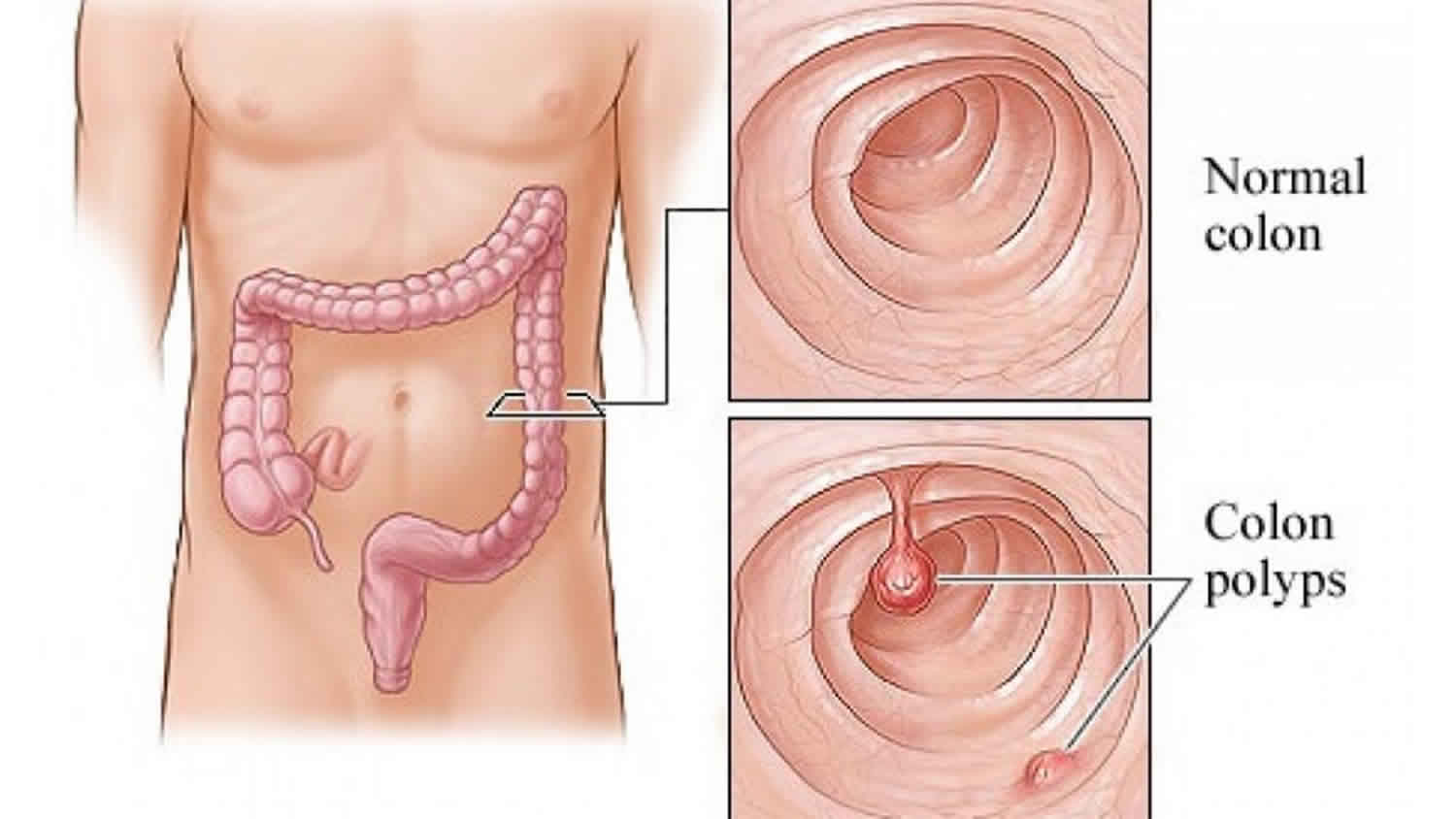
What is tubular adenoma
Tubular adenoma refers broadly to any benign tumor of glandular tissue in the intestines 1. This article will address specifically adenomas of the colon, occurring within polyps. Adenomas are defined as possessing at least the characteristics of low-grade dysplasia 1. Some adenomas may progress over an extended period from low-grade dysplasia to high-grade dysplasia, carcinoma in situ, or invasive adenocarcinoma 1. Although physicians debate this, and the evidence does not completely confirm, thus far, if villous features indicate an increased risk of developing colorectal cancer, multiple studies demonstrate that histologic villous features of adenomas may be associated with developing advanced forms of neoplasia 1. Size at baseline of adenomas has been shown in some studies to influence future advanced disease; lesions greater than 1 cm increase a patient’s risk 1. One source states that villous lesions have an increased risk of containing adenocarcinoma correlated to size specifically, with a 10% to 20% risk in adenomas larger than 2 cm, and a 5% risk in adenomas from 1 cm to 2 cm in size. However, these findings are inconsistent when considered across multiple studies regarding the subject 1. A recent study proposes using the overall sum of all adenoma diameters (called the adenoma bulk) at initial surveillance colonoscopy to stratify patients as either high or low-risk for predicting the development of advanced neoplasia 1. This study states that the adenoma bulk is comparatively predictive as the conventional current model using histology features when using a risk stratification cutoff of 10mm for the adenoma bulk, with high-risk adenoma bulk being greater than or equal to 10 mm 1.
Adenomas can have several different growth patterns that can be seen under the microscope by the pathologist. There are 2 major growth patterns: tubular and villous. Many adenomas have a mixture of both growth patterns, and are called tubulovillous adenomas. Most adenomas that are small (less than ½ inch) have a tubular growth pattern. Larger adenomas may have a villous growth pattern. Larger adenomas more often have cancers developing in them. Adenomas with a villous growth pattern are also more likely to have cancers develop in them.
The most important thing is that your polyp has been completely removed and does not show cancer. The growth pattern is only important because it helps determine when you will need your next colonoscopy to make sure you don’t develop colon cancer in the future.
