Heck’s disease
Heck’s disease also known as focal epithelial hyperplasia or multifocal epithelial hyperplasia, is a relatively benign and rare infection in the mouth caused by oral human papillomavirus (HPV) types 13 or 32 1. Moreover, the HPV-32 tends to cause Heck’s disease in the older age groups while HPV-13 seems to be equally involved in the development of Heck’s disease in both young and old patients 1. Heck’s disease is predominantly found in children and adolescents with indigenous heritage and has a variable female predilection 2. Heck’s disease often affects multiple members of given family. This familial tendency may be related to either genetic susceptibility or HPV transmission between family members 3.
Clinically, Heck’s disease is characterized by presence of multiple, soft, sessile, white or pink papules and nodules, measuring 1 – 10 mm in diameter, presenting a color similar to circumjacent mucosa and usually affecting lips, buccal mucosa and the tongue 4. Although the papules will spontaneously regress without treatment over time, some patients opt for excisional biopsy for functional or cosmetic purposes 5.
Heck’s disease was first identified by Dr Heck and his team in 1965 6 among a Navajo population in the United States and has since been reported among other indigenous groups throughout the world 7. Worldwide Heck’s disease is rare; however, it is frequently observed among specific ethnic and racial groups. Heck’s disease is found in a high frequency among Inuits and Indians resident in North, Central and South America, Eskimos from Greenland and North Canada, and descendants of Khoi-San in South Africa 8. An incidence as high as 30% has been reported among Inuit and children from some communities in Venezuala. It is rare in Caucasian populations. As the disorder is rare in other communities, there are few population studies, most of which being of geographical regions and/or ethnic groups in which Heck’s disease is common.
The diagnosis ofHeck’s disease is mainly based on the clinical features of the lesions and a biopsy pathology report. Since the discovery of HPV particles in these lesions in 1970, there has been an increase in the diagnostic use of molecular biology techniques, including in situ DNA hybridization and polymerase chain reaction (PCR), which have proven highly useful in identifying the types of HPV involved 9.
Treatment options for Heck’s disease comprise surgical removal, laser excision, electrocautery, cryotherapy and topical agents such as imiquimod, retinoic acid or trichloroacetic acid 10. However, treatment is not always required, as most lesions are asymptomatic and frequently undergo spontaneous regression 5; there is no tendency to malignant transformation 11.
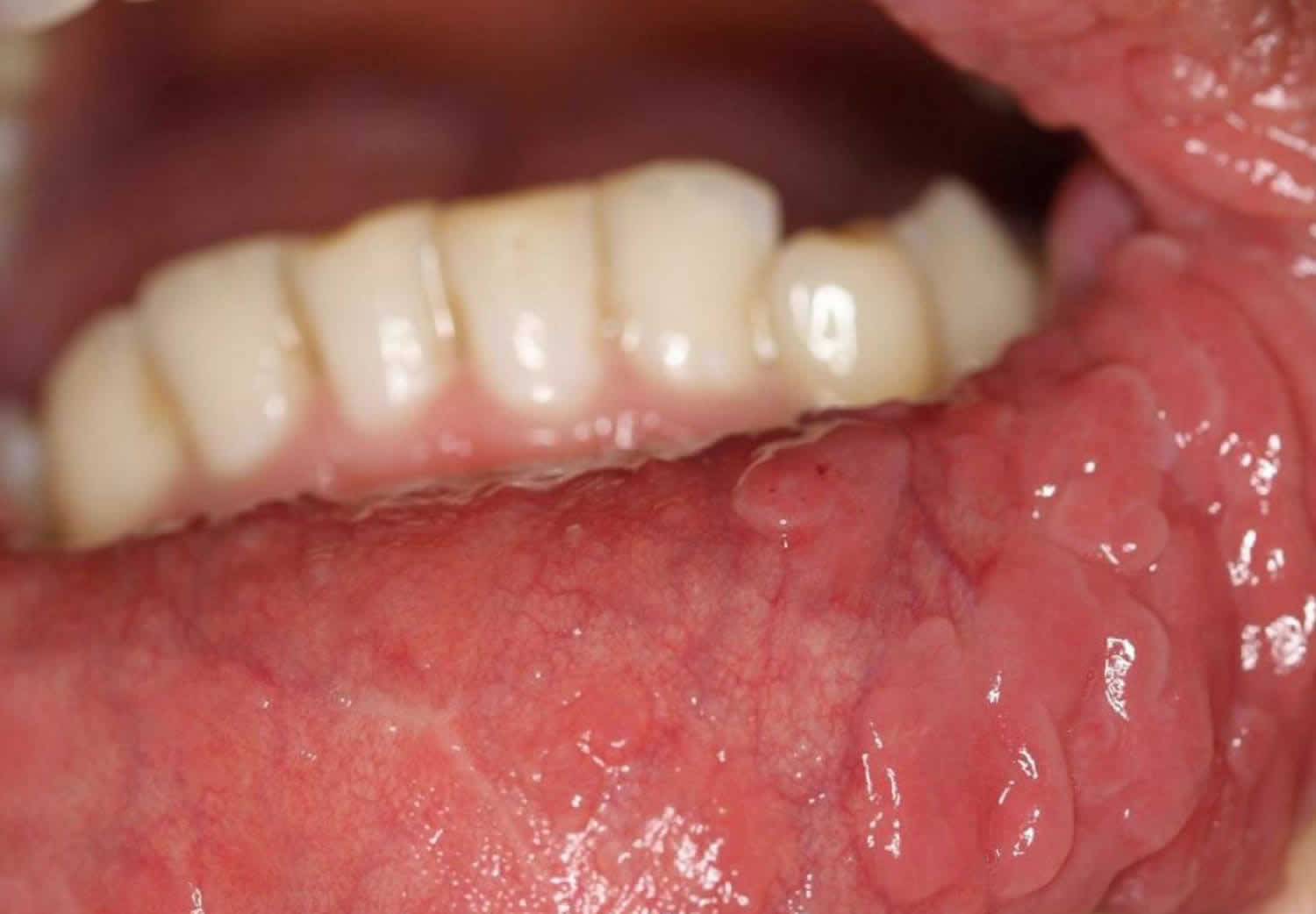
