100,000. In obese young females the incidence of pseudotumor cerebri is about 20 per 100,000. An estimated 100,000 Americans have pseudotumor cerebri and the number is rising as more people become obese or overweight. The disorder is most common in women between the ages of 20 and 50. Obesity, defined as a body mass index (BMI) greater than 30, is a major risk factor. BMI is a number based on your weight and height. A recent gain of 5-15 percent of total body weight is also considered a risk factor for this disorder, even for people with a BMI less than 30. However, weight is not usually a factor in men and in children under 10 years of age.
Obesity dramatically increases a young woman’s risk of pseudotumor cerebri. In fact, even in women who aren’t obese a moderate amount of weight gain may increase the risk. Losing extra pounds and maintaining a healthy weight may help reduce your chances of developing this potentially sight-stealing disorder.
Symptoms of pseudotumor cerebri mimic symptoms of large brain tumors, such as headache, nausea, vomiting, and pulsating sounds within the head (tinnitus). Pseudotumor cerebri may also cause a rare condition called empty sella syndrome.
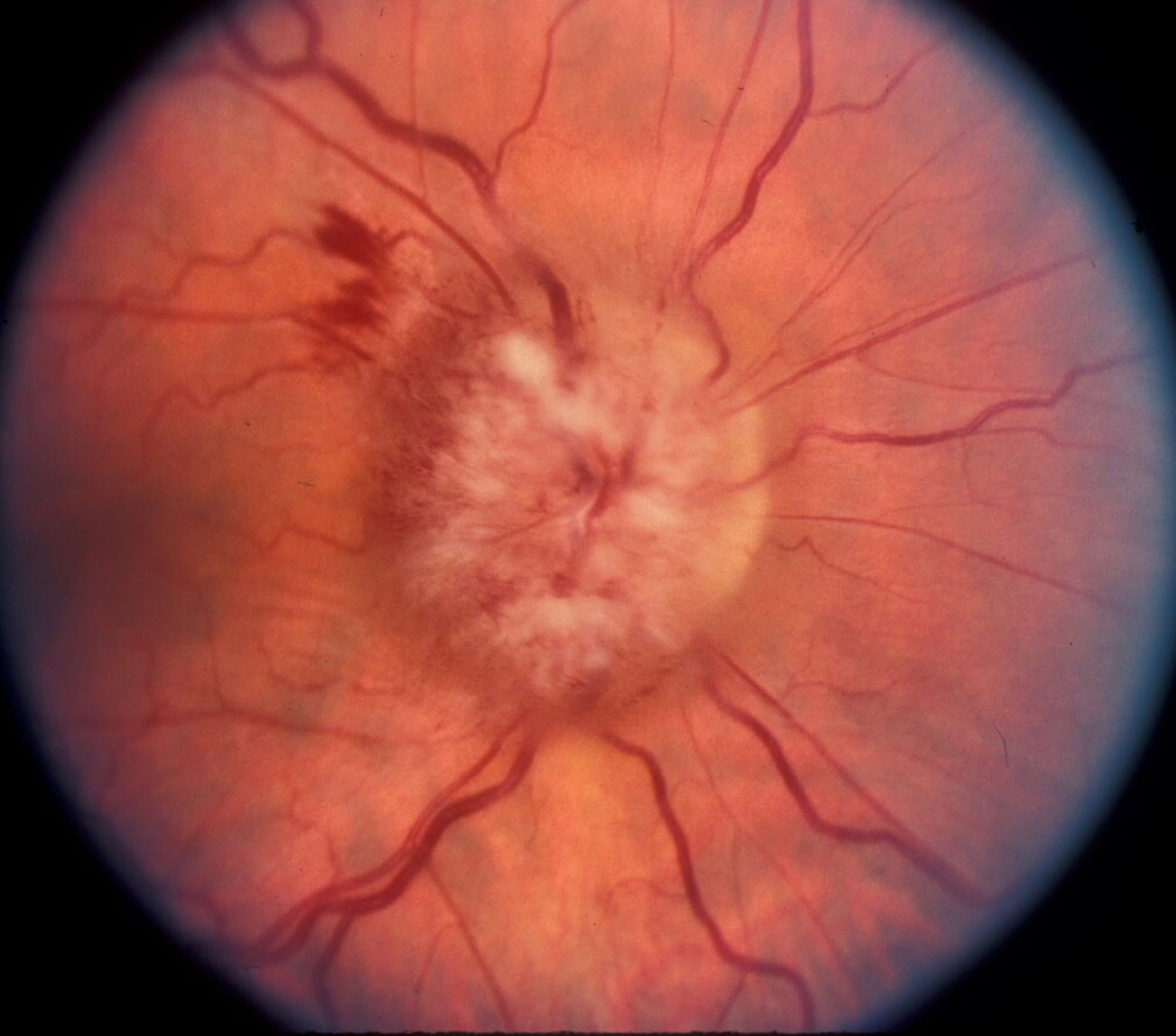
The increased intracranial pressure associated with pseudotumor cerebri can cause swelling of the optic nerve (papilledema) and result in vision loss, including blind spots, poor peripheral (side) vision, double vision, and short temporary episodes of blindness (see Figures 3 to 5). Many patients experience permanent vision loss. Medications often can reduce this pressure, but in some cases, surgery is necessary.
For most people, pseudotumor cerebri usually improves with treatment. For others, it progressively worsens with time, or it can resolve and then recur. About 5-10 percent of women with pseudotumor cerebri experience disabling vision loss.
Pseudotumor cerebri mirena
Because of a previous association of pseudotumor cerebri with levonorgestrel (mirena IUD), Valenzuela et al 3 evaluated the use of levonorgestrel-eluting intrauterine devices (“levonorgestrel intrauterine systems”, mirena) in the University of Utah and Rigshospitalet pseudotumor cerebri patients. In their retrospective series comparing the levonorgestrel intrauterine systems (mirena) population versus non-mirena population, they found no significant differences in pseudotumor cerebri signs and symptoms among the two groups 3. That study suggests that women with an levonorgestrel (mirena IUD) may have increased risk of developing pseudotumor cerebri but does not suggest levonorgestrel (mirena IUD) can cause pseudotumor cerebri.
Brain Ventricles and Cerebrospinal Fluid
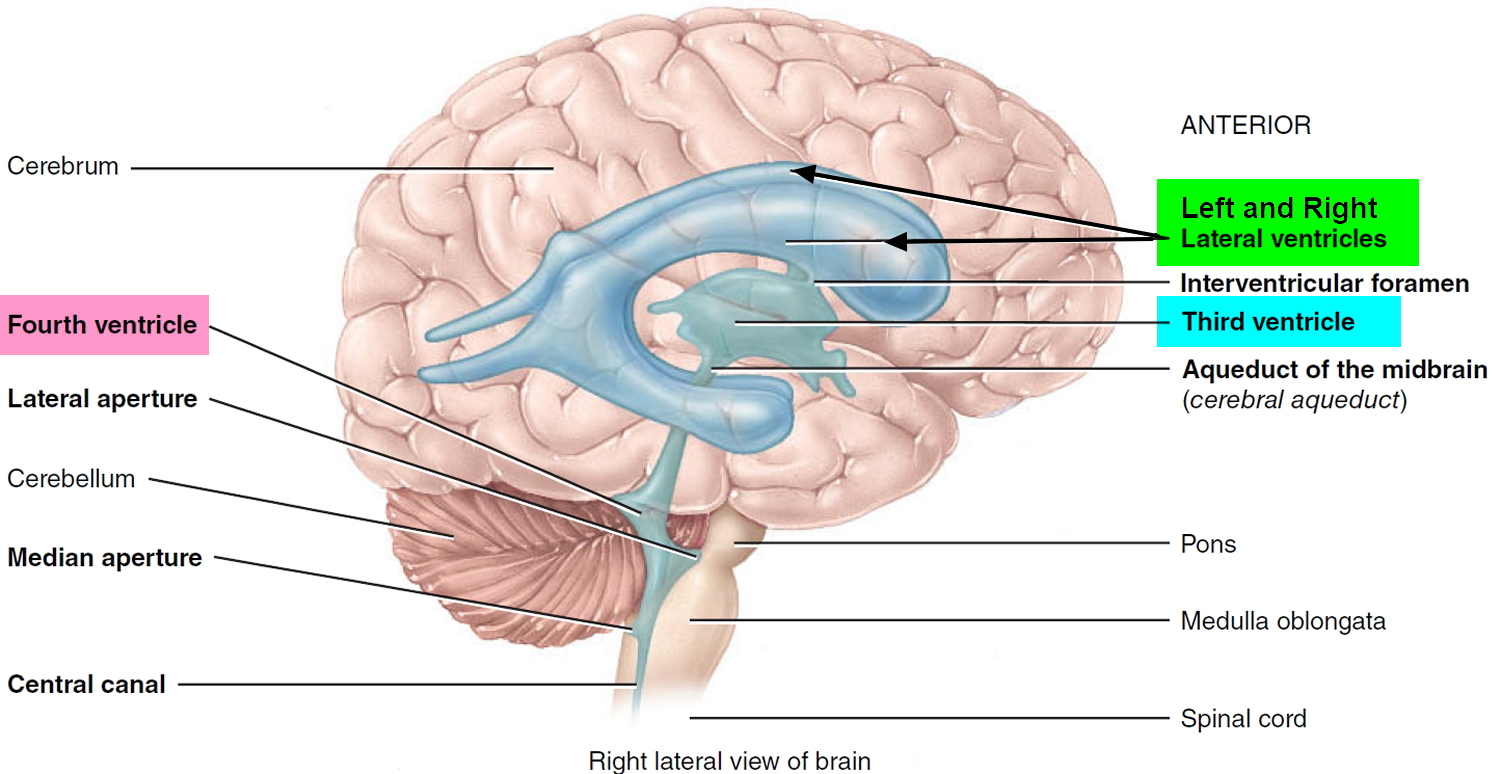
The brain has four internal chambers called ventricles. The largest and most frontal ones are the two lateral ventricles, which form an arc in each cerebral hemisphere. Through a tiny pore called the interventricular foramen, each lateral ventricle is connected to the third ventricle, a narrow median space inferior to the corpus callosum. From here, a canal called the cerebral aqueduct passes down the core of the midbrain and leads to the fourth ventricle, a small triangular chamber between the pons and cerebellum. Caudally, this space narrows and forms a central canal that extends through the medulla oblongata into the spinal cord.
Figure 1. Ventricles of the brain
On the floor or wall of each ventricle is a spongy mass of blood capillaries called a choroid plexus, named for its histological resemblance to a fetal membrane called the chorion. Ependyma, a type of neuroglia that resembles a cuboidal epithelium, lines the ventricles and canals and covers the choroid plexuses. It produces cerebrospinal fluid (CSF).
Cerebrospinal fluid is a clear, colorless liquid that fills the ventricles and canals of the CNS and bathes its external surface. The brain produces about 500 mL of cerebrospinal fluid per day, but the fluid is constantly reabsorbed at the same rate and only 100 to 160 mL is normally present at one time. About 40% of it is formed in the subarachnoid space external to the brain, 30% by the general ependymal lining of the brain ventricles, and 30% by the choroid plexuses. Cerebrospinal fluid production begins with the filtration of blood plasma through the capillaries of the brain. Ependymal cells modify the filtrate as it passes through them, so the cerebrospinal fluid has more sodium chloride than blood plasma, but less potassium, calcium, and glucose and very little protein.
Cerebrospinal fluid serves three functions for the brain:
- Buoyancy. Because the brain and cerebrospinal fluid are similar in density, the brain neither sinks nor floats in the cerebrospinal fluid. It hangs from delicate specialized fibroblasts of the arachnoid meninx. A human brain removed from the body weighs about 1.5 kg, but when suspended in cerebrospinal fluid its effective weight is only about 50 g. This buoyancy allows the brain to attain considerable size without being impaired by its own weight. If the brain rested heavily on the floor of the cranium, the pressure would kill the nervous tissue.
- Protection. Cerebrospinal fluid also protects the brain from striking the cranium when the head is jolted. If the jolt is severe, however, the brain still may strike the inside of the cranium or suffer shearing injury from contact with the angular surfaces of the cranial floor. This is one of the common findings in head injuries (concussions) from contact sports like NFL, rugby and boxing.
- Chemical stability. Cerebrospinal fluid rinses metabolic wastes from the nervous tissue and regulates its chemical environment. Slight changes in cerebrospinal fluid composition can cause malfunctions of the nervous system. For example, a high glycine concentration disrupts the control of body temperature and blood pressure, and a high pH causes dizziness and fainting.
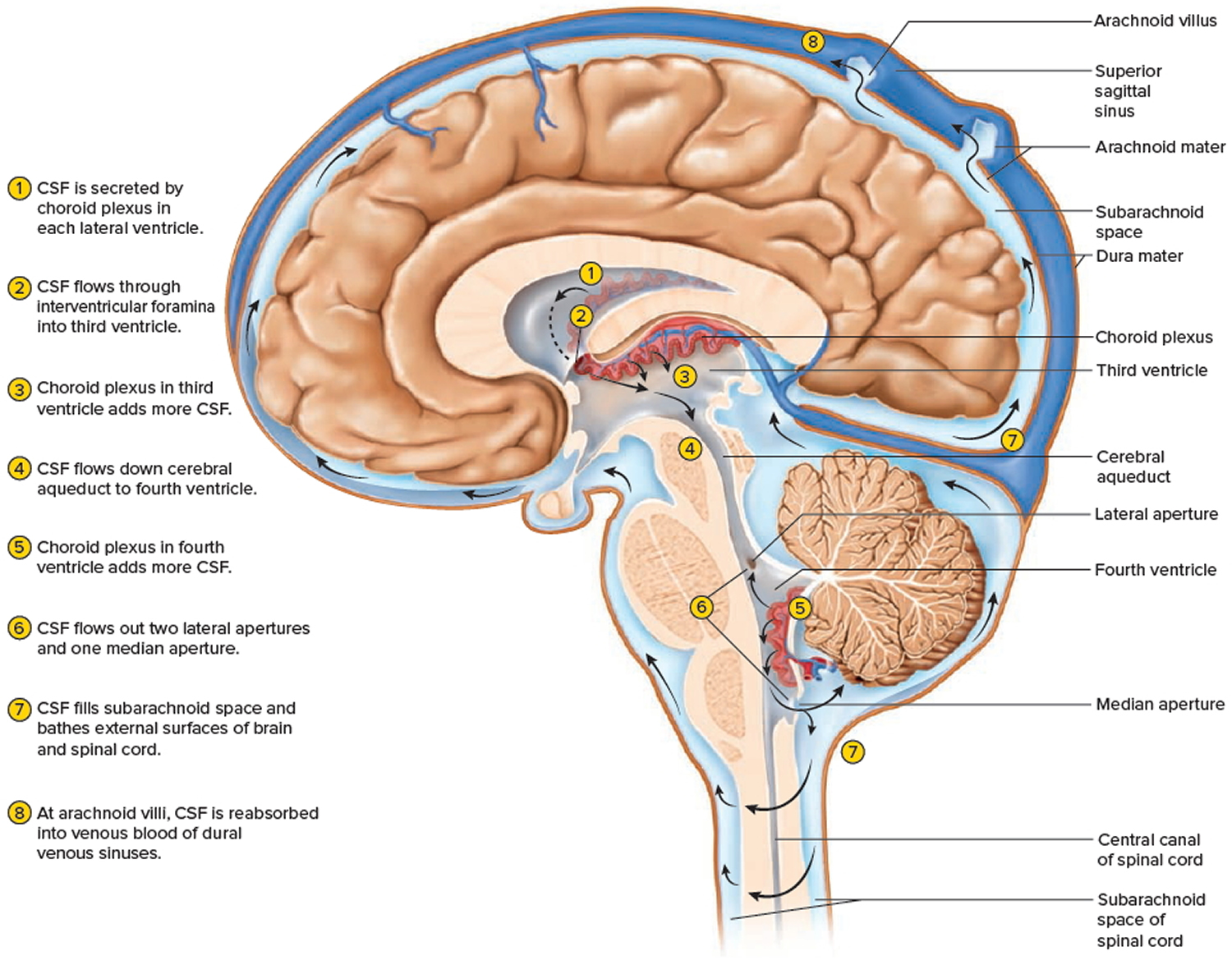
Cerebrospinal fluid continually flows through and around the brain and spinal cord, driven partly by its own pressure, partly by the beating of ependymal cilia, and partly by rhythmic pulsations of the brain produced by each heartbeat. The cerebrospinal fluid of the lateral ventricles flows through the interventricular foramina into the third ventricle, then down the cerebral aqueduct to the fourth ventricle. The third and fourth ventricles and their choroid plexuses add more cerebrospinal fluid along the way. A small amount of cerebrospinal fluid fills the central canal of the spinal cord, but ultimately, all of it escapes through three pores in the fourth ventricle—a median aperture and two lateral apertures. These lead into the subarachnoid space on the brain and spinal cord surface. From here, cerebrospinal fluid is reabsorbed by arachnoid granulations, extensions of the arachnoid meninx shaped like little sprigs of cauliflower, protruding through the dura mater into the superior sagittal sinus. Cerebrospinal fluid penetrates the walls of the granulations and mixes with blood in the sinus.
Figure 2. Cerebrospinal fluid formation, absorption and circulation around and within the brain
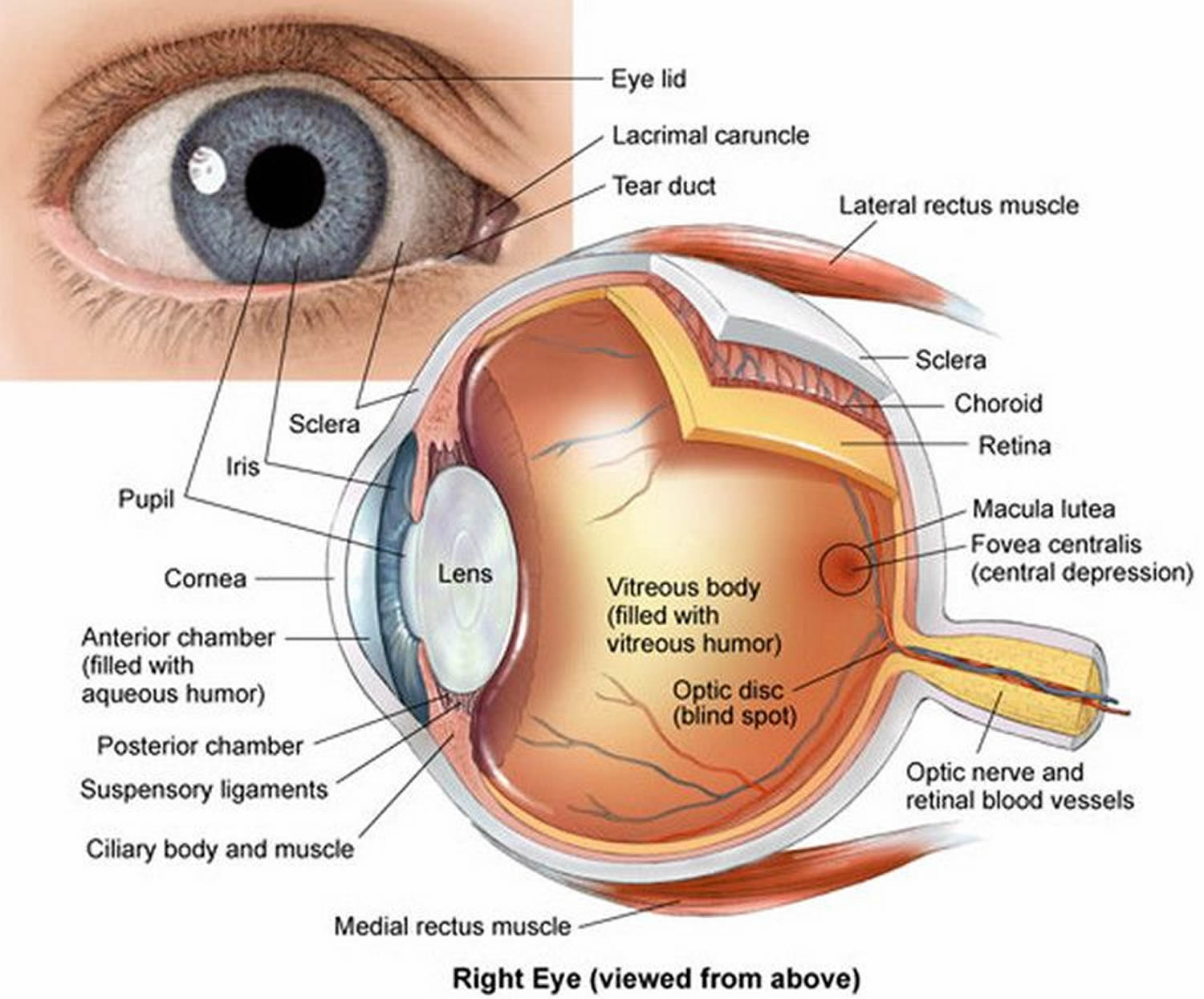
Figure 3. Structure of the human eye
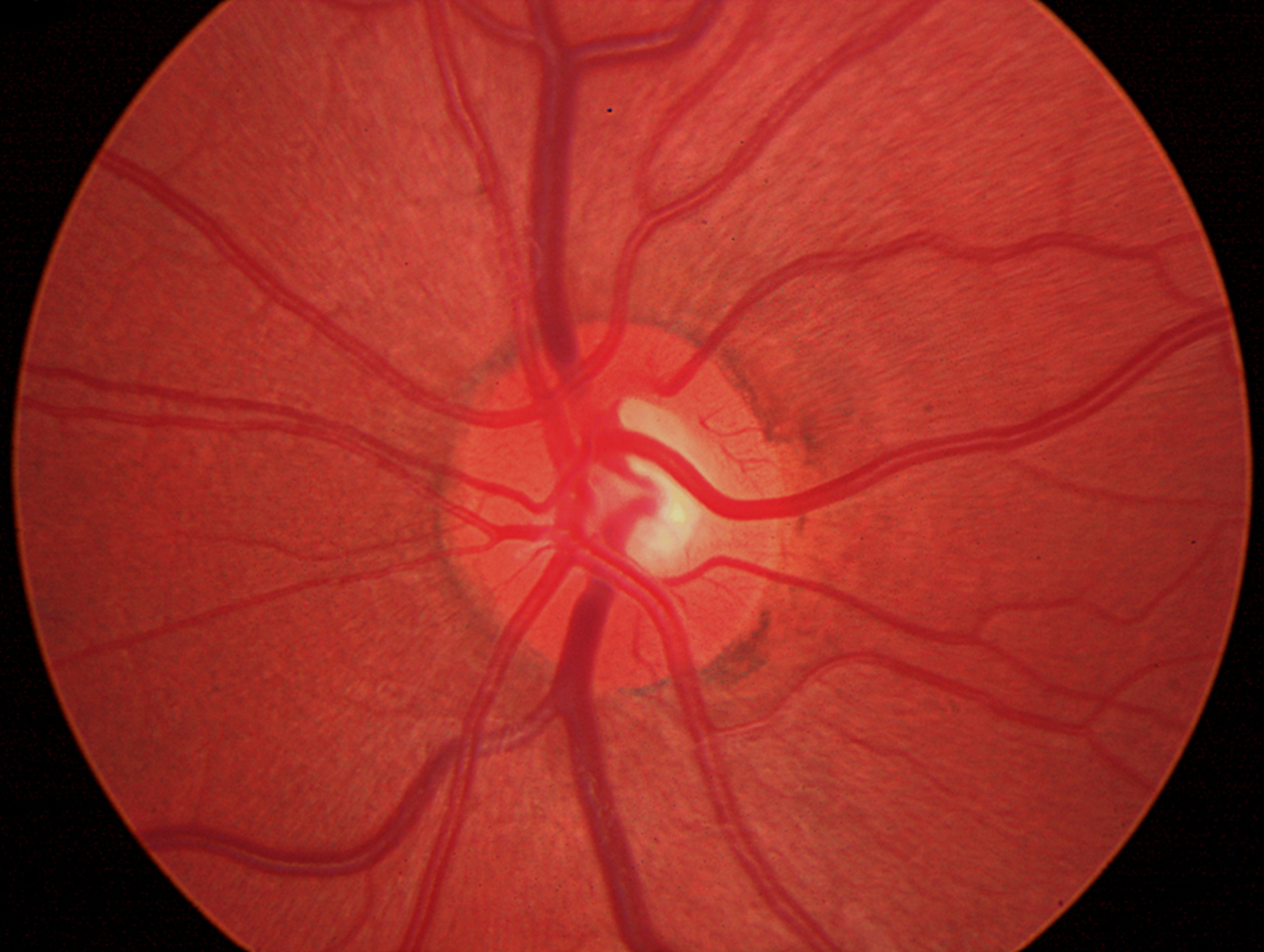
Figure 4. Normal optic disc (human eye)
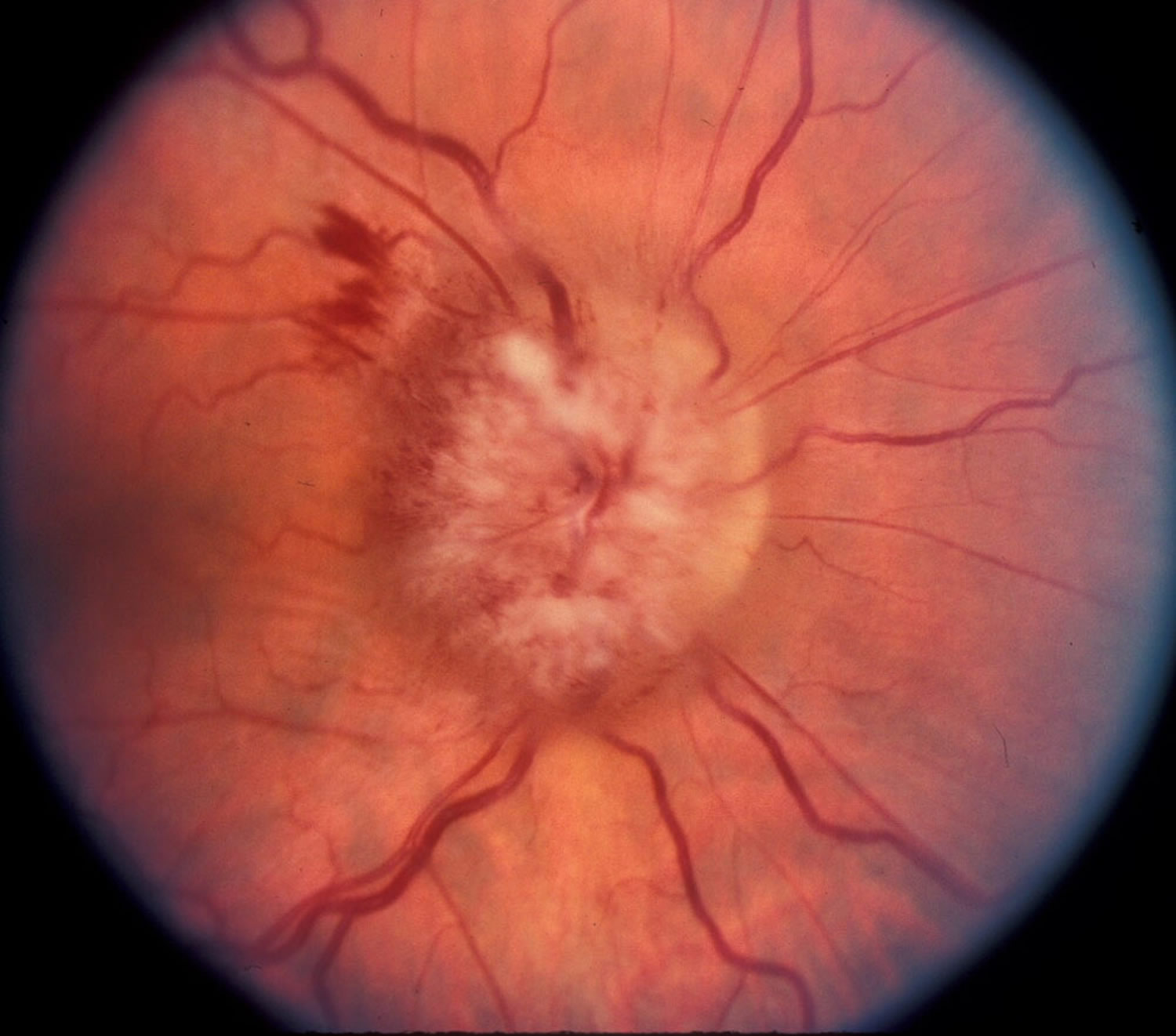
Figure 5. Papilledema – optic disc with papilledema in a patient with pseudotumor cerebri (idiopathic intracranial hypertension).
Pseudotumor cerebri prognosis
The outlook (prognosis) of this condition is quite variable: in some cases, the condition disappears on its own; in up to 20% of affected individuals, symptoms may return; and a small number of patients have symptoms that slowly get worse and may lead to blindness 4. It has been shown that regaining weight that was previously lost is associated with a return of symptoms 5.
The main concern with pseudotumor cerebri is the risk of worsening vision, so treatment aims to monitor and protect vision 6. Because of this, individuals with pseudotumor cerebri are encouraged to have regular eye examinations to check for changes in vision. Additionally, individuals who have undergone shunting procedures are recommended to visit a neurologist or neurosurgeon regularly to ensure that the shunt continues to work properly 6. More than half of individuals who have undergone this type of procedure experience occasional headaches following the procedure 7.
Pseudotumor cerebri complications
Some people with pseudotumor cerebri experience progressively worsening vision and may eventually become blind.
Even if your symptoms have resolved, they can recur months or even years later.
Pseudotumor cerebri causes
The exact cause of pseudotumor cerebri in most individuals is unknown, but it may be linked to an excess amount of cerebrospinal fluid within the bony confines of your skull.
Your brain and spinal cord are surrounded by cerebrospinal fluid, which acts like a cushion to protect these vital tissues from injury. This fluid is produced in the brain and eventually is absorbed into the bloodstream. The increased intracranial pressure of pseudotumor cerebri may be a result of a problem in this absorption process.
In general, your intracranial pressure increases when the contents of your skull exceed its capacity. For example, a brain tumor generally increases your intracranial pressure because there’s no room for the tumor. The same thing happens if your brain swells or if you have too much cerebrospinal fluid.
Several studies indicate that many people with pseudotumor cerebri have a narrowing (stenosis) in two large sinuses in the brain (transverse sinuses). Studies are determining whether this is an effect or a cause of the condition.
Risk factors for pseudotumor cerebri
The following factors have been associated with pseudotumor cerebri:
Obesity
Obesity has been associated with pseudotumor cerebri, which occurs in 1 to 2 people in 100,000. In women who are obese, about 4 to 21 in 100,000 develop the condition. Women under the age of 44 who are obese are more likely to develop the disorder.
Medications
Substances that have been linked to pseudotumor cerebri include:
- Growth hormone
- Tetracycline
- Excess vitamin A
- Birth control pills
- Cyclosporine
- Isotretinoin
- Minocycline
- Nalidixic acid
- Nitrofurantoin
- Phenytoin
- Steroids (starting or stopping them)
- Sulfa drugs
- Tamoxifen
- Tetracycline
Other medications may be associated with pseudotumor cerebri, but further research is needed.
Health problems
The following conditions and diseases have been linked to pseudotumor cerebri:
- Addison’s disease
- Anemia
- Behcet’s syndrome
- Blood-clotting disorders
- Lupus
- Chronic kidney failure
- Cushing disease
- Hypoparathyroidism
- Iron deficiency anemia
- Obesity
- Onset of menstruation
- Pregnancy
- Polycystic ovary syndrome (PCOS)
- Sleep apnea
- Underactive parathyroid glands (hypoparathyroidism)
- Uremia.
Pseudotumor cerebri symptoms
Patients with pseudotumor cerebri usually present with symptoms related to increased intracranial pressure and papilledema.
Pseudotumor cerebri signs and symptoms may include:
- Headaches (typically nonspecific and varying in type, location, and frequency)
- Visual loss (typically visual field but rarely visual acuity loss)
- Double vision (diplopia) (typically horizontal due to nonlocalizing sixth nerve palsy but rarely vertical)
- Ringing in the ears that pulses in time with your heartbeat (pulsatile tinnitus)
- Radicular pain (typically in the arms, uncommon symptom)
- Moderate to severe headaches that may originate behind your eyes and worsen with eye movement
- Nausea, vomiting or dizziness
- Blurred or dimmed vision
- Brief episodes of blindness, lasting only a few seconds and affecting one or both eyes (visual obscurations)
- Difficulty seeing to the side
- Seeing light flashes (photopsia)
- Neck, shoulder or back pain
Rarely, patients presenting with increased intracranial pressure with related optic nerve edema may be asymptomatic.
Visual symptoms of papilledema may include the following:
- Transient visual obscurations, often predominantly or uniformly orthostatic
- Progressive loss of peripheral vision in one or both eyes (nerve fiber layer defects, enlargement of the blind spot)
- Blurring and distortion (ie, metamorphopsia) of central vision due to macular edema or optic neuropathy
- Sudden visual loss (e.g., fulminant idiopathic intracranial hypertension)
Nonspecific symptoms of pseudotumor cerebri may include dizziness, nausea, vomiting, photopsias, and retrobulbar pain 8.
The most significant physical finding is bilateral optic disc edema secondary to the increased intracranial pressure. Rarely, in more pronounced cases, macular involvement with subsequent edema and diminished central vision may be present. Sixth nerve palsy may occur as a nonlocalizing sign of increased intracranial pressure.
High-grade and atrophic papilledema in addition to subretinal hemorrhages are poor visual prognostic signs. Uncontrolled papilledema results in progressive peripheral visual field constriction or nerve fiber bundle defects.
Rarely, sudden loss of central vision results from an associated anterior ischemic optic neuropathy or a vascular occlusion.
Pseudotumor cerebri diagnosis
Obesity, other treatable diseases, and some medications can cause raised intracranial pressure and symptoms of pseudotumor cerebri. A thorough medical history and physical examination is needed to evaluate these factors in order to determine the best approach to treatment of this condition.
Eye exams
A doctor trained in eye conditions (ophthalmologist) may conduct an eye examination.
Visual function tests are the most important parts of the neurologic examination for diagnosing and monitoring patients with pseudotumor cerebri.
If pseudotumor cerebri is suspected, your eye doctor will look for a distinctive type of swelling affecting the optic nerve, called papilledema, in the back of your eye.
You’ll also undergo a visual fields test to see if there are any blind spots in your vision besides your so-called normal blind spot in each eye where the optic nerve enters the retina.
Such eye tests include the following:
- Ophthalmoscopy, optic nerve photography, and, in some cases, optical coherence tomography (OCT)
- Formal visual field assessment
- Ocular motility examination
Typically, no laboratory testing is mandatory, but some doctors have recommended blood tests depending on clinical circumstances, including the following:
- Complete blood count
- Electrolytes, bicarbonate
- Full procoagulant profile (only in patients with a previous history of thrombosis or magnetic resonance imaging [MRI] evidence of a dural venous sinus occlusion on magnetic resonance venography [MRV])
Brain imaging
Neuroimaging studies are essential to rule out the possibility of an intracranial lesion prior to performing a lumbar puncture. In addition, it is necessary to rule out the potential associated diagnosis of a dural sinus thrombosis. As a result, a combined MRI/MRV of the brain with gadolinium is the preferred study. At the very least, initially, CT scanning of the brain to rule out an intracranial lesion can be performed if a MRI is not immediately available.
Spinal tap (lumbar puncture)
Once an intracranial mass lesion is ruled out, a lumbar puncture is usually indicated.
Cerebrospinal fluid studies may include the following:
- Opening pressure
- White blood cell and differential counts
- Red blood cell count
- Total protein
- Quantitative protein electrophoresis
- Glucose
- Aerobic bacterial culture and sensitivity
- Acid-fast bacilli culture
- Cryptococcal antigen (especially in patients with HIV)
- Syphilis markers (e.g., rapid plasma reagin)
- Tumor markers and cytology (in patients with a history of cancer or with clinical features suggesting occult malignancy)
The diagnosis is also confirmed by detecting a high spinal CSF pressure reading, usually greater than 250 mmH2O (200-250 considered borderline high) and normal laboratory and imaging studies including CT scans and MRIs.
Pseudotumor cerebri treatment
If a diagnosis of pseudotumor cerebri is confirmed, careful and repeated eye (ophthalmologic) exams are required to check for any changes in vision 9. The goal is to preserve optic nerve function while managing increased intracranial pressure.
Visual loss in one or both of the eyes can evolve rapidly despite the best efforts to arrest the process. Idiopathic intracranial hypertension has been a frequent cause of litigation. Almost uniformly, cases center on the delay of maximum medical and surgical management for patients who present with rapidly declining vision.
The exact time window within which vision loss can be reversed after symptomatic decline is not known. Opinions among experts in the field vary as to how rapidly and aggressively any given patient should have been treated. As a rule, it is better to err on the side of rapid intervention (i.e., within hours to days) in such patients. This is a dramatic opportunity to save vision that can be easily lost. A major medicolegal pitfall is created when poor outcome is coupled with the perception of delayed treatment.
One of the standard teachings regarding this condition has been that pregnancy exacerbates or triggers the onset of symptomatic pseudotumor cerebri. At present, however, there is little statistical evidence of a causal association between pregnancy and pseudotumor cerebri, beyond the fact that pregnancy is common in the age group and gender that is predominantly affected by pseudotumor cerebri 10.
Pharmacologic therapy may include the following:
- Medications such as acetazolamide (the most effective agent for lowering intracranial pressure) and furosemide may be used to reduce fluid build-up and relieve pressure 9. Losing weight and stopping certain medications (including oral contraceptives, tetracycline, and a variety of steroids) may lead to improvement.
- Primary headache prophylaxis (e.g., amitriptyline, propranolol, other commonly prescribed migraine prophylaxis agents, or topiramate)
- Corticosteroids for lowering intracranial pressure in pseudotumor cerebri of inflammatory etiology or for supplementing acetazolamide. Typically, this should be considered only on a short-term basis and only in patients who present with marked loss of visual function because steroids can cause weight gain and withdrawal of steroids may worsen pseudotumor cerebri.
In a 2014 National Institutes of Health funded study 11 of 165 patients with idiopathic intracranial hypertension and mild vision loss (the Idiopathic Intracranial Hypertension Treatment Trial), researchers found that acetazolamide treatment for six months in conjunction with a low-sodium weight-reduction diet modestly improved vision, reduced intracranial pressure, improved quality of life, and reduced papilledema. Average improvement in perimetric mean deviation (PMD) was 1.43 dB with acetazolamide and 0.71 dB with placebo. Data show that the effect of the drug was independent of weight loss 12. Compared with the placebo group, patients on acetazolamide treatment had statistically significant improvement of retinal nerve fiber layer thickness, total retinal thickness, and optic nerve volume based on optical coherence tomography measurements 13.
Acetazolamide appears to be the most effective agent for lowering intracranial pressure. Most patients experience adequate relief of symptoms (typically, headache) with this first-line agent. The brand-name formulation Diamox appears to be better tolerated than generic acetazolamide.
The initial dosage should be 0.5-1 g/day. Although many physicians start patients on 250 mg twice daily, others consider this dosage too low. A 500-mg oral dose of Diamox Sequels twice daily is preferred; however, some insurers only cover an oral dose of 250 mg 4 times per day. Most patients respond to a dosage of 1-2 g/day. This can be increased to 3-4 g/day, but most patients cannot tolerate the adverse effects (e.g., extremity paresthesias, fatigue, metallic taste from carbonated beverages, and decreased libido) that occur at this high dosage 14. In the Idiopathic Intracranial Hypertension Treatment Trial, of the 86 patients in the acetazolamide treatment group, 38 (44.1%) of patients tolerated the maximum dose of 4 g/day 11. Seventy-seven (89.5%) of the 86 patients taking acetazolamide tolerated dosages of 1 g/day or more. Compared with the 79 patients from the placebo group, the 86 patients taking acetazolamide were more likely to experience paresthesia, dysgeusia (foul or metallic taste), vomiting and diarrhea, nausea, and fatigue 11.
In the event of intolerance to acetazolamide, furosemide may be used as a replacement diuretic in this group. Unfortunately, it does not appear to be as effective as acetazolamide.
If visual function deteriorates while on maximal medical therapy, surgical interventions should be strongly considered. Such interventions include the following:
- Optic nerve sheath fenestration (a protective measure to decompress the fluid surrounding the optic nerve)
- Cerebrospinal fluid diversion (i.e., via a lumboperitoneal or ventriculoperitoneal shunt). Therapeutic shunting, which involves surgically inserting a tube to drain cerebrospinal fluid (CSF) from the lower spine into the abdominal cavity, may be needed to remove excess CSF and relieve CSF pressure 9.
- Intracranial venous sinus stenting (in the presence of an area of evident focal stenosis of the dural sinus venous system)
Optic nerve sheath fenestration. The ophthalmic surgical approach to managing patients with progressive vision loss and papilledema involves cutting slits or rectangular patches in the dura surrounding the optic nerve immediately behind the globe 15. This allows direct egress of CSF into the orbital fat, where it is absorbed into the venous circulation.