Colon polyps are a common entity, increasing in prevalence with age. They are more common in the western countries. By age 50, colorectal cancer screening studies have demonstrated a prevalence of 25% to 30%, increasing to 50% by age 70 in high-risk Western countries such as the United States 2. Polyps are most prevalent in non-white men 3. Colon polyps are rare in the younger population, present in only one to four percent of 20 to 30-year-olds. However, 6% of children are affected, and the percentage goes up to 12% in children with lower gastrointestinal bleeding. The three different colon polyps are villous, tubular and tubulovillous. Adenomatous polyps will gradually show dysplastic changes, which differentiates them from hyperplastic polyps. In general, colonic polyps are benign but those that develop high-grade dysplasia will become malignant with time 4.
Adenomatous polyps are the most common type of polyp in the colon, accounting for about 60% to 70% of all colonic polyps. Conventional type adenomatous polyps can be classified as tubular, villous, or tubulovillous. Villous adenomas are characterized by more than 75% villous features, where villous refers to finger-like or leaf-like epithelial projections. Tubulovillous adenomas have between 25% and 75% villous features. Less than 25% villous features indicate a tubular adenoma. Adenomas are usually asymptomatic and found on routine colorectal cancer screening. Adenomas with villous features may be associated with a slight increase in development of more advanced neoplasia or dysplasia compared to other types of adenomas 5.
Colon polyps are protrusions occurring in the colon lumen most commonly sporadic or as part of other syndromes 4. Colon polyps are classified as diminutive if 5 mm in diameter or less, small if 6 to 9 mm, or large if they are 1 cm in diameter or more. Colon polyps can be depressed, flat, sessile or pedunculated. Few polyps arise from submucosa including lipomas, carcinoids or lymphoid aggregates. Most commonly, however, they result from the mucosa, and they can be adenomatous (tubular more than 80%, villous 5-15%, tubulovillous 5-15%), serrated (sessile or traditional), or non-neoplastic (hyperplastic and juvenile). Hyperplastic polyps are very common and have a very low malignant potential. They are encountered more in the distal colon. Juvenile polyps are benign hamartomas and are common in childhood 6.