Figure 1. Heck’s disease
Footnote: Multifocal epithelial neoplasia on the lower labial mucosa of a 7-year-old boy with Heck disease, manifesting as numerous flat, white papules and coalescing plaques.
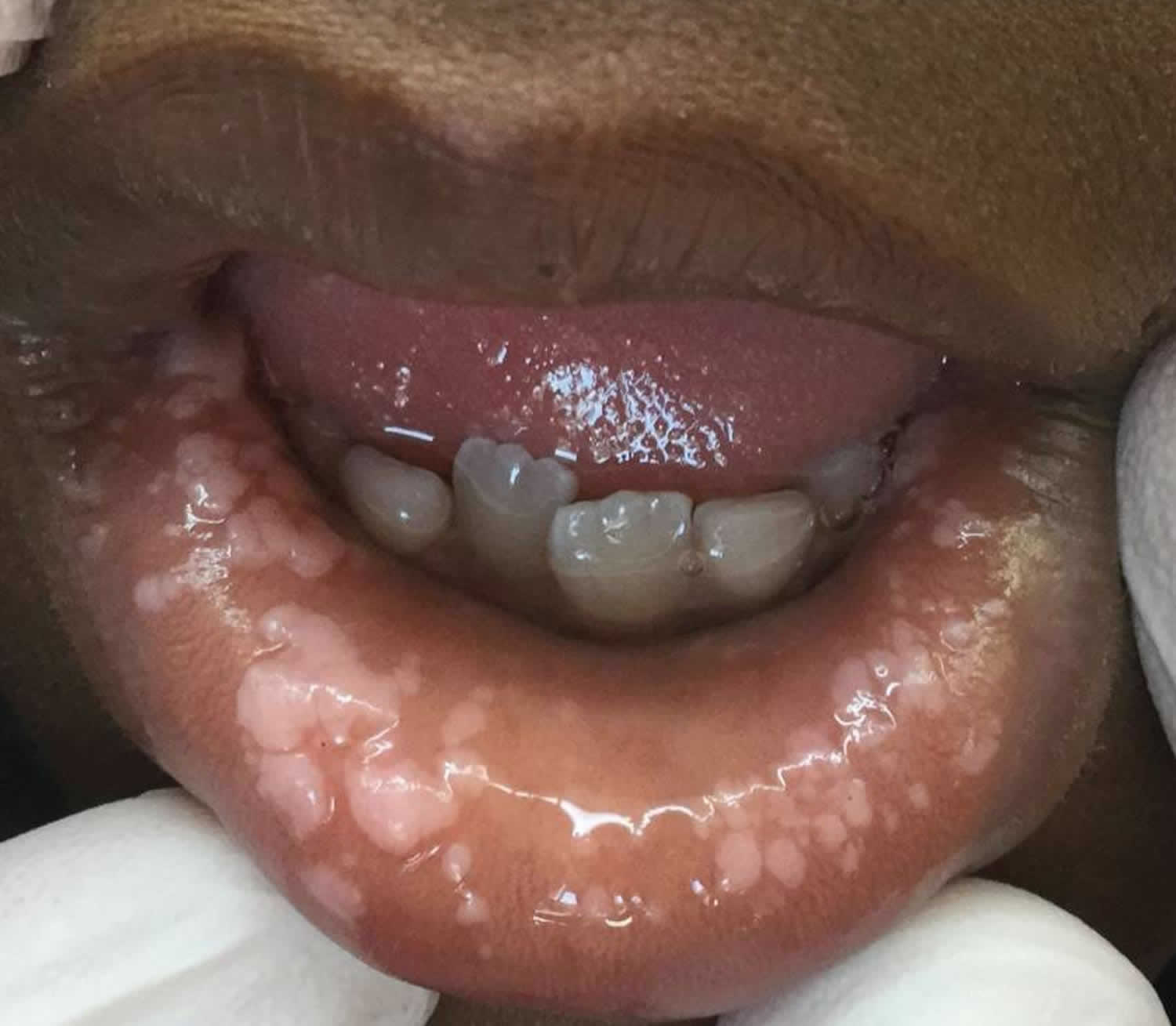
[Source 10 ]Figure 2. Heck’s disease
Footnote: Clinical photograph to show the multiple lesions of focal epithelial hyperplasia on the left buccal mucosa of the 7-year-old girl patient.
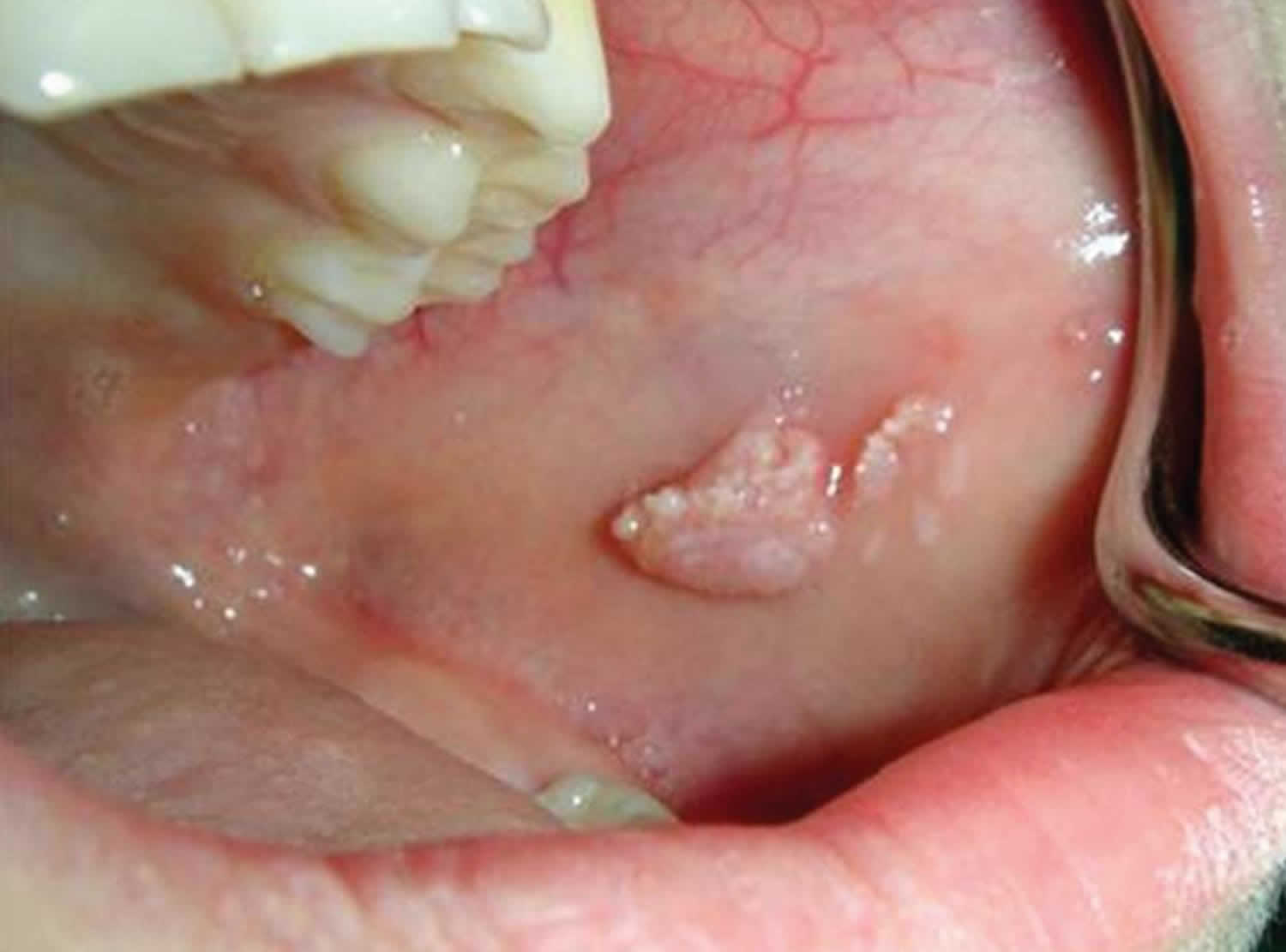
[Source 1 ]Figure 3. Heck’s disease
Footnote: Multiple, soft, sessile, smooth surface papules and nodules involving upper, and lower labial mucosa, That are so close together that form a cobble stone appearance.
[Source 12 ]Heck’s disease causes
Heck’s disease is a rare viral infection of the oral mucosa caused by human papilloma virus (HPV) subtypes 13 or 32 13. In spite of the fact that the most common associated HPV subtypes are 13 and 32, cross-reaction with other subtypes has also been reported 8. Moreover, the HPV subtype 32 (HPV-32) tends to cause the disease in the older age groups while the HPV subtype 13 (HPV-13) seems to be equally involved in the development of disease in both young and old patients 1.
Risk factors for Heck’s disease include poor hygiene, poverty and communal lifestyles. Extensive lesions have been reported in association with HIV infection or AIDS. In a systematic review of 9 studies that collected oral HPV data from 3762 cancer-free, HIV-negative individuals from the United States, Brazil, Mexico, and Finland, Wood et al 14 reported that 7.5 % had an oral infection with any HPV type at baseline. In a study involving 307 Australian university students (aged 18-35 years), 7 students (6 men and 1 woman; 2.3%) tested positive for oral HPV infection. Those positive for oral HPV were more likely to have received oral sex from more partners in their lifetime 15.
Factors that determine the disease susceptibility are unclear, but genetics and having human lymphocytic antigen (HLA-DR4, DRB10404) allele in particular, are thought to play a major role in Heck’s disease 13.
Genetic predisposition is also frequently mentioned as an etiological factor for Heck’s disease 16. The familial presence of Heck’s disease was first highlighted by Gómez et al in Columbia in 1969 17. In 1993, Premoli-De-Percoco 18 found that members of six generations of a Venezuelan family had been diagnosed with Heck’s disease and concluded that the disease has a genetic basis, noting that all generations were born in a small village with a very strong American Indian genetic component. In 2004, García-Corona et al 19 assessed the effect of genetics by studying the association of HLA-DR4 alleles (DRB10404) with HPV infection, yielding results in support of the genetic basis of Heck’s disease.
Nutritional deficiencies and environmental factors have been proposed by certain authors 20, including poverty and the lack of hygiene 21. The role of immunosuppression is also under investigation. Feller et al 22 reported the case of a seropositive child undergoing highly active antiretroviral treatment who was also diagnosed with Heck’s disease and successfully treated by diode laser; the authors suggested that the anti-HIV therapy may have been responsible for the disease. In another report on an HIV-positive patient with Heck’s disease, Marvan et al 23 concluded that the correlation between the immune state and HPV infection warranted further investigation.
Heck’s disease symptoms
In general, Heck’s disease is asymptomatic. It may present because of the appearance, a feeling of roughness or accidental biting of a lesion.
Heck’s disease or focal epithelial hyperplasia only affects the lining of the mouth, most commonly:
- inside of the lips
- inside of the cheeks
- sides of the tongue
However, the gums and tonsil area can also be involved.
Heck’s disease most commonly presents as a soft smooth flat-topped slightly raised cobblestoned appearance. It is usually the same color as the rest of the mouth, but sometimes paler. There are often multiple lesions of less than 1cm in diameter.
Heck’s disease diagnosis
Heck’s disease may be suspected clinically, particularly in high risk populations.
A biopsy from a lesion in the mouth will show diagnostic pathology – the epithelium is very thickened and raised above the surrounding mucosa with typical ‘mitosoid’ cells.
The diagnosis can be confirmed by the detection of HPV 13 or 32 DNA following polymerase chain reaction (PCR) on a scraping taken from a lesion, or by in situ hybridization of biopsy specimens. In some cases, particularly in children, a mucosal scraping to confirm the specific HPV types, rather than a more invasive biopsy, is all that is required to confirm the diagnosis. The identification of the specific HPV type may be important in cases of suspected child sexual abuse as HPV 13 and 32 have only ever been detected in the oral mucosa.
Heck’s disease treatment
Heck’s disease is a benign condition with no cases of oral cancer have been reported. It therefore usually does not require treatment unless lesions are of cosmetic concern or if accidental or deliberate biting of lesions occurs. Treatment options then may include excisional biopsy, CO2 laser or diathermy. Successful use of topical imiquimod 5% cream has been reported. Followup is recommended in all cases.
Heck’s disease prognosis
The prognosis of Heck’s disease is good, given that most lesions remit spontaneously, but periodical clinical follow-ups are considered crucial 24. Lesions that do not remit or cause functional and/or cosmetic problems may be removed by various means, including surgery, cryotherapy, electrocoagulation, laser, chemical agents (e.g., retinoic acid) or immunostimulants (e.g., interferon).
The natural history of Heck’s disease is currently unclear, although it does seem to persist for many years. Some surveys claim there is no difference in the incidence between children or adults although the clinical appearance may be different, with adults having fewer and flatter lesions than children. Others have reported involvement only of children, and therefore a presumed resolution of the condition by adult life.
References- Ozden B, Gunduz K, Gunhan O, Ozden FO. A Case Report of Focal Epithelial Hyperplasia (Heck’s disease) with PCR Detection of Human Papillomavirus. J Maxillofac Oral Surg. 2011;10(4):357-360. doi:10.1007/s12663-011-0184-2 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3267916
- Falaki F, Amir Chaghmaghi M, Pakfetrat A, Delavarian Z, Mozaffari PM, Pazooki N. Detection of human papilloma virus DNA in seven cases of focal epithelial hyperplasia in Iran. J Oral Pathol Med. 2009;38(10):773–776. doi: 10.1111/j.1600-0714.2009.00784.x
- Nevile B, Damm D, Allen C, Bouquot J. Oral and Maxillofacial Pathology. Philadelphia: Saunders Elsevier; 2009.
- Said AK, Leao JC, Fedele S, Porter SR. Focal epithