Condylomata acuminata
Condylomata acuminata is a medical term for anogenital warts or venereal warts, which are growths on the cervical or vulval mucosa in females, or on the glans of penis or prepuce in males. Anogenital warts or genital warts are a sexually transmitted disease (STD) caused by the human papillomavirus (HPV), which has over 100 different strains. Subtypes number 6 and 11 cause 90% of genital warts and are considered low risk because they very rarely will cause genital or anal cancer 1. On the other hand, subtypes 16 and 18, for example, are considered high risk because, although they rarely cause genital warts, they can lead to cervical or anal precancer and cancer. The warts usually appear as a small bump or group of bumps in the genital area. They are flesh-colored and can be flat or look bumpy like cauliflower. Some genital warts are so small you cannot see them. In women, genital warts usually occur in or around the vagina, on the cervix, or around the anus. In men, genital warts are less common. They may have warts on the tip of the penis, around the anus, or on the scrotum, thigh, or groin.
You can get genital warts during oral, vaginal, or anal sex with an infected partner. HPV is also spread by skin-to-skin contact during sexual activity; there does not need to be vaginal or anal intercourse to spread the infection. Correct usage of latex condoms greatly reduces, but does not completely eliminate, the risk of catching or spreading human papillomavirus (HPV). The most reliable way to avoid HPV infection is to not have anal, vaginal, or oral sex. HPV vaccines may help prevent some of the HPV infections that cause genital warts.
Since anogenital warts are not a reportable disease, the incidence of condyloma acuminata is difficult to estimate. However, recent estimates report the incidence of anogenital warts in the United States at 1.1 to 1.2 cases per 1000 person-years 2.
Most people who become infected with HPV will not have symptoms and will clear the infection on their own. Your health care provider usually diagnoses genital warts by seeing them. For people who do develop genital warts, there are many options for treatment, all of which are meant to remove the visible warts. There is no cure for genital warts, the human papillomavirus (HPV) stays in your body even after treatment, so warts can come back.
There are several topical treatment options available, including podophyllotoxin solutions and creams, imiquimod cream, and sinecatechins ointment. Cryotherapy, trichloroacetic acid solution, and several surgical modalities are also available treatments. There is a chance for condyloma acuminata to recur after topical treatments. Surgical excision is the only available treatment with clearance rates close to 100 percent 1. Patients should receive counsel on treatment options, the importance of follow up appointments, and safe sex practices.
In the United States three HPV vaccines are licensed: Ceravrix, a 2-valent vaccine (2vHPV) that targets HPV types 16 and 18; Gardasil, a 4-valent vaccine (4vHPV) that targets HPV types 6, 11, 16, and 18; and Gardasil 9, a 9-valent vaccine (9vHPV) that targets HPV types 6, 11, 16, 18, 31, 33, 45, 52, and 58. Types 16 and 18 account for 65-70% of all cervical cancers, whereas the five additional types targeted by the 9-valent vaccine account for 10% 3. HPV types 6 and 11 cause >90% of genital warts. Only Gardasil 9 (9vHPV) vaccine is available in the United States 4.
- Gardasil, a 4-valent vaccine (4vHPV) that protects against the 4 strains of HPV (HPV types 6, 11, 16, and 18) that cause 70% of cervical cancers and 90% of genital warts. In the US, Gardasil is approved for girls/women and boys/men ages 9–26. In 2011, Health Canada approved the vaccine for women up to the age of 45.
- Gardasil 9, a 9-valent vaccine (9vHPV) that targets HPV types 6, 11, 16, 18, 31, 33, 45, 52, and 58, is highly effective in preventing urogenital cancers and anogenital warts.
Condylomata acuminata (anogenital warts) key points
- Anogenital warts are a recognized symptom of genital HPV infections.
- About 90% of those exposed who contract HPV will not develop genital warts.
- Only about 10% who are infected may transmit the virus.
- HPV types 6 and 11 cause genital warts. There are over 100 different known types of HPV viruses.
- HPV is spread through direct skin-to-skin contact with an infected individual, usually during sex.
- While some types of HPV cause cervical and anal cancer, these are not the same viral types that cause genital warts.
- It is possible to be infected with different types of HPV at the same time.
- There is conflicting evidence about the effect of condoms on prevention.
- Approximately three out of four unaffected partners of patients with warts develop them within eight months of contact.
- Although 90% of HPV infections are cleared within two years of infection, it is possible for a latency period to occur, with the first occurrence or a recurrence happening months or even years later.
- Latent HPV is transmissible, and if an individual has unprotected sex with an infected partner, there is a 70% chance they will become infected.
- In individuals with a prior HPV infection, the appearance of new warts may be either from a new exposure or a recurrence.
- Anal or genital warts may be transmitted during birth and may be an indicator of sexual abuse.
- Genital warts may sometimes result from autoinoculation by warts elsewhere on the body, such as from the hands.
