What is apheresis
Apheresis is the process of withdrawing blood, filtering something out of the blood, and then putting the filtered blood back into the body, using a cell-separating machine. Apheresis is often done on donors where whole blood is centrifuged to obtain individual components (eg, red blood cells, platelets, plasma based on specific gravity) to use for transfusion in different patients. Apheresis may also be used therapeutically to treat various disorders 1.
Apheresis has different names depending on what is being filtered from the blood. Other names include therapeutic plasma exchange, white blood cell reduction and red blood cell exchange.
Apheresis may be used to treat certain blood disorders, cancers, and other diseases. Apheresis also may be used to remove harmful things from the blood, such as toxins or extra iron.
Apheresis can be used to:
- Obtain healthy blood components from a donor to transfuse to a person with a disorder
- Remove harmful substances or excessive numbers of blood cells from the blood of a person with a disorder (termed therapeutic apheresis)
The different components of blood that can be separated include:
- Red blood cells
- Platelets
- Plasma
- White blood cells
- Lipoproteins 2
Plasma contains antibodies (immunoglobulins) and clotting factors, which are sometimes separated from plasma.
The two most common types of apheresis that are used to remove harmful substances from blood are:
- Plasma exchange
- Cytapheresis
Red blood cell exchange also involves apheresis.
Plasma exchange
In plasma exchange, the person’s blood is removed, and the plasma is separated from the blood cells and platelets. The plasma is discarded and the blood cells and platelets are returned to the person along with a plasma-replacing fluid, such as albumin.
Plasma exchange is used to treat disorders in which the person’s plasma contains harmful substances (often antibodies). Such disorders include multiple sclerosis, myasthenia gravis and Guillain-Barré syndrome (neurologic disorders that cause muscle weakness), Goodpasture syndrome (an autoimmune disorder involving bleeding in the lungs and kidney failure), cryoglobulinemia (a disorder involving formation of abnormal antibodies), and thrombotic thrombocytopenic purpura (a rare clotting disorder). Apheresis can help control some chronic diseases but generally does not cure them. However, thrombotic thrombocytopenic purpura can be reversed with apheresis.
To be helpful, plasma exchange must be done often enough to remove the undesirable substance faster than the body produces it. However, apheresis is repeated only as often as necessary because the large fluid shifts between blood vessels and tissues that occur as blood is removed and returned may cause complications in people who are already ill.
Cytapheresis
Cytapheresis is the selective removal of red blood cells, white blood cells, or platelets and can be accomplished by using identical centrifuge-based equipment as plasma apheresis. Cytapheresis can be used to treat polycythemia (an excess of red blood cells), certain types of leukemia (a type of cancer in which there are excess white blood cells), and thrombocythemia (an excess of platelets).
Cytapheresis applications include the following:
- Erythrocytapheresis (selective removal of red blood cells) is used in conditions such as sickle cell disease or malarial infection, in which RBCs are selectively removed and replaced with donor erythrocytes
- Leukapheresis (selective removal of white blood cells) is used in conditions such as hyperleukocytosis, in which pathologically high number of white cells are present (as, for example, in leukemia); it can also be used to collect peripherally circulating stem cells that can then be infused in an autologous or allogeneic stem cell transplant
- Platelet apheresis (selective removal of platelets) can be used in conditions of thrombocytosis (eg, polycythemia vera)
Red blood cell exchange
In red blood cell exchange, diseased or abnormal red blood cells are removed by apheresis and then replaced by donor red blood cells. Red blood cell exchange is used to treat or sometimes prevent serious complications of sickle cell disease, such as stroke and acute chest syndrome.
Plasma apheresis
Plasma apheresis is the collection of plasma by apheresis. Apheresis machines separate the plasma for collection and return the remaining components (red cells, white cells, and platelets) to the donor. This process allows the Blood Service to collect a greater amount of plasma than can be separated from individual whole blood donations.
Extracorporeal photopheresis
Extracorporeal photopheresis is a type of apheresis. During extracorporeal photopheresis, blood is removed. Then white blood cells are removed from the blood. These cells are combined with a medicine and exposed to ultraviolet light to help the medicine work. The treated cells are then put back into the body. After the treatment, the white blood cells are less likely to cause inflammation.