Optic nerve sheath fenestration has been demonstrated to reverse optic nerve edema and to bring about some recovery of optic nerve function. In addition, it may decrease headache in many patients. The approach to the optic nerve may be from either the medial or the lateral aspect of the orbit; each approach has its benefits and drawbacks 16.
Although intracranial pressure typically remains elevated in these patients postoperatively, the local filtering effect of the optic nerve sheath fenestration acts as a safety valve and keeps the pressure from being transmitted to the optic nerve. Despite the general lack of an intracranial pressure-lowering effect, unilateral surgery occasionally has a bilateral curative effect on the papilledema. However, if this is not the case, the opposite nerve must undergo the same procedure.
Complications related to optic nerve sheath fenestration include the following:
- Diplopia
- Optic nerve injury
- Vascular occlusion
- Tonic pupil
- Hemorrhage and infection (risks inherent to intraconal surgery)
Unfortunately, the procedure may not have lasting benefits. In most cases, visual function stabilizes or improves after optic nerve sheath decompression in the short run 17, but in at least one third of cases, secondary visual decline may occur within 3-5 years and may necessitate repeat surgery or an alternative treatment; Spoor and McHenry 18 found the long-term success rate of this operation to be only 16%.
A study of optic nerve sheath fenestration performed in 41 eyes from 21 patients with vision loss from either idiopathic intracranial hypertension or intracranial hypertension from cerebral venous thrombosis found best-corrected visual acuity and visual field to be stabilized or improved in 32 of 34 eyes (94%) over a 3-month follow-up interval 19. Transient benign complications were apparent in 4 eyes. Only marginal improvement was shown in 4 eyes with no light perception vision; these were not analyzed with the remainder of the group.
Treatment of pseudotumor cerebri with repeated lumbar punctures is considered to be of historic interest only, as the CSF volume reforms rapidly. However, serial lumbar punctures have been considered in some patients who refuse or cannot undergo conventional medical or surgical therapy (e.g., pregnant patients). Placement of a lumbar drain as a temporizing measure in hospitalized fulminant idiopathic intracranial hypertension cases can be considered while awaiting a definitive surgical procedure.
CSF diversion procedures
CSF diversion procedures are highly effective in lowering intracranial pressure 20. In some facilities, they remain the procedures of choice for treating pseudotumor cerebri patients who do not respond to maximum medical treatment 16. Shunts are also indicated for patients with intractable headaches, patients in regions where there is no access to a surgeon comfortable with optic nerve sheath fenestration, and patients in whom optic nerve sheath fenestration has failed.
Lumboperitoneal shunting is the traditional method for providing prompt reduction of intracranial pressure in patients with pseudotumor cerebri 21. However, this procedure has a high 1-year failure rate. Some neurosurgeons currently prefer ventriculoperitoneal or ventriculoatrial shunting to lumboperitoneal shunting.
The main reason why these neurosurgeons prefer ventricular shunts is that such shunts can be monitored for function by using an extracranial subcutaneous compressible bulb and a 1-way valve (permitting intracranial-to-abdominal flow) in series with the intracranial and abdominal ends of the shunt. The bulb will resist digital compression if the distal (abdominal or atrial) end is obstructed. It will collapse under digital pressure but will fail to reinflate if the intracranial end is obstructed.
Many neurosurgeons have been reluctant to place ventricular shunts in patients with pseudotumor cerebri because the ventricles are small and difficult to cannulate without radiographic guidance. In addition, there is a significant risk of complications (e.g., infection, stroke, seizures, and shunt failure). However, Woodworth et al 22 reported that with a frameless stereotactic approach, they were able to place ventricular shunts in a single pass in all patients, even into slit ventricles, with good long-term viability.
Sinclair et al 23 found that although CSF diversion minimizes visual decline and improves visual acuity, 68% of patients continued to have headaches and 28% had low-pressure headaches that complicated surgery. Shunt revision was required for 51% of patients, with most requiring multiple revisions. For those with otherwise untreatable rapidly declining vision, CSF diversion shunting should be conducted as a last resort. Other treatments, such as weight reduction, may be more effective and may have less associated morbidity.
Intracranial venous sinus stenting
Bussière et al 24 studied 13 patients with pseudotumor cerebri that was refractory to medical management who were found to have stenosis of one or both of the transverse dural sinuses on time-of-flight magnetic resonance (MR) venography. Of these, 10 also had increased pressure gradient across the stenotic dural venous sinus (>10 mm Hg), and these underwent stent placement without significant complications.
All 10 of the patients experienced complete resolution or significant improvement of their headaches, and 8 experienced complete or near-complete resolution of papilledema 24. The authors suggested only that a randomized, controlled study of transverse dural venous sinus stenting in the management of pseudotumor cerebri was needed to establish the safety and efficacy of the procedure in this setting.
Arac et al 25 reported 1 case of endovascular stenting for pseudotumor cerebri and reviewed the same published series that Bussière et al did, arriving at essentially the same conclusions.
A meta-analysis comparing visual outcomes after these interventions reported the following findings 26:
- 252 optic nerve sheath decompressions: Visual defects improved or resolved in 80%
- 31 ventriculoperitoneal shunt shunt placements: Visual defects improved or resolved in 38.7%
- 44 lumboperitoneal shunt placements: Visual defects improved or resolved in 44.6%
- 17 intracranial venous sinus stent placements: Visual defects improved or resolved in 47%
There are distinct risks and benefits associated with each of these interventions.
Visual worsening was rare for all procedures. Study author 26 concluded that visual outcome was best documented for optic nerve sheath fenestration, which appeared to be the best surgical procedure for vision loss in pseudotumor cerebri. While optic nerve sheath decompression improves visual defects, many patients continue to have significant headaches that require CSF shunt placement 27.
Diet and Weight Loss
Most patients with pseudotumor cerebri are females who are overweight. Weight loss is a cornerstone in the long-term management of these patients. No physical activity restriction is required; in fact, exercise programs are strongly recommended in conjunction with the weight-reduction diet. As little as a 5-10% weight loss has been demonstrated to yield a reduction in intracranial pressure with accompanying resolution of papilledema 28. Unfortunately, weight reduction generally proves to be a difficult task for these patients 29. To formalize the process of weight reduction, referral to a dietitian is appropriate.
On initial diagnosis, a weight-reduction diet should be strongly recommended to all patients with pseudotumor cerebri. Often, a formal weight-loss program is required.
Some patients with pseudotumor cerebri have experienced dramatic improvement after bariatric surgery.
In a review of the literature on bariatric surgery for obese patients with pseudotumor cerebri, Fridley et al 30 found a total of 62 patients, of whom 52 (92%) experienced resolution of the presenting symptoms. Of 35 patients who underwent postoperative funduscopy, 34 had resolution of papilledema. Of 12 patients who underwent pre- and postoperative visual field examinations, 11 showed resolution of visual field defects.