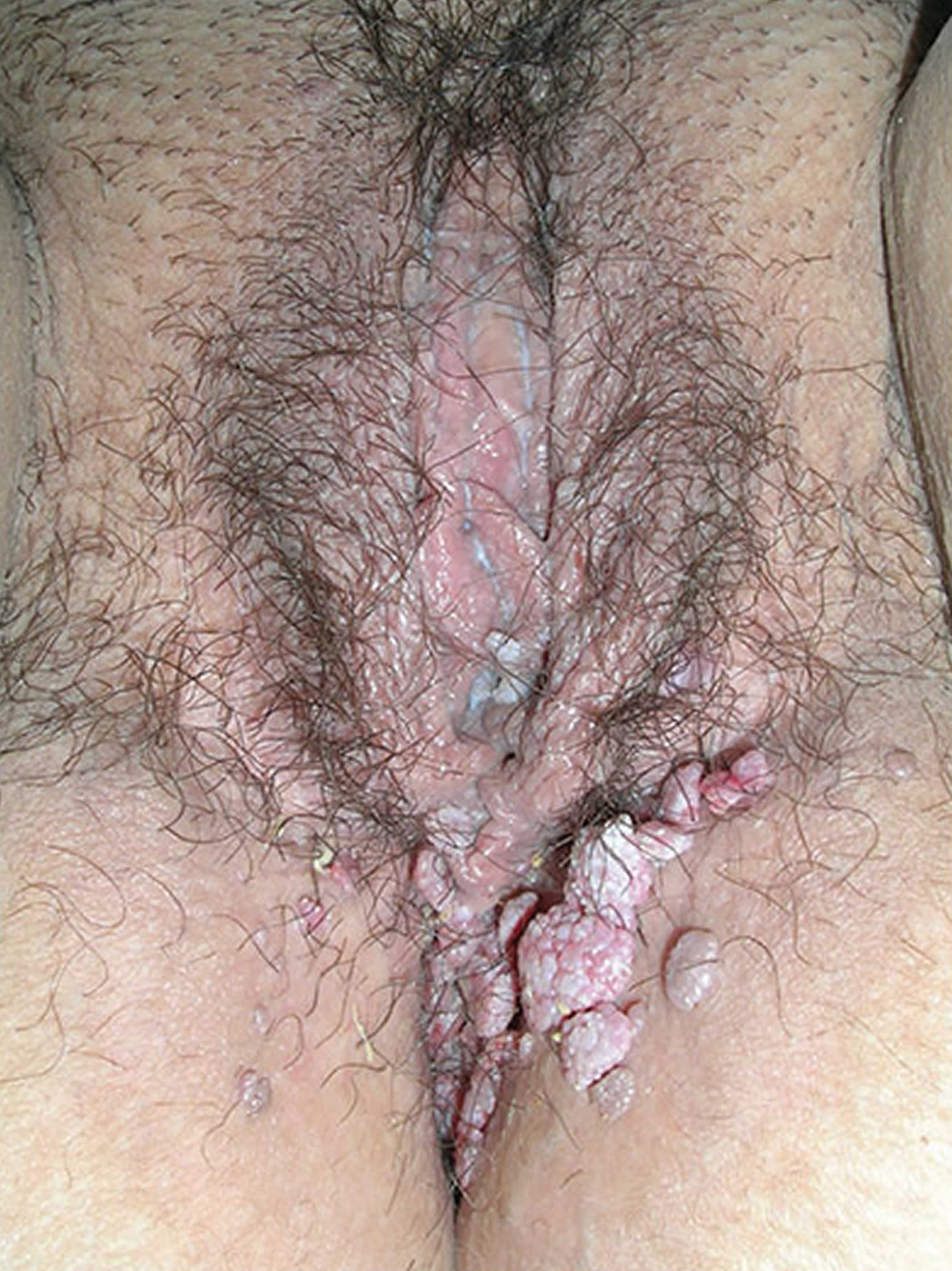
Figure 1. Condylomata acuminata in female
Footnote: Genital warts are a common sexually transmitted infection. They can appear on the genitals, in the pubic area or in the anal canal. In women, genital warts can also grow inside the vagina.
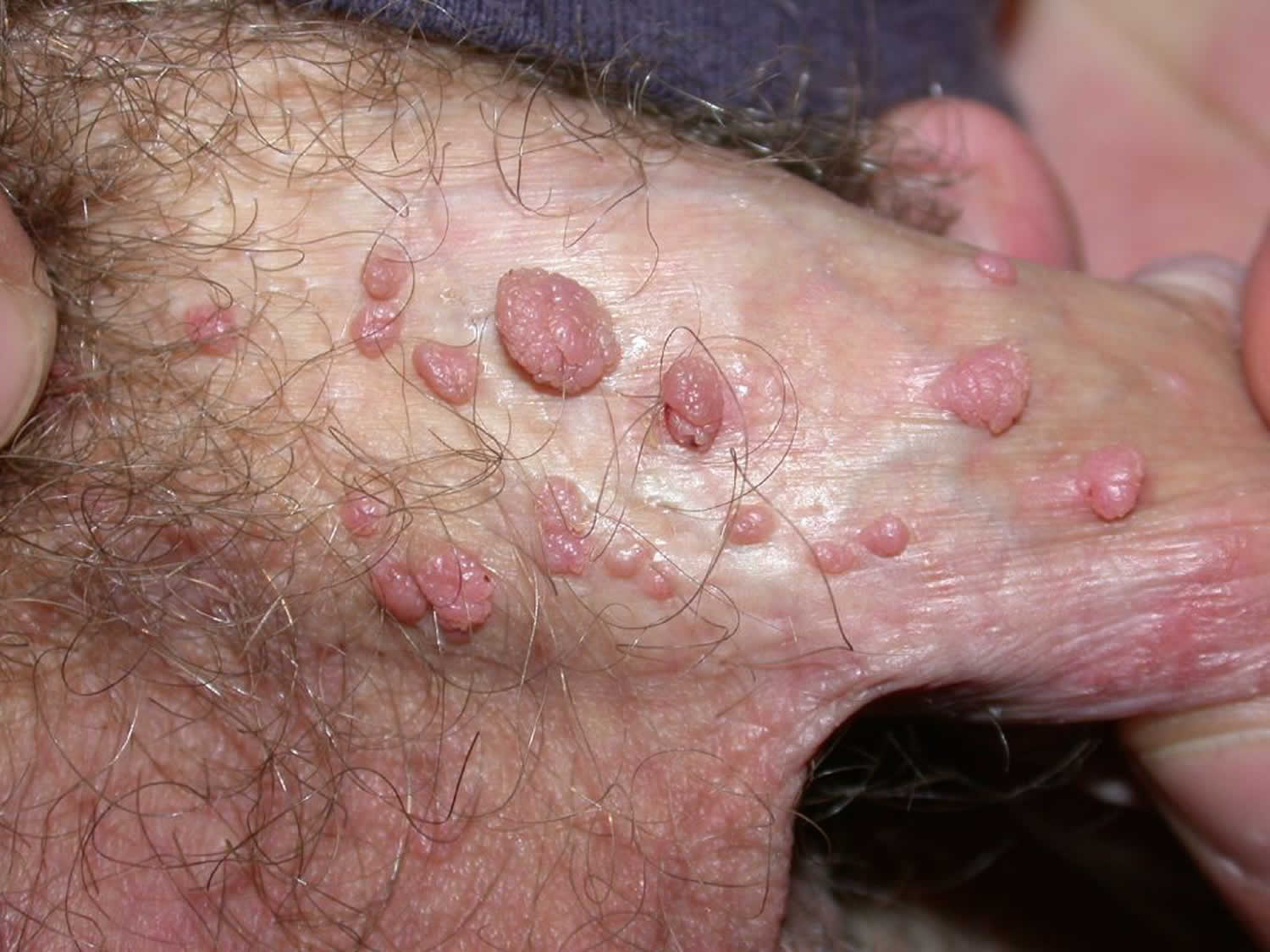
Figure 2. Condylomata acuminata in male
See a doctor or go to a STD clinic if you or your partner develops bumps or warts in the genital area. Genital warts affect the moist tissues of the genital area. They can look like small, flesh-colored bumps or have a cauliflower-like appearance. In many cases, the warts are too small to be visible.