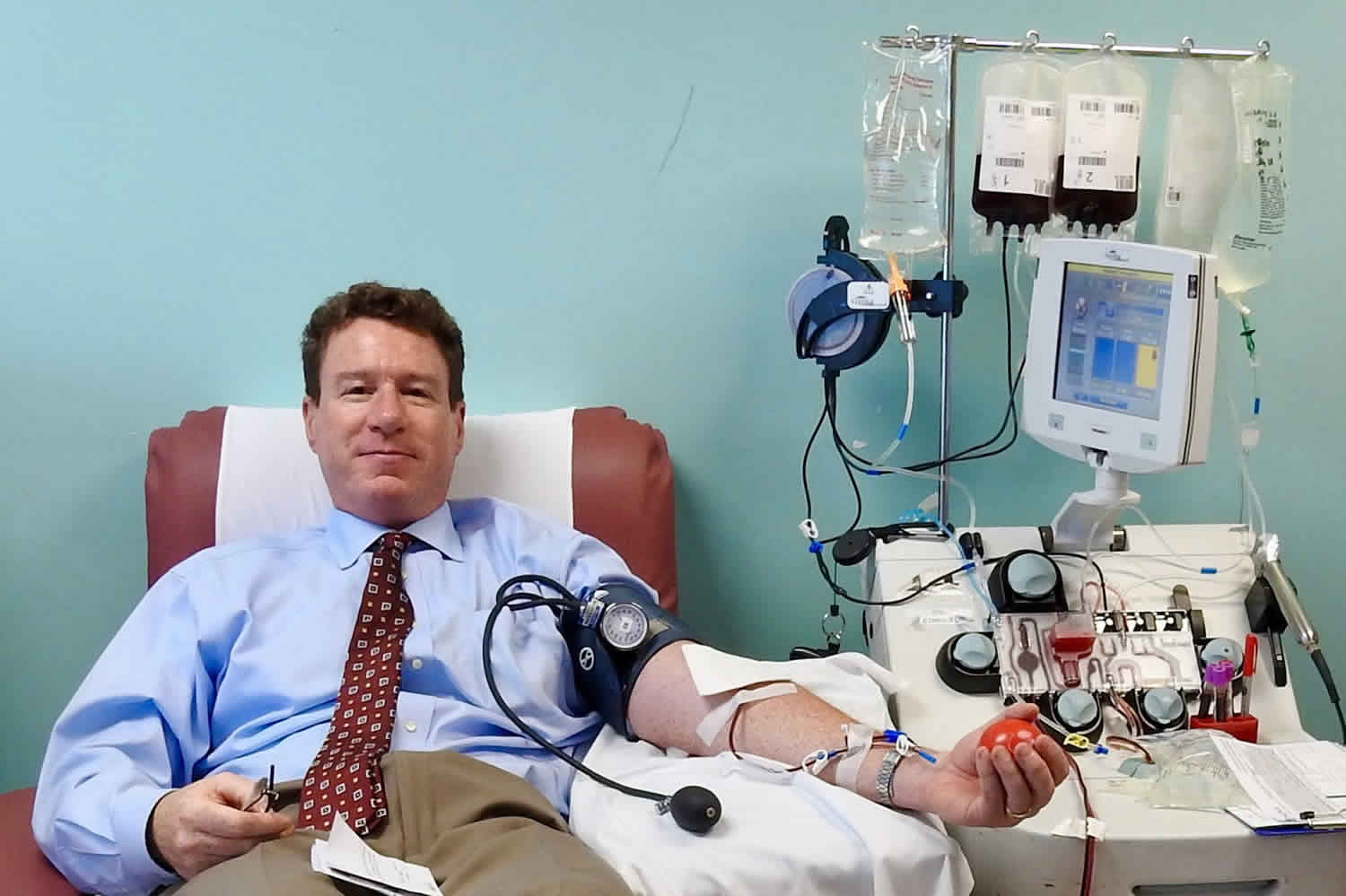
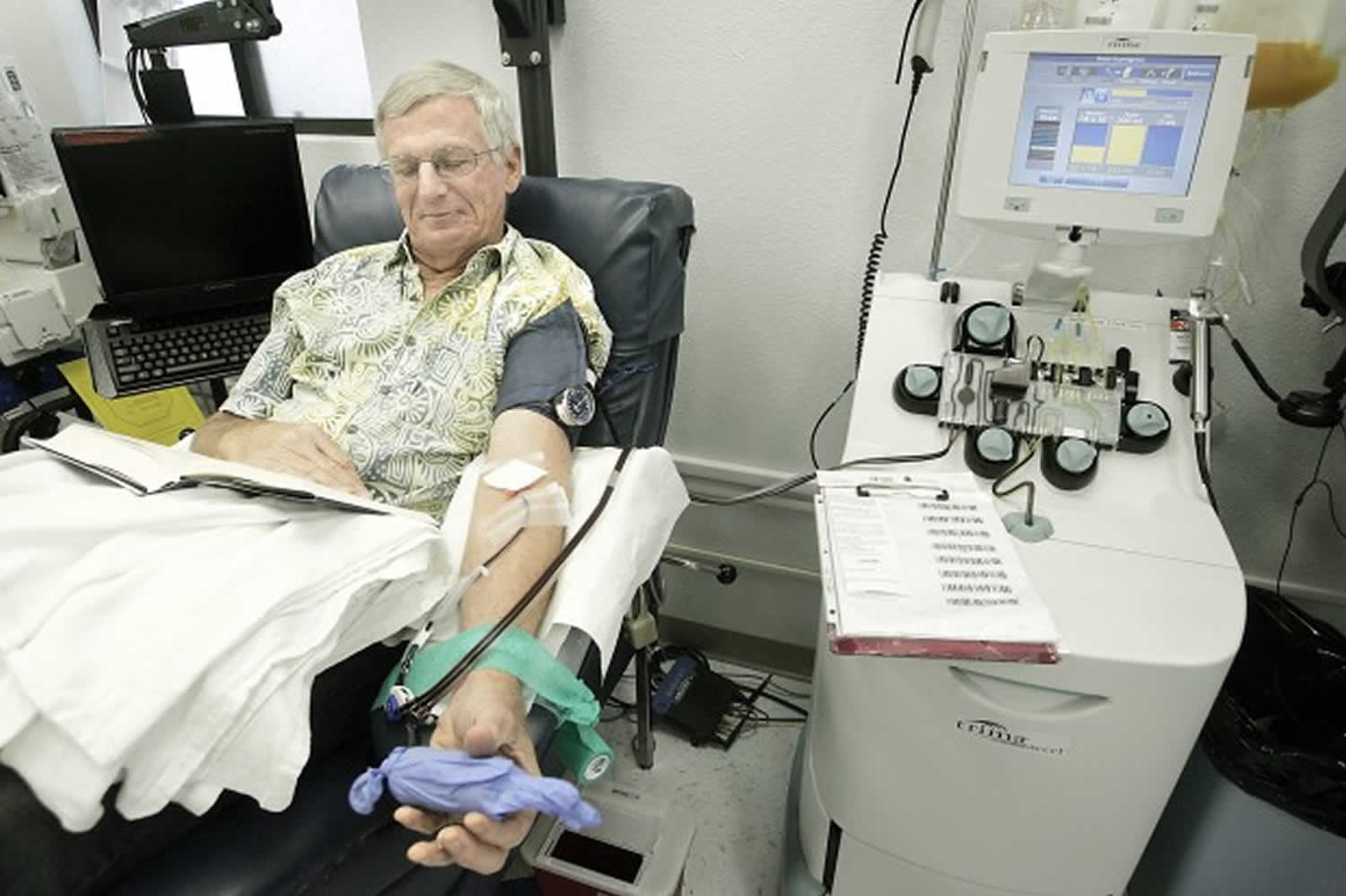
Figure 1. Apheresis machine
What is plasma apheresis?
Plasmapheresis refers to the process of separating plasma from blood, typically by centrifugation or filtration 3. Plasmapheresis is often done on donated blood to obtain plasma for transfusion. Because donors typically give only 1 unit (about 500 mL) of blood and must be in good health, there is no need to replace the removed plasma.
Plasmapheresis also may be done therapeutically to remove certain deleterious substances (eg, autoantibodies, immune complexes) that circulate in plasma. Because large volumes of plasma must be removed, patients are transfused with plasma from healthy donors; thus the process is termed plasma exchange.
The filtering of plasma from whole blood can be accomplished via centrifugation or the use of semipermeable membranes 4. Centrifugation takes advantage of the different specific gravities inherent to various blood products, such as red blood cells (RBCs), white blood cells (WBCs), platelets, and plasma 5. Membrane plasma separation uses differences in particle size to filter plasma from the cellular components of blood 4.
Traditionally, in the United States, most plasmapheresis is done with automated centrifuge-based technology 6. In certain instances—in particular, in patients already undergoing hemodialysis—plasmapheresis can be carried out using semipermeable membranes to filter plasma 5.