The clinical significance of colon polyps arises from the fact that more than 95% of colon adenocarcinoma originate from polyps. Sessile serrated polyps are common, found more in the proximal colon, and have low malignant potential if there is no dysplasia, and significant malignant potential if they are dysplastic. Traditional serrated adenoma is uncommon, more often noted in the distal colon, with significant malignant potential. Non-neoplastic polyps include hyperplastic, juvenile polyps, hamartomas, and inflammatory pseudopolyps. Hyperplastic polyps are very common, found more in the distal colon, with very low malignant potential. Juvenile polyps are benign hamartomas and are not premalignant. They are common in childhood and more in the rectosigmoid. Polyps develop into cancer after inactivation of tumor suppressor genes as adenomatous polyposis coli (APC), or mismatch repair genes (MLH1). Genetics play a significant role; familial adenomatous polyposis (FAP) is autosomal dominant, and the majority of affected patients develop colorectal cancer by age 40. Juvenile polyposis syndrome is also autosomal dominant. Fifty to sixty percent of patients have a mutation in the SMAD4 or BMPR1A gene. By age 35, 20% of patients develop colorectal cancer. Other forms of inheritance exist as in MUTYH-associated polyposis (MAP) which is autosomal recessive caused by biallelic mutations in MUTYH gene 7.
Recommendations that may decrease the incidence of colon polyps include low-fat, high fiber diet, decreased alcohol intake, and smoking cessation. No benefit was noted to decrease rates of colorectal cancer with the use of multivitamins, folate, calcium, statins, azathioprine or 6-mercaptopurine 4. Some studies found benefit using nonsteroidal anti-inflammatory drugs (NSAIDs) and calcium in patients with familial adenomatous polyposis (FAP) and MUTYH-associated polyposis (MAP), where they may decrease the incidence and recurrence of polyps.
How does having an adenoma affect my future follow-up care?
Since you had an adenoma, you will need to have another colonoscopy to make sure that you don’t develop any more adenomas. When your next colonoscopy should be scheduled depends on a number of things, like how many adenomas were found, if any were villous, and if any had high-grade dysplasia. The timing of your next colonoscopy should be discussed with your treating doctor, as he or she knows the details of your specific case.
What if my adenoma was not completely removed?
If your adenoma was biopsied but not completely removed, you will need to talk to your doctor about what other treatment you’ll need. Most of the time, adenomas are removed during a colonoscopy. Sometimes, though, the adenoma may be too large to remove during colonoscopy. In such cases you may need surgery to have the adenoma removed.
Tubular adenoma histopathology
It is important to differentiate degree of dysplasia present in adenomas. The degree of dysplasia present in an adenoma is determined by both cytological and architectural features. Adenomas are tumors of dysplastic epithelium which can be characterized as having a low grade or high-grade dysplasia, which indicates the level of maturation of the epithelium. By definition, adenomas have at least low-grade dysplasia.
The cytological features of low-grade dysplasia include crowded, pseudo-stratification to early stratification of spindled or elongated nuclei which occupy the basal half of the cytoplasm. Pleomorphism and atypical mitoses should be absent or minimally present. Mitotic activity and minimal loss of cell polarity are allowed. Architecturally, the crypts should maintain a resemblance to normal colon, without significant crowding, cribriform, or complex forms.