- 1 or more painless growths or lumps around your vagina, penis or anus
- itching or bleeding from your genitals or anus
- a change to your normal flow of pee (for example, it’s begun to flow sideways) that does not go away
- a sexual partner who has genital warts, even if you do not have symptoms
These symptoms mean you may have genital warts. Go to a sexual health clinic to be checked.
Sexual health clinics are sometimes called STD clinics, or sexual and reproductive health services.
Treatment can help remove the warts and stop the infection being passed on.
What is the human papillomavirus?
The human papillomavirus (HPV) is actually a group of double-stranded DNA viruses.
- There are at least 100 different types of HPV; at least 40 can infect the anogenital area. Many others cause warts on other areas of skin.
- At least 75% of sexually active adults have been infected with at least one type of anogenital HPV at some time in their life.
- HPV is incorporated into skin cells and stimulates them to proliferate, causing a visible wart.
- Visible anogenital warts are often easy to diagnose by their typical appearance. They are usually due to HPV types 6 and 11.
- Most do not develop visible warts. However, the infection may show up on a cervical smear. This is known as subclinical infection.
- Some strains of HPV cause anogenital cancer. These strains may not cause visible warts but they remain contagious.
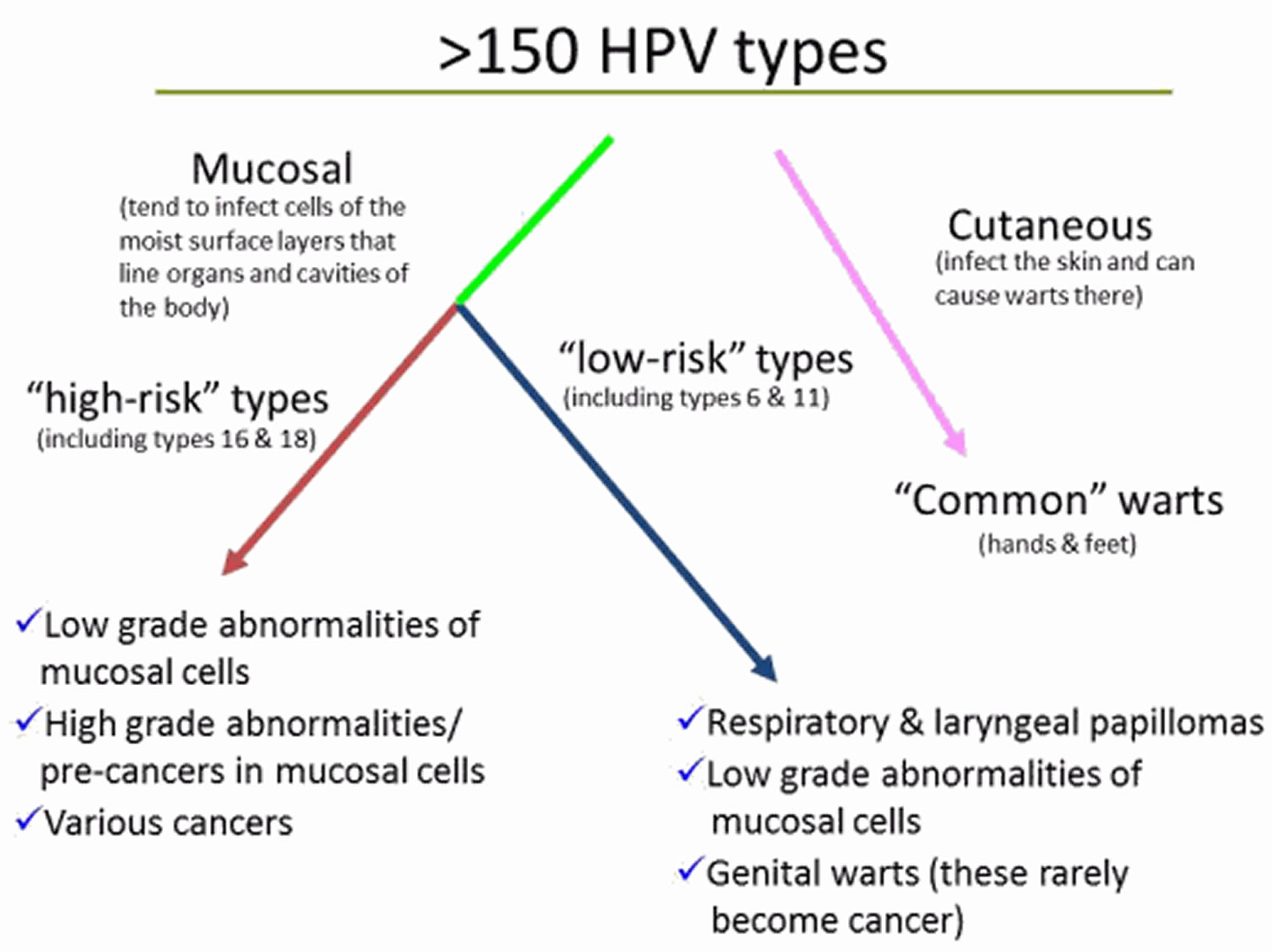
Figure 1. HPV types
How is human papillomavirus transmitted?
Visible genital warts and subclinical human papillomavirus (HPV) infection nearly always arise from direct skin to skin contact.
- Sexual contact. This is the most common way amongst adults.
- Transmission is more likely from visible warts than from subclinical HPV infection.
- Oral sex. HPV appears to prefer the genital area to the mouth however.
- Vertical (mother to baby) transmission through the birth canal.
- Auto (self) inoculation from one site to another.
- Fomites (ie from objects like bath towels). It remains very controversial whether warts can spread this way.
Often, warts will appear three to six months after infection but they may appear months or even years later.
Who’s at risk of condylomata acuminata?
Genital warts are the most common sexually transmitted disease and affect millions of people throughout the world. It is estimated that 75–80% of sexually active men and women will be infected with HPV at some point in their lives. As anogenital warts are sexually acquired during close skin contact, they are most commonly observed in young adults between the ages of 15 and 30 years. They are highly contagious, and occur in equal numbers in unvaccinated males and females. Approximately 15% of the United States population is infected with HPV. HPV infection occurs in people of all ages and both sexes. However, they are rare in people that have been vaccinated against HPV in childhood before beginning sexual activity. Your risk of acquiring the virus is higher if you have had many sexual partners and if you first had sexual intercourse at a young age. Over 50% of girls will get HPV within 2 years of becoming sexually active.
HPV is spread through skin-to-skin contact and does not require actual intercourse to be passed from one person to another. A person can be infected without any visible signs of infection and, therefore, can pass the infection on without knowing it. Condoms can decrease the risk of spreading the virus, but they do not completely prevent transmission.
Condylomata acuminata causes
Condyloma acuminata results from specific human papillomavirus (HPV) types, most commonly HPV types 6 and 11. HPV is the most common sexually transmitted infection worldwide, with 9 to 13 percent of the global population infected 5. Patients who are between 20 and 39 years of age are most commonly affected 6. Anogenital warts will usually appear three to six months after infection but they may appear months or even years later. There are several known risk factors for acquiring HPV. Prevalence of infection increases with an increased number of lifetime sexual partners, a history of chlamydia and gonorrhea infections, smoking, and human immunodeficiency virus (HIV) infection 7.Transmission rates of HPV between sex partners are high, and transmission often occurs from persons with HPV who are asymptomatic or have subclinical infection 8, 9. Consistent and correct use of condoms reduces the risk for genital HPV acquisition or transmission, and therefore reduces the risk for HPV-associated diseases 10. Condom use, however, does not entirely prevent transmission of HPV, since exposure to HPV can occur in areas that are not fully covered or protected by a condom 11. Treatment of warts or cervical cellular abnormalities will reduce, but not eliminate, the risk of transmission. Transmission of HPV through nonsexual routes can occur, but is uncommon 12, 13. Mothers with low-risk genital HPV types can rarely transmit HPV to their newborn during delivery, but if transmission does occur, HPV has the potential to cause recurrent respiratory papillomatosis in children 14, 15.
There have been over 200 types of HPV identified, with 40 strains known to affect the anogenital area. HPVs are called papillomaviruses because some HPV types cause papillomas (warts), which are non-cancerous tumors. Each HPV virus is given a number, which is called an HPV type. Figure 1 above shows the different groups of HPV types and the problems each group can cause.
HPV subtypes number 6 and 11 cause 90% of condyloma acuminata (genital warts) and are considered low risk because they very rarely will cause genital or anal cancer 3, 16, 17, 18. On the other hand, HPV subtypes 16 and 18, for example, are considered high risk because, although they rarely cause genital warts, they can cause cancer, including cancers of the cervix (the base of the womb at the top of the vagina), vagina, vulva (the area around the outside of the vagina), penis, anus, and parts of the mouth and throat 11, 19, 5. Approximately 65-70% of cervical cancers are caused by HPV types 16 or 18 20. In addition, about 10% of HPV-related cancers are caused by HPV types 31, 33, 45, 52, and 58 3. In the United States, approximately 45,000 HPV-related cancers are diagnosed each year, with cancer of the cervix the most common among women and cancer of the throat (oropharynx) the most common in men 19.
Genital warts are almost always spread through sexual activity. Your warts don’t have to be visible for you to spread the infection to your sexual partner.
- Sexual contact. This is the most common way amongst adults. Transmission is more likely from visible warts than from subclinical or latent HPV infection.
- Oral sex. HPV appears to prefer the genital area to the mouth.
- Vertical (mother to baby) transmission through the birth canal.
- Auto (self) inoculation from one site to another.
As anogenital warts are usually sexually acquired, they are most commonly observed in young adults between the ages of 15 and 30 years. They are highly contagious. However, anogenital warts are rare in people who have been vaccinated against the benign HPV types in childhood before beginning sexual activity. Anogenital warts have been reported in a number of studies to be more common in males than females.
Patients who are immunocompromised due to drug-induced immunosuppression or HIV infection are at particular risk of acquiring HPV and developing anogenital warts.
Anogenital warts can also affect infants and young children. The virus may be acquired during birth or from the hands of carers.
Risk factors for getting condyloma acuminata (genital warts)
Most people who are sexually active get infected with genital HPV at some time. Factors that can increase your risk of becoming infected include:
- Having unprotected sex with multiple partners
- Having had another sexually transmitted infection
- Having sex with a partner whose sexual history you don’t know
- Becoming sexually active at a young age
- Having a compromised immune system, such as from HIV or drugs from an organ transplant
Pathophysiology
HPV is a double-stranded DNA virus that primarily infects the nucleus of differentiated squamous epithelial cells. The DNA virus can remain in a latent phase for several months, resulting in an incubation period of one month to two years 21. The genome of HPV contains oncogene, which encodes proteins that stimulate cell proliferation. These proteins enable the virus to replicate via the host cell’s DNA polymerase while the host cells undergo cell division. As the number of virally infected host cells grows, the basal, spinous, and granular layers of the epidermis thicken, leading to acanthosis and the macroscopic appearance of warts. Condyloma acuminata generally take three to four months to form. In otherwise healthy individuals, an adequate immune response can halt viral replication and resolve the infection over time. However, prolonged HPV infection increases the risk of developing malignant transformation 22.
Condylomata acuminata prevention
Limiting your number of sexual partners and being vaccinated will help prevent you from getting genital warts. Using a condom every time you have sex is a good idea, but won’t necessarily protect you from genital warts.
HPV vaccination
The Centers for Disease Control and Prevention (CDC) recommends routine HPV vaccination for girls and boys ages 11 and 12, although it can be given as early as age 9. It’s ideal for girls and boys to receive the HPV vaccine before they have sexual contact 4.
Side effects from the HPV vaccines are usually mild and include soreness at the injection site, headaches, a low-grade fever or flu-like symptoms.
The CDC now recommends that all 11- and 12-year-olds receive two doses of HPV vaccine at least six months apart, instead of the previously recommended three-dose schedule. Younger adolescents ages 9 and 10 and teens ages 13 and 14 also are able to receive vaccination on the updated two-dose schedule. Research has shown that the two-dose schedule is effective for children under 15.
Teens and young adults who begin the vaccine series later, at ages 15 through 26, should continue to receive three doses of the vaccine.
The CDC now recommends catch-up HPV vaccinations for all people through age 26 who aren’t adequately vaccinated.
The U.S. Food and Drug Administration (FDA) recently approved the use of Gardasil 9 for males and females ages 9 to 45. If you’re ages 27 to 45, discuss with your doctor whether he or she recommends that you get the HPV vaccine.
In the United States three HPV vaccines are licensed: Ceravrix, a 2-valent vaccine (2vHPV) that targets HPV types 16 and 18; Gardasil, a 4-valent vaccine (4vHPV) that targets HPV types 6, 11, 16, and 18; and Gardasil 9, a 9-valent vaccine (9vHPV) that targets HPV types 6, 11, 16, 18, 31, 33, 45, 52, and 58. Types 16 and 18 account for 65-70% of all cervical cancers, whereas the five additional types targeted by the 9-valent vaccine account for 10% 3. HPV types 6 and 11 cause >90% of genital warts. Since late 2016, only Gardasil 9 (9vHPV) vaccine is available in the United States 4.
- Gardasil, a 4-valent vaccine (4vHPV) that protects against the 4 strains of HPV (HPV types 6, 11, 16, and 18) that cause 70% of cervical cancers and 90% of genital warts. In the US, Gardasil is approved for girls/women and boys/men ages 9–26. In 2011, Health Canada approved the vaccine for women up to the age of 45.
- Gardasil 9, a 9-valent vaccine (9vHPV) that targets HPV types 6, 11, 16, 18, 31, 33, 45, 52, and 58, is highly effective in preventing urogenital cancers and anogenital warts.
HPV vaccine may be available at doctor offices, community health clinics, school-based health centers, and health departments.
Who should NOT get HPV vaccine?
Tell your doctor about any severe allergies. Some people should NOT get some HPV vaccines if:
- They have ever had a life-threatening allergic reaction to any ingredient of an HPV vaccine, or to a previous dose of HPV vaccine.
- They have an allergy to yeast (Gardasil and Gardasil 9).
- They are pregnant.
HPV vaccines are safe for children who are mildly ill, like those with a low-grade fever of less than 101 degrees, a cold, runny nose, or cough. People with a moderate or severe illness should wait until they are better.
Condylomata acuminata symptoms
Condylomata acuminata or genital warts appear as flesh-colored, round bumps of varying sizes (few millimeters in diameter). They can be smooth and flat or cauliflower-like appearance with a small stalk. Warts may join together to form plaques up to several centimeters across. They can be seen on the labia, vagina, penis, scrotum, anus, skin around the anus, and urethra. Warts usually do not cause any symptoms, although the warts can bleed and become painful with intercourse if they are located within the vagina. A linear pattern may be seen if the HPV virus has been inoculated along a scratch
Condylomata acuminata or anogenital warts may occur in the following sites:
- Vulva
- Vagina
- Cervix
- Urethra
- Penis
- Scrotum
- Anus.
In women, genital warts can grow on the vulva, the walls of the vagina, the area between the external genitals and the anus, the anal canal, and the cervix. In men, they may occur on the tip or shaft of the penis, the scrotum, or the anus.
Genital warts can also develop in the mouth, throat, around the lips and conjunctiva of the eye of a person who has had oral sexual contact with an infected person.
The signs and symptoms of genital warts include:
- Small, flesh-colored, brown or pink swellings in your genital area
- A cauliflower-like shape caused by several warts close together
- Itching or discomfort in your genital area
- Bleeding with intercourse
Genital warts can be so small and flat as to be invisible. Rarely, however, genital warts can multiply into large clusters in someone with a suppressed immune system.
Normal anatomical structures may be confused with warts. These include:
- Pearly papules (these are in a ring around the glans of the penis )
- Sebaceous glands on the labia (known as “Fordyce spots”)
- Vestibular papillae (the fronds found in the opening to the vagina).
Condylomata acuminata complications
Condyloma acuminata or anogenital warts are contagious and easily spread to your sexual partners. Anogenital warts can impact psychosexual functioning and quality of life.
HPV infection complications can include:
- Cancer. Cervical cancer has been closely linked with genital HPV infection. Certain types of HPV also are associated with cancers of the vulva, anus, penis, and mouth and throat. HPV infection doesn’t always lead to cancer, but it’s important for women to have regular Pap tests, particularly those who’ve been infected with higher risk types of HPV.
- Problems during pregnancy. Rarely during pregnancy, warts can enlarge and multiply, making it difficult to urinate. Warts on the vaginal wall can inhibit the stretching of vaginal tissues during childbirth. Large warts on the vulva or in the vagina can bleed when stretched during delivery. Extremely rarely, a baby born to a mother with genital warts develops warts in the throat. The baby might need surgery to keep the airway from being blocked. HPV can be transmitted to the baby resulting in recurrent respiratory papillomatosis in the infant.
Condyloma acuminata can have significant psychological effects on patients, including anxiety, guilt, and anger. Patients may also have concerns about the loss of fertility and cancer. Premalignant and malignant lesions can be present within genital warts. Condyloma acuminata can also develop into malignant lesions. Concerning signs for malignant transformation include bleeding, irregular pigmentation, ulceration, and lesions with palpable dermal infiltration 23. Also, as previously mentioned, Buschke-Lowenstein tumors are a known complication of condyloma acuminata 24.
Condylomata acuminata and cancer
The HPV types that cause external visible warts (HPV Types 6 and 11) rarely cause cancer. Other HPV types (most often Types 16, 18, 31, 33 and 35) are less common in visible warts but are strongly associated with anogenital cancer, including:
Squamous intraepithelial lesion of penile, vulval and anal skin
Invasive squamous cell carcinoma (SCC) of cervix, penis, vulva and anus
HPV also causes some cases of oral and nasopharyngeal cancer.
Only a small percentage of infected people develop genital cancer. This is because HPV infection is only one factor in the process; cigarette smoking and how well the immune system is working are also important.
Cervical smears, as recommended in the National Cervical Screening guidelines, detect squamous intraepithelial lesions of the cervix, which can be treated. If these abnormalities were ignored over a long period, they could progress to cervical cancer.
Condylomata acuminata diagnosis
Genital warts are usually diagnosed clinically by their appearance.
Skin biopsy is sometimes necessary to confirm the diagnosis of viral wart, particularly if there is concern of anogenital cancer.
In some circumstances, researchers and clinicians may wish to confirm the presence or absence of HPV. One commercially available qualitative test for HPV is the COBAS 4800 Human Papillomavirus (HrHPV) Test, which evaluates 14 high-risk (HR oncogenic) HPV types.
Pap tests
For women, it’s important to have regular pelvic exams and Pap tests, which can help detect vaginal and cervical changes caused by genital warts or the early signs of cervical cancer.
During a Pap test, your doctor uses a device called a speculum to hold open your vagina and see the passage between your vagina and your uterus (cervix). He or she will then use a long-handled tool to collect a small sample of cells from the cervix. The cells are examined with a microscope for abnormalities.
HPV test
Only a few types of genital HPV have been linked to cervical cancer. A sample of cervical cells, taken during a Pap test, can be tested for these cancer-causing HPV strains.
This test is generally reserved for women age 30 and older. It isn’t as useful for younger women because for them, HPV usually goes away without treatment.
Condylomata acuminata differential diagnosis
- Condyloma lata
- Molluscum contagiosum
- Lichen planus
- Psoriasis
- Malignancy
- Pearly penile papules
- Acrochordon
- Sebaceous cysts
- Buschke-Lowenstein tumor 25.
Condylomata acuminata treatment
There is no cure for HPV 26. The goal of treatment is to remove visible warts and decrease the risk of spreading the virus 27. However, available therapies reduce but do not completely eliminate the risk of HPV transmission 28. In addition, there are no data that suggest treatment of genital warts impacts the subsequent risk of anogenital HPV-related cancer 28. Although appropriate treatment can induce wart-free periods, subsequent recurrences are common, with a 20 to 50% recurrence rate at 3 to 6 months post-treatment 29. Reappearance of genital warts within the first several months usually indicates recurrence rather than reinfection. If left untreated, visible genital warts persist with or without rapid increase in the number, but some regress spontaneously. Because spontaneous resolution of anogenital warts can occur, some individuals may choose to defer treatment and await possible spontaneous resolution 28. In addition, there are no data that suggest treatment of genital warts impacts the subsequent risk of anogenital HPV-related cancer 28.
If your warts aren’t causing discomfort, you might not need treatment. But if you have itching, burning and pain, or if you’re concerned about spreading the infection, your doctor can help you clear an outbreak with medications or surgery. However, warts often return after treatment. There is no treatment for the HPV virus itself.
The primary goal of treatment is to eliminate warts that cause physical or psychological symptoms such as:
- Pain
- Bleeding
- Itch
- Embarrassment.
Options include:
- No treatment
- Self-applied treatments at home
- Treatment at a doctor’s surgery or medical clinic.
The underlying viral infection may persist after the visible warts have cleared. Warts sometimes re-emerge years later because the immune system has weakened.
Self-applied treatments
To be successful you must identify and reach the warts, and follow the application instructions carefully. Available treatments include:
- Podophyllotoxin 0.5% gel or solution (Condylox®). This should not be used during pregnancy.
- Imiquimod 3.75% cream (Zyclara). Imiquimod cream appears to boost your immune system’s ability to fight genital warts. Apply once daily for up to 8 weeks. Avoid sexual contact while the cream is on your skin. It might weaken condoms and diaphragms and irritate your partner’s skin. One possible side effect is skin redness. Other side effects might include blisters, body aches or pain, a cough, rashes, and fatigue.
- Imiquimod 5% cream (Aldara). Apply once daily (at bedtime) 3 times a week for up to 16 weeks.
- Sinecatechins (Veregen). Sinecatechins cream is used for treatment of external genital warts and warts in or around the anal canal. Sinecatechins 15% ointment should be administered 3 times/day using a finger to apply a 0.5 cm strand of ointment to each anogenital wart; the goal is to cover the wart entirely with a thin layer of the sinecatechins. The ointment should not be washed off after application. Genital, anal, and oral sexual contact should be avoided while sinecatechins ointment is present on the skin. This treatment can be administered for up to 16 weeks. Treatment with sinecatechins is not recommended for persons with genital herpes or for immunocompromised persons, including those with HIV. Side effects, such as reddening of the skin, itching or burning, and pain, are often mild.
Don’t try to treat genital warts with over-the-counter wart removers. These medications aren’t intended for use in the genital area.
Treatment at the clinic
Genital wart treatments that can be applied directly to your skin by a physician:
- Imiquimod (Aldara, Zyclara). This cream appears to boost your immune system’s ability to fight genital warts. Avoid sexual contact while the cream is on your skin. It might weaken condoms and diaphragms and irritate your partner’s skin. One possible side effect is skin redness. Other side effects might include blisters, body aches or pain, a cough, rashes, and fatigue.