In therapeutic plasma exchange, using an automated centrifuge, filtered plasma is discarded and red blood cells along with replacement colloid (e.g., donor plasma or albumin) are returned to the patient.
In membrane plasma filtration, secondary membrane plasma fractionation can selectively remove undesired macromolecules, which then allows return of the processed plasma to the patient instead of donor plasma or albumin. Examples of secondary membrane plasma fractionation include cascade filtration 7, thermofiltration, cryofiltration 8 and low-density lipoprotein pheresis.
Plasma apheresis indications
Plasma apheresis is currently used as a therapeutic modality in a wide array of conditions 3. Generally, it is used when a substance in the plasma, such as immunoglobulin, is acutely toxic and can be efficiently removed. Myriad conditions that fall into this category (including neurologic, hematologic, metabolic, dermatologic, rheumatologic, and renal diseases, as well as intoxications) can be treated with plasmapheresis.
The Apheresis Applications Committee of the American Society for Apheresis periodically evaluates potential indications for apheresis and categorizes them from I to IV in the basis of the available medical literature. The following are some of the indications, and their categorization, from the society’s guidelines 3.
Category I (disorders for which apheresis is accepted as first-line therapy, either as a primary standalone treatment or in conjunction with other modes of treatment) are as follows:
- Guillain-Barre syndrome
- Myasthenia gravis
- Chronic inflammatory demyelinating polyneuropathy
- Hyperviscosity in monoclonal gammopathies
- Thrombotic thrombocytopenic purpura
- Goodpasture syndrome (unless it is dialysis-dependent and there is no diffuse alveolar hemorrhage)
- Hemolytic uremic syndrome (atypical, due to autoantibody to factor H)
- Wilson disease, fulminant 9
Category II (disorders for which apheresis is accepted as second-line therapy, either as a standalone treatment or in conjunction with other modes of treatment) are as follows:
- Lambert-Eaton myasthenic syndrome
- Multiple sclerosis (acute central nervous system demyelination disease unresponsive to steroids)
- Red blood cell alloimmunization in pregnancy
- Mushroom poisoning
- Acute disseminated encephalomyelitis
- Hemolytic uremic syndrome (atypical, due to complement factor mutations)
- Autoimmune hemolytic anemia (life-threatening cold agglutinin disease)
- Systemic lupus erythematosus (severe)
- Myeloma cast nephropathy
Category III (disorders for which the optimal role of apheresis therapy is not established; decision-making should be individualized) are as follows:
- Posttransfusion purpura
- Autoimmune hemolytic anemia (warm autoimmune hemolytic anemia)
- Hypertriglyceridemic pancreatitis 9
- Thyroid storm
Category IV (disorders in which published evidence demonstrates or suggests apheresis to be ineffective or harmful; institutional review board [IRB] approval is desirable if apheresis treatment is undertaken in these circumstances) are as follows:
- Stiff person syndrome
- Hemolytic uremic syndrome (typical diarrhea-associated)
- Systemic lupus erythematosus (nephritis)
- Immune thrombocytopenia
Plasma apheresis contraindications
Plasma apheresis is contraindicated in the following patients:
- Patients who cannot tolerate central line placement
- Patients who are actively septic or are hemodynamically unstable – Although plasmapheresis can remove various mediators from the blood and so, in theory, would appear to be potentially useful in treating sepsis, septic patients are often coagulopathic and hemodynamically unstable, which poses significant risks when catheters are inserted and connected to a mechanical circuit; there are some ongoing trials evaluating plasmapheresis in sepsis (including two in Germany), but at present, there are no clear data to suggest that the clinical benefits of using plasmapheresis in this setting outweigh the risks associated with its use
- Patients who have allergies to fresh frozen plasma or albumin, depending on the type of plasma exchange
- Patients with heparin allergies should not receive heparin as an anticoagulant during plasmapheresis
- Patients with hypocalcemia are at risk for worsening of their condition because citrate is commonly used to prevent clotting and can potentiate hypocalcemia
- Patients taking angiotensin-converting enzyme (ACE) inhibitors are advised to stop taking the medication for at least 24 hours before starting plasmapheresis
What is therapeutic apheresis?
Apheresis may also be used therapeutically to treat various disorders 1. Therapeutic apheresis includes plasma exchange and cytapheresis
Plasma exchange
Therapeutic plasma exchange removes plasma components from blood. A blood cell separator extracts the patient’s plasma and returns RBCs and platelets in plasma or a plasma-replacing fluid; for this purpose, 5% albumin is preferred to fresh frozen plasma (except for patients with thrombotic thrombocytopenic purpura) because it causes fewer reactions and transmits no infections. Therapeutic plasma exchange resembles dialysis but, in addition, can remove protein-bound toxic substances. A one-volume exchange removes about 65% of such components.
To be of benefit, plasma exchange should be used for diseases in which the plasma contains a known pathogenic substance, and plasma exchange should remove this substance more rapidly than the body produces it. For example, in rapidly progressive autoimmune disorders, plasma exchange may be used to remove existing harmful plasma components (eg, cryoglobulins, antiglomerular basement membrane antibodies) while immunosuppressive or cytotoxic drugs suppress their future production.
There are numerous, complex indications. Clinicians typically follow Guidelines on the Use of Therapeutic Apheresis from the American Society for Apheresis 1. The frequency of plasma exchange, the volume to be removed, the replacement fluid, and other variables are individualized.
Low density lipoprotein cholesterol can be selectively removed from plasma by adsorption over a column (called LDL apheresis) 2.
In photopheresis, mononuclear cells are selectively removed by centrifugation and treated with photoactivatable drugs (eg, 8-methoxypsoralen) that are then activated with ultraviolet light; it is a form of immunomodulatory therapy.
In immunoadsorption, an antibody or antigen is removed from plasma by combining with an antigen or antibody chosen to bind the target antibody or antigen over a column.
Complications of plasma exchange are similar to those of therapeutic cytapheresis.
Cytapheresis
In cytapheresis, the cellular components of blood (eg, red blood cells, white blood cells, platelets) are separated. This is often done on donated blood so that each component may be given to a different recipient. Cytapheresis also may be done therapeutically to remove excess or defective cellular components.
Therapeutic cytapheresis removes cellular components from blood, returning plasma.
It is most often used to remove defective red blood cells and substitute normal ones in patients with sickle cell disease who have the following conditions: acute chest syndrome, stroke, pregnancy, or frequent, severe sickle cell crises. Red blood cell exchange achieves hemoglobin S levels of < 30% without the risk of increased viscosity that can occur because of increased hematocrit with simple transfusion.
Therapeutic cytapheresis may also be used to reduce severe thrombocytosis or leukocytosis (cytoreduction) in acute leukemia or in accelerated or blast crisis phase of chronic myeloid leukemia when there is risk of hemorrhage, thrombosis, or pulmonary or cerebral complications of extreme leukocytosis (leukostasis).
Therapeutic platelet removal (plateletpheresis) is effective in thrombocytosis because platelets are not replaced as rapidly as white blood cells. One or two procedures may reduce platelet counts to safe levels.
Therapeutic white blood cell removal (leukapheresis) can remove kilograms of buffy coat in a few procedures, and it often relieves leukostasis. However, the reduction in white blood cell count itself may be mild and only temporary.
Other uses of cytapheresis include collection of peripheral blood stem cells for autologous or allogeneic bone marrow reconstitution (an alternative to bone marrow transplantation) and collection of lymphocytes for use in immune modulation cancer therapy (adoptive immunotherapy).
What apheresis donation?
Plasma donation volume varies for each donor but the maximum donation volume is 800ml. The volume of plasma a donor can safely donate is calculated based on their gender, height and weight. A plasma donation usually takes between 45 and 60 minutes but donors are advised to set aside 90 minutes to allow for
the interview process beforehand, and to rest and have refreshment afterwards.
During a plasma donation, blood is drawn from the donation arm by a pump into a sterile single use disposable kit inside the cell separating machine. Inside the apheresis machine there is a blood processing chamber that spins (centrifuges) the blood, separating it into layers. Red cells are heaviest and sit at the bottom, platelets and white cells are in the middle and plasma is at the top as it is the lightest.
The apheresis machine is programmed to detect the plasma and divert it into a collection bag with the rest of the blood given back to the donor (red cells, white cells, and platelets). This cycle is repeated until the desired collection volume is achieved.
A small amount of citrate anticoagulant is mixed with each withdrawal of blood to prevent clotting. This is controlled by the cell separator. The anticoagulant works by inactivating calcium, which is required for clotting to occur. The donor will receive a small amount of anticoagulant as it is returned with the components that are not being collected.
The amount to be collected is entered into the machine, by a trained staff member, at the beginning of the donation. The amount can be changed during the donation if required but the cell separator will only collect the amount of plasma entered.