High-grade dysplasia cytologically boasts increased nucleus to cytoplasm ratio, more significant loss of polarity, more “open” appearing nuclei with increasingly prominent nucleoli. Other features which distinguish a high grade from low-grade dysplasia include significant pleomorphism, rounded nuclei, atypical mitoses, and significant loss of polarity. Mitotic figures may be observed as in low-grade dysplasia. Cribriform and crowding, back-to-back glands indicate high-grade dysplasia and can be important architectural features to aid in differentiating some cases of low vs. high-grade dysplasia which may have less distinct cytological features.
The villous component of adenomas refers to epithelial finger-like projections away from the muscularis mucosae. There should be deep crypts, and the projections should contain fibrovascular cores lined by dysplastic epithelium. As stated above, the amount of villous differentiation distinguishes villous adenomas (over 75% villous features) versus tubulovillous adenomas (mixed tubular and villous features with 25% to 75% villous features).
What if my report mentions dysplasia?
Dysplasia is a term that describes how much your polyp looks like cancer under the microscope:
- Polyps that are only mildly abnormal (don’t look much like cancer) are said to have low-grade (mild or moderate) dysplasia.
- Polyps that are more abnormal and look more like cancer are said to have high-grade (severe) dysplasia.
The most important thing is that your polyp has been completely removed and does not show cancer. If high-grade dysplasia is found in your polyp, it might mean you need to have a repeat (follow-up) colonoscopy sooner than if high-grade dysplasia wasn’t found, but otherwise you do not need to worry about dysplasia in your polyp.
What if my report also mentions hyperplastic polyps?
Hyperplastic polyps are typically benign (they aren’t pre-cancers or cancers) and are not a cause for concern.
What is adenocarcinoma of the colon?
Adenocarcinoma is a type of cancer that starts in the cells that form glands making mucus to lubricate the inside of the colon and rectum. This is the most common type of colon cancer.
Tubular adenoma causes
The risk of developing colorectal polyps is related to both environmental and genetic factors. Risk factors include advancing age, male gender, high-fat, low-fiber diet, tobacco use, and excess alcohol intake (more than eight drinks a week). Individuals with a family history of polyps, colorectal cancer, and intestinal polyposis carry a higher risk of developing colon polyps. Inflammatory bowel disease was found to be associated with decreased prevalence of polyps 8.
The majority of polyps in the colon arise sporadically even though genetics play the most important role in influencing the individual risk of developing colon polyps and subsequent colorectal cancer. Personal history of colon adenomas places a patient at increased risk for future development of colon cancer 9.
Colorectal cancer has been shown to arise mainly from pre-existing adenomatous colon polyps. Adenomatous polyps form as mentioned above primarily in a sporadic fashion. The sporadic nature of these polyps is a result of either a mutation in the adenomatous coli pathway or DNA mismatch repair.
Familial adenomatous polyposis (FAP) is a genetic disorder which, in particular, predisposes affected patients to the development of multiple adenomatous polyps 1. Familial adenomatous polyposis (FAP) is an autosomal dominant disorder caused by a mutation in the tumor suppressor APC gene, located on chromosome 5q. This mutation can either be inherited or in as many as one out of three patients, can present as a new germline mutation. The frequency of familial adenomatous polyposis (FAP) is from 1 in 8,000 to 1 in 14,000, with large numbers of adenomas occurring beginning in late childhood. Classically, patients with FAP have over 100 colorectal adenomatous polyps, but many patients have several hundred, or even thousands, of polyps. These patients usually develop adenocarcinoma by the age of 30 to 40. Even given the likely development of colon cancer in these patients, FAP only accounts for around 1% of colon cancers.
Tubular adenoma diagnosis
Medical history and physical examination
Patients with polyps are usually asymptomatic and usually found on screening colonoscopies for colorectal cancer. Patients may also present with painless rectal bleeding of bright or dark red color, on wiping or with bowel movements mixed with stools or dripping. Other presentations include diarrhea, constipation, abdominal pain, mucus with stools, or with symptoms and signs of iron deficiency anemia secondary to chronic bleeding. A physical exam is usually normal, although rectal polyps may be palpated on digital rectal exam or if they are prolapsed. Fecal occult blood test (FOBT) by digital rectal exam is not as effective as fecal occult blood test (FOBT) done on stool passed spontaneously.
Current US Preventive Services Task Force recommendation for average-risk adults is that beginning at age 50 patients should initiate regular colon cancer screening. Methods include a fecal occult blood test (FOBT), fecal immunochemical test (FIT), and direct visualization methods. Direct visualization through colonoscopy, biopsy, and histologic examination of colon lesions is necessary to examine and diagnose colorectal polyps and cancers.
Tests
Initial testing includes CBC (complete blood count) to check for anemia found in chronic bleeding and basic metabolic panel (BMP) to check for electrolyte disorders as with hypersecretory adenomas. Guaiac (gFOBT) uses a chemical indicator with color change in the presence of blood and immunochemical (iFOBT or fecal immunochemical test [FIT]) uses antibodies against human hemoglobin. Stool DNA test is more sensitive but less specific than fecal immunochemical test (FIT) 10.
The gold standard test for polyps detection is colonoscopy, where polypectomy is done, and management is based on pathology results. This is however not a faultless screening test, as it has increased miss rate with low colon prep quality, smaller polyp, right-sided colon polyps, and less experience of the endoscopist. Patients found to have more than ten colorectal adenomas should be tested for MUTYH and APC. A newer way of screening is computed tomographic colonography (CTC) which requires excellent bowel preparation and is less sensitive with flat polyps. Other ways to diagnose are with double-contrast barium enemas and colon capsule endoscopies. Further research is still needed for enhanced optical technologies which may be used to differentiate between neoplastic and non-neoplastic colonic polyps, and they include narrowed spectrum endoscopy narrow-band imaging [NBI], confocal laser endomicroscopy [CLE], Fujinon intelligent chromoendoscopy [FICE] and image-enhanced endoscopy [i-scan].
Tubular adenoma treatment
Surgeons should excise adenomatous polyps of any type completely and confirm clear margins 11. Colonic polypectomy is done during colonoscopy for diagnostic and therapeutic purposes using snare polypectomy with electrocautery for pedunculated polyps, or mucosal resection for sessile polyps.
Based on the morphology and a number of lesions removed, surveillance guidelines according to the American Cancer Society are as follows:
- Average risk (no first-degree relative to colon cancer): Colonoscopy at age 50
- No adenoma or carcinoma, repeat in 10 years
- One to two small (no more than 1 cm) tubular adenomas with low-grade dysplasia, repeat in 5 to 10 years
- Three to ten adenomas, or a large (at least 1 cm) adenoma, or any adenomas with high-grade dysplasia or villous features, repeat in 3 years.
- More than ten adenomas on a single exam, repeat within 3 years
- Increased risk (positive family history in the first-degree relative before age 60, or in two or more first-degree relatives at any age if not a hereditary syndrome) Colonoscopy at age 40, or 10 years before the youngest case in the immediate family (whichever is earlier). Repeat with above surveillance guidelines with the caveat that maximum time between screening should be 5 years.
- High-risk (hereditary colon cancer/polyposis syndromes):
- Familial Adenomatous Polyposis (FAP): Annual flexible sigmoidoscopy; if genetically proven FAP, consider colectomy.
Total colectomy with ileorectal anastomosis or proctocolectomy ileal pouch anal anastomosis is done for prophylactic reasons for Familial Adenomatous Polyposis (FAP) and MUTYH-associated polyposis (MAP). They may also be done with persistent bleeding or when there are too many polyps. Guidelines for colorectal cancer screening recommend a colonoscopy starting at age 50 for the general population, earlier for at-risk patients, and to stop screening if life expectancy is less than ten years. Risk factors for colon cancer include polyp pathology (adenomatous, serrated, with high-grade dysplasia, with more than 25% villous histology), polyp more than 1 cm in diameter, polyps located in the proximal colon and the number of polyps more than three. Hence, a follow-up colonoscopy is recommended to be done in 10 years if no polyps or distal small hyperplastic polyps are found; in five years if there are small sessile serrated polyps with no dysplasia; in three years if they are large sessile serrated polyp(s) or with dysplasia or traditional serrated adenoma. Other guidelines include screening in five to ten years if one or two small tubular adenomas are seen; in three years if there are three to ten adenomas; in less than three years if there are more than ten adenomas; and in three years if at least one adenoma is ten mm in diameter or more. Alternatively, also screen in three years if one or more adenomas show villous features of any or with high-grade dysplasia. Colectomy is recommended in cases of resected polyps with high-risk features of malignancy (lower third of the submucosa invasion, indeterminate or positive resection margins, margin less than 1 mm, lymphovascular invasion, or poor differentiation) as there is a high incidence of lymph node metastasis 12.
References- Myers DJ, Arora K. Villous Adenoma. [Updated 2018 Dec 13]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2019 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK470272
- Zhou H, Shen Z, Zhao J, Zhou Z, Xu Y. [Distribution characteristics and risk factors of colorectal adenomas]. Zhonghua Wei Chang Wai Ke Za Zhi. 2018 Jun 25;21(6):678-684
- Click B, Pinsky PF, Hickey T, Doroudi M, Schoen RE. Association of Colonoscopy Adenoma Findings With Long-term Colorectal Cancer Incidence. JAMA. 2018 May 15;319(19):2021-2031
- Meseeha M, Attia M. Colon Polyps. [Updated 2018 Nov 18]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2019 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK430761
- Chubak J, McLerran D, Zheng Y, Singal AG, Corley DA, Doria-Rose VP, Doubeni CA, Kamineni A, Haas JS, Halm EA, Skinner CS, Zauber AG, Wernli KJ, Beaber EF., PROSPR consortium. Receipt of Colonoscopy Following Diagnosis of Advanced Adenomas: An Analysis within Integrated Healthcare Delivery Systems. Cancer Epidemiol. Biomarkers Prev. 2019 Jan;28(1):91-98
- Chen EY, Vaccaro GM. Small Bowel Adenocarcinoma. Clin Colon Rectal Surg. 2018 Sep;31(5):267-277
- Dabbous HK, Mohamed YAE, El-Folly RF, El-Talkawy MD, Seddik HE, Johar D, Sarhan MA. Evaluation of Fecal M2PK as a Diagnostic Marker in Colorectal Cancer. J Gastrointest Cancer. 2018 Apr 07
- Yoshizawa N, Yamaguchi H, Kaminishi M. Differential diagnosis of solitary gastric Peutz-Jeghers-type polyp with stomach cancer: a case report. Int J Surg Case Rep. 2018;51:261-264
- Grunwald D, Landau A, Jiang ZG, Liu JJ, Najarian R, Sheth SG. Further Defining the 2012 Multi-Society Task Force Guidelines for Surveillance of High-risk Adenomas: Is a 3-Year Interval Needed for All Patients? J. Clin. Gastroenterol. 2018 Jul 20
- Hakimian S, Jawaid S, Guilarte-Walker Y, Mathew J, Cave D. Video capsule endoscopy as a tool for evaluation of obscure overt gastrointestinal bleeding in the intensive care unit. Endosc Int Open. 2018 Aug;6(8):E989-E993
- Park SK, Yang HJ, Jung YS, Park JH, Sohn CI, Park DI. Risk of advanced colorectal neoplasm by the proposed combined United States and United Kingdom risk stratification guidelines. Gastrointest. Endosc. 2018 Mar;87(3):800-808
- Jover R, Dekker E, Schoen RE, Hassan C, Pellise M, Ladabaum U., WEO Expert Working Group of Surveillance after colonic neoplasm. Colonoscopy quality requisites for selecting surveillance intervals: A World Endoscopy Organization Delphi Recommendation. Dig Endosc. 2018 Nov;30(6):750-759