- Podophyllin and podofilox (Condylox). Podophyllin is a plant-based resin that destroys genital wart tissue. Your doctor applies 15–25% solution. Podofilox contains the same active compound, but you can apply it at home. Never apply podofilox internally. Additionally, this medication isn’t recommended for use during pregnancy. Podophyllin resin has been associated with multiple reports of systemic toxicity when this agent was applied to large lesions or when left on for longer periods than recommended 30. Side effects can include mild skin irritation, sores or pain.
- Trichloroacetic acid (TCA) or bichloroacetic acid (BCA) 80%–90% solution: The application of trichloroacetic acid or bichloroacetic acid 80-90% solution should only be performed by an individual with training in this application and for treatment of small lesions and can be used for internal warts. These acid agents destroy wart tissue by causing chemical coagulation of proteins. Only a small amount of the solution should be applied to the wart and the solution should dry before the patient moves. If too much is applied or the patient experiences pain, sodium bicarbonate (i.e. baking soda) can be applied to neutralize the acid; the sodium bicarbonate is typically applied as a liquid soap preparation or powdered with talc. Side effects can include mild skin irritation, sores or pain.
- Freezing with liquid nitrogen (cryotherapy). Liquid nitrogen is used to freeze the warts. Freezing works by causing a blister to form around your wart. As your skin heals, the lesions slough off, allowing new skin to appear. You might need to repeat the treatment. The main side effects include pain and swelling.
- Surgical removal. This may need to be performed by a specialist with local anesthesia; usually for large amounts of warts and you might have pain afterward. You might need surgery to remove larger warts, warts that don’t respond to medications or, if you’re pregnant, warts that your baby can be exposed to during delivery.
- Laser treatment. Carbon dioxide laser treatment is useful for a large amount of urethral or vaginal warts. Side effects can include scarring and pain.
- Electrocautery. This procedure uses an electrical current to burn off warts. You might have some pain and swelling after the procedure.
Most of these treatments require multiple applications. If there is no improvement after 3 treatment cycles or if the warts are not gone after 6 treatment cycles, your doctor will usually switch you to a different medication. Common side effects of all treatments include skin irritation and redness. Some people may also develop scars at the site of the warts.
Experimental therapies for genital warts include:
- Interferon
- 5-fluorouracil/epinephrine-gel implant. A 5% 5-fluorouracil (5-FU) cream is no longer considered acceptable due to the side effects.
- Cidofovir
- Application of 5-aminolevulinic acid (ALA) with photodynamic therapy is an emerging treatment for condyloma acuminata. This treatment modality was found to be more effective, simpler, and have lower recurrence rates than CO2 laser treatment. Photodynamic therapy with 5-aminolevulinic acid may be a useful adjuvant to other traditional treatment modalities 31.
Condyloma acuminata during pregnancy
Genital warts can proliferate rapidly and become more friable during pregnancy. Watchful waiting is acceptable with smaller lesions. During pregnancy, cryotherapy, trichloroacetic acid, bichloroacetic acid, and surgical removal may be safely used 27. Cytotoxic agents (podophyllin, podofilox, imiquimod) should not be used during pregnancy 27. Although pregnant women with genital warts have a risk of transmitting HPV to their child during a vaginal birth, Cesarean section delivery should not be performed solely to prevent transmission to the neonate, since it is unclear whether Cesarean delivery prevents respiratory papillomatosis among infants 27.
Condyloma acuminata in immunocompromised patients
The general approach to the treatment of genital warts in persons with HIV is the same as for those without HIV 27. Persons with HIV, particularly those individuals with advanced immunosuppression, often have larger or more numerous warts that do not respond as well to therapy, and recurrences occur more frequently after treatment. High-grade squamous intraepithelial lesions (HSIL) and invasive cancer arising within the region of a genital wart (or resembling genital warts) are more frequent in immunocompromised patients; therefore, hyperpigmented lesions and lesions that persist despite treatment (especially in the perianal area) should be promptly evaluated by biopsy. The role of genital warts (or irritated post-treatment sites) in HIV transmission has not been well characterized.
Condylomata acuminata prognosis
While condyloma acuminata can be challenging to treat with the lesions requiring multiple treatments or a combination of treatments over time. Surgical excision is the only treatment option with clearance rates near 100 percent, although condyloma acuminata can recur after all treatment types. In fact, recurrence of lesions after clearance is seen 20 to 30 percent of the time. The recurrence rates for genital warts treated with topical podophyllotoxin treatments have a clearance rate of 45 to 83 percent and a recurrence rate of 6 to 100 percent. Imiquimod treatments have a lower rate of recurrence at 6 to 26 percent and a clearance rate of 35 to 68 percent. Sinecatechins ointment clears 47 to 59 percent of condyloma acuminata, and only 7 to 11 percent of patients have a recurrence of lesions after clearance. Cryotherapy with liquid nitrogen clears 44 to 75 percent of genital warts with a 21 to 42 percent recurrence rate. Treatment with trichloroacetic acid solution clears 56 to 81 percent of lesions and 36 percent of lesions recur after clearance 23.
References- Pennycook KB, McCready TA. Condyloma Acuminata. [Updated 2019 Oct 9]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2020 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK547667
- Park IU, Introcaso C, Dunne EF. Human Papillomavirus and Genital Warts: A Review of the Evidence for the 2015 Centers for Disease Control and Prevention Sexually Transmitted Diseases Treatment Guidelines. Clin. Infect. Dis. 2015 Dec 15;61 Suppl 8:S849-55.
- Schiffman M, Doorbar J, Wentzensen N, de Sanjosé S, Fakhry C, Monk BJ, Stanley MA, Franceschi S. Carcinogenic human papillomavirus infection. Nat Rev Dis Primers. 2016 Dec 1;2:16086. doi: 10.1038/nrdp.2016.86
- Human Papillomavirus (HPV) Vaccination: What Everyone Should Know. https://www.cdc.gov/vaccines/vpd/hpv/public
- Kaderli R, Schnüriger B, Brügger LE. The impact of smoking on HPV infection and the development of anogenital warts. Int J Colorectal Dis. 2014 Aug;29(8):899-908.
- Fleischer AB, Parrish CA, Glenn R, Feldman SR. Condylomata acuminata (genital warts): patient demographics and treating physicians. Sex Transm Dis. 2001 Nov;28(11):643-7.