What is platelet apheresis?
In platelet apheresis or plateletpheresis or platelet donation, a donor gives only platelets rather than whole blood. Whole blood is drawn from the donor, and a apheresis machine that separates the blood into its components selectively removes the platelets and returns the rest of the blood to the donor. Because donors get most of their blood back, they can safely give 8 to 10 times as many platelets during one of these procedures as they would give in a single donation of whole blood. They can also give platelets more often, once every 3 days (but no more than 24 donations during a year). Collecting platelets from a donor takes about 1 to 2 hours, compared with collecting whole blood, which takes about 10 minutes.
Apheresis procedure
Blood is taken by inserting an intravenous (IV) needle into a vein. The blood is processed in a machine. Then the blood goes back into the body through another IV. If it’s not possible to use an IV, a central venous catheter may be used instead. It’s a thin, flexible tube that goes into a large vein in the neck or chest.
The treatment takes about 2 to 5 hours, depending on which type of apheresis you get. Your doctor may have you go to the bathroom first, as once you’re attached to the machine you can’t leave it until the procedure is finished.
You may need only one treatment. Or you may need many treatments over weeks or months. It depends on what type of apheresis is used and what is being treated.
Apheresis side effects
Apheresis is generally tolerated by healthy donors. However, some people have side effects from apheresis. These may include an allergic reaction, fatigue, nausea, dizziness, or low blood pressure. You may feel numbness, tingling, and itching. Most side effects will stop when the treatment ends. You may feel tired for a few days.
You may get calcium supplements during the procedure to help with numbness or tingling.
If you need a central venous catheter, your doctor will talk to you about the risks. There is a low risk of things such as bleeding, infection, lung puncture, and an air bubble blocking a blood vessel (air embolism).
Minor and a few major risks:
- Insertion of the large IV catheters necessary for apheresis can cause complications (eg, bleeding, infection, pneumothorax).
- Citrate anticoagulant may decrease serum ionized calcium.
- Replacement of patient’s plasma with a colloidal solution (eg, 5% albumin or fresh frozen plasma does not replace IgG and coagulation factors.
Most complications can be managed with close attention to the patient and manipulation of the procedure, but some severe reactions and a few deaths have occurred.
References- Schwartz J, Padmanabhan A, Aqui N, et al: Guidelines on the Use of Therapeutic Apheresis in Clinical Practice–Evidence-Based Approach from the Writing Committee of the American Society for Apheresis: The Seventh Special Issue. J Clin Apheresis 31: 149–338, 2016. doi:10.1002/jca.21470
- Feingold K, Grunfeld C. Lipoprotein Apheresis. [Updated 2017 Feb 21]. In: Feingold KR, Anawalt B, Boyce A, et al., editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK425700
- [Guideline] Schwartz J, Padmanabhan A, Aqui N, Balogun RA, Connelly-Smith L, Delaney M, et al. Guidelines on the Use of Therapeutic Apheresis in Clinical Practice-Evidence-Based Approach from the Writing Committee of the American Society for Apheresis: The Seventh Special Issue. J Clin Apher. 2016 Jun. 31 (3):149-62.
- Siami GA, Siami FS. Membrane plasmapheresis in the United States: a review over the last 20 years. Ther Apher. 2001 Aug. 5(4):315-20.
- Gerhardt RE, Ntoso KA, Koethe JD, Lodge S, Wolf CJ. Acute plasma separation with hemodialysis equipment. J Am Soc Nephrol. 1992 Mar. 2(9):1455-8
- Gurland HJ, Lysaght MJ, Samtleben W, Schmidt B. A comparison of centrifugal and membrane-based apheresis formats. Int J Artif Organs. 1984 Jan. 7(1):35-8
- Agishi T, Kaneko I, Hasuo Y, Hayasaka Y, Sanaka T, Ota K, et al. Double filtration plasmapheresis. 1980. Ther Apher. 2000 Feb. 4(1):29-33
- Siami GA, Siami FS. Current topics on cryofiltration technologies. Ther Apher. 2001 Aug. 5(4):283-6
- Chang CT, Tsai TY, Liao HY, Chang CM, Jheng JS, Huang WH, et al. Double Filtration Plasma Apheresis Shortens Hospital Admission Duration of Patients With Severe Hypertriglyceridemia-Associated Acute Pancreatitis. Pancreas. 2016 Apr. 45 (4):606-12