Among 13 patients in whom CSF pressures were measured pre- and postoperatively, there was an average postoperative decrease of 254 mm water 30. The authors called for prospective controlled studies to confirm the effectiveness of this surgical approach for pseudotumor cerebri patients in long-term follow-up.
References- Dessardo NS, Dessardo S, Sasso A, Sarunic AV, Dezulovic MS. Pediatric idiopathic intracranial hypertension: clinical and demographic features. Coll Antropol. 2010 Apr. 34 Suppl 2:217-21.
- Jindal M, Hiam L, Raman A, Rejali D. Idiopathic intracranial hypertension in otolaryngology. Eur Arch Otorhinolaryngol. 2009 Jun. 266(6):803-6.
- An Estimation of the Risk of Pseudotumor Cerebri among Users of the Levonorgestrel Intrauterine Device. Neuroophthalmology. 2017 Apr 19;41(4):192-197. doi: 10.1080/01658107.2017.1304425. eCollection 2017 Aug. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5762147/
- Dugdale DC, Hoch DB. Pseudotumor cerebri. MedlinePlus. https://medlineplus.gov/ency/article/000351.htm
- Wakerley B, Tan M, Ting E. Idiopathic intracranial hypertension. Cephalalgia. 2014; Epub ahead of print:http://www.ncbi.nlm.nih.gov/pubmed/24847166 https://www.ncbi.nlm.nih.gov/pubmed/24847166
- Kosmorsky GS. Idiopathic intracranial hypertension: pseudotumor cerebri. Headache. 2014; 54(2):389-393. https://www.ncbi.nlm.nih.gov/pubmed/24512582
- Dykhuizen MJ, Hall J.. Cerebral venous sinus system and stenting in pseudotumor cerebri. Current Opinions in Ophthalmology. 2011; 22(6):458-462. https://www.ncbi.nlm.nih.gov/pubmed/21986881
- Wall M. Idiopathic intracranial hypertension (pseudotumor cerebri). Curr Neurol Neurosci Rep. 2008 Mar. 8(2):87-93.
- Pseudotumor Cerebri Information Page. https://www.ninds.nih.gov/Disorders/All-Disorders/Pseudotumor-Cerebri-Information-Page
- Giuseffi V, Wall M, Siegel PZ, Rojas PB. Symptoms and disease associations in idiopathic intracranial hypertension (pseudotumor cerebri): a case-control study. Neurology. 1991 Feb. 41;2,Pt 1:239-44.
- ten Hove MW, Friedman DI, Patel AD, Irrcher I, Wall M, McDermott MP, et al. Safety and Tolerability of Acetazolamide in the Idiopathic Intracranial Hypertension Treatment Trial. J Neuroophthalmol. 2016 Mar. 36 (1):13-9.
- Wall M, McDermott MP, Kieburtz KD, Corbett JJ, Feldon SE, Friedman DI, et al. Effect of acetazolamide on visual function in patients with idiopathic intracranial hypertension and mild visual loss: the idiopathic intracranial hypertension treatment trial. JAMA. 2014 Apr 23-30. 311(16):1641-51.
- Optical Coherence Tomography Substudy Committee., NORDIC Idiopathic Intracranial Hypertension Study Group. Papilledema Outcomes from the Optical Coherence Tomography Substudy of the Idiopathic Intracranial Hypertension Treatment Trial. Ophthalmology. 2015 Sep. 122 (9):1939-45.e2
- Wall M. Idiopathic intracranial hypertension (pseudotumor cerebri). Curr Neurol Neurosci Rep. 2008 Mar. 8(2):87-93.
- Goh KY, Schatz NJ, Glaser JS. Optic nerve sheath fenestration for pseudotumor cerebri. J Neuroophthalmol. 1997 Jun. 17(2):86-91.
- Brazis PW. Clinical review: the surgical treatment of idiopathic pseudotumour cerebri (idiopathic intracranial hypertension). Cephalalgia. 2008 Dec. 28(12):1361-73.
- Chandrasekaran S, McCluskey P, Minassian D, Assaad N. Visual outcomes for optic nerve sheath fenestration in pseudotumour cerebri and related conditions. Clin Experiment Ophthalmol. 2006 Sep-Oct. 34(7):661-5.
- Spoor TC, McHenry JG. Long-term effectiveness of optic nerve sheath decompression for pseudotumor cerebri. Arch Ophthalmol. 1993 May. 111(5):632-5.
- Nithyanandam S, Manayath GJ, Battu RR. Optic nerve sheath decompression for visual loss in intracranial hypertension: report from a tertiary care center in South India. Indian J Ophthalmol. 2008 Mar-Apr. 56(2):115-20.
- Yadav YR, Parihar V, Agarwal M, Bhatele PR, Saxena N. Lumbar peritoneal shunt in idiopathic intracranial hypertension. Turk Neurosurg. 2012. 22(1):21-6.
- Burgett RA, Purvin VA, Kawasaki A. Lumboperitoneal shunting for pseudotumor cerebri. Neurology. 1997 Sep. 49(3):734-9.
- Woodworth GF, McGirt MJ, Elfert P, Sciubba DM, Rigamonti D. Frameless stereotactic ventricular shunt placement for idiopathic intracranial hypertension. Stereotact Funct Neurosurg. 2005. 83(1):12-6.
- Sinclair AJ, Kuruvath S, Sen D, Nightingale PG, Burdon MA, Flint G. Is cerebrospinal fluid shunting in idiopathic intracranial hypertension worthwhile? A 10-year review. Cephalalgia. 2011 Dec. 31(16):1627-33.
- Bussière M, Falero R, Nicolle D, Proulx A, Patel V, Pelz D. Unilateral transverse sinus stenting of patients with idiopathic intracranial hypertension. AJNR Am J Neuroradiol. 2010 Apr. 31(4):645-50.
- Arac A, Lee M, Steinberg GK, Marcellus M, Marks MP. Efficacy of endovascular stenting in dural venous sinus stenosis for the treatment of idiopathic intracranial hypertension. Neurosurg Focus. 2009 Nov. 27(5):E14.
- Feldon SE. Visual outcomes comparing surgical techniques for management of severe idiopathic intracranial hypertension. Neurosurg Focus. 2007. 23(5):E6.
- Banta JT, Farris BK. Pseudotumor cerebri and optic nerve sheath decompression. Ophthalmology. 2000 Oct. 107 (10):1907-12.
- Rowe FJ, Sarkies NJ. Assessment of visual function in idiopathic intracranial hypertension: a prospective study. Eye (Lond). 1998. 12 ( Pt 1):111-8.
- Johnson LN, Krohel GB, Madsen RW, March GA Jr. The role of weight loss and acetazolamide in the treatment of idiopathic intracranial hypertension (pseudotumor cerebri). Ophthalmology. 1998 Dec. 105(12):2313-7.
- Fridley J, Foroozan R, Sherman V, Brandt ML, Yoshor D. Bariatric surgery for the treatment of idiopathic intracranial hypertension. J Neurosurg. 2011 Jan. 114(1):34-9.