- Chelimo C, Wouldes TA, Cameron LD, Elwood JM. Risk factors for and prevention of human papillomaviruses (HPV), genital warts and cervical cancer. J. Infect. 2013 Mar;66(3):207-17.
- Burchell AN, Tellier PP, Hanley J, Coutlée F, Franco EL. Human papillomavirus infections among couples in new sexual relationships. Epidemiology. 2010 Jan;21(1):31-7. doi: 10.1097/EDE.0b013e3181c1e70b
- Burchell AN, Coutlée F, Tellier PP, Hanley J, Franco EL. Genital transmission of human papillomavirus in recently formed heterosexual couples. J Infect Dis. 2011 Dec 1;204(11):1723-9. doi: 10.1093/infdis/jir644
- Lam JU, Rebolj M, Dugué PA, Bonde J, von Euler-Chelpin M, Lynge E. Condom use in prevention of Human Papillomavirus infections and cervical neoplasia: systematic review of longitudinal studies. J Med Screen. 2014 Mar;21(1):38-50. doi: 10.1177/0969141314522454
- Human Papillomavirus (HPV) Infection. https://www.cdc.gov/std/treatment-guidelines/hpv.htm
- Ryndock EJ, Meyers C. A risk for non-sexual transmission of human papillomavirus? Expert Rev Anti Infect Ther. 2014 Oct;12(10):1165-70. doi: 10.1586/14787210.2014.959497
- Sabeena, S., Bhat, P., Kamath, V., and Arunkumar, G. (2017) Possible non-sexual modes of transmission of human papilloma virus. J. Obstet. Gynaecol. Res., 43: 429– 435. doi: 10.1111/jog.13248
- Maturo SC, Hartnick CJ. Juvenile-onset recurrent respiratory papillomatosis. Adv Otorhinolaryngol. 2012;73:105-8. doi: 10.1159/000334457
- Derkay CS, Bluher AE. Update on Recurrent Respiratory Papillomatosis. Otolaryngol Clin North Am. 2019 Aug;52(4):669-679. doi: 10.1016/j.otc.2019.03.011
- Wangu Z, Hsu KK. Impact of HPV vaccination on anogenital warts and respiratory papillomatosis. Hum Vaccin Immunother. 2016 Jun 2;12(6):1357-62. doi: 10.1080/21645515.2016.1172754
- Suzanne M. Garland, Marc Steben, Heather L. Sings, Margaret James, Shuang Lu, Radha Railkar, Eliav Barr, Richard M. Haupt, Elmar A. Joura, Natural History of Genital Warts: Analysis of the Placebo Arm of 2 Randomized Phase III Trials of a Quadrivalent Human Papillomavirus (Types 6, 11, 16, and 18) Vaccine, The Journal of Infectious Diseases, Volume 199, Issue 6, 15 March 2009, Pages 805–814, https://doi.org/10.1086/597071
- Ball, S.L.R., Winder, D.M., Vaughan, K., Hanna, N., Levy, J., Sterling, J.C., Stanley, M.A. and Goon, P.K.C. (2011), Analyses of human papillomavirus genotypes and viral loads in anogenital warts. J. Med. Virol., 83: 1345-1350. https://doi.org/10.1002/jmv.22111
- Cancers Associated with Human Papillomavirus, United States—2013–2017. https://www.cdc.gov/cancer/uscs/about/data-briefs/no18-hpv-assoc-cancers-UnitedStates-2013-2017.htm
- Saraiya M, Unger ER, Thompson TD, Lynch CF, Hernandez BY, Lyu CW, Steinau M, Watson M, Wilkinson EJ, Hopenhayn C, Copeland G, Cozen W, Peters ES, Huang Y, Saber MS, Altekruse S, Goodman MT; HPV Typing of Cancers Workgroup. US assessment of HPV types in cancers: implications for current and 9-valent HPV vaccines. J Natl Cancer Inst. 2015 Apr 29;107(6):djv086. doi: 10.1093/jnci/djv086
- Varma S, Lathrop E, Haddad LB. Pediatric condyloma acuminata. J Pediatr Adolesc Gynecol. 2013 Dec;26(6):e121-2.
- Costa-Silva M, Fernandes I, Rodrigues AG, Lisboa C. Anogenital warts in pediatric population. An Bras Dermatol. 2017 Sep-Oct;92(5):675-681.
- Lacey CJ, Woodhall SC, Wikstrom A, Ross J. 2012 European guideline for the management of anogenital warts. J Eur Acad Dermatol Venereol. 2013 Mar;27(3):e263-70.
- Sporkert M, Rübben A. [Buschke-Lowenstein tumors]. Hautarzt. 2017 Mar;68(3):199-203.
- O’Mahony C, Gomberg M, Skerlev M, Alraddadi A, de Las Heras-Alonso ME, Majewski S, Nicolaidou E, Serdaroğlu S, Kutlubay Z, Tawara M, Stary A, Al Hammadi A, Cusini M. Position statement for the diagnosis and management of anogenital warts. J Eur Acad Dermatol Venereol. 2019 Jun;33(6):1006-1019.
- Leslie SW, Sajjad H, Kumar S. Genital Warts. [Updated 2019 Oct 8]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2020 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK441884
- Anogenital Warts. https://www.cdc.gov/std/treatment-guidelines/anogenital-warts.htm
- Human Papillomavirus Infection. https://www.std.uw.edu/go/comprehensive-study/hpv/core-concept/all
- D’Ambrogio A, Yerly S, Sahli R, Bouzourene H, Demartines N, Cotton M, Givel JC. Human papilloma virus type and recurrence rate after surgical clearance of anal condylomata acuminata. Sex Transm Dis. 2009 Sep;36(9):536-40. doi: 10.1097/OLQ.0b013e3181a866a3
- Filley CM, Graff-Richard NR, Lacy JR, Heitner MA, Earnest MP. Neurologic manifestations of podophyllin toxicity. Neurology. 1982 Mar;32(3):308-11. doi: 10.1212/wnl.32.3.308
- Scheinfeld N. Update on the treatment of genital warts. Dermatol. Online J. 2013 Jun 15;19(6):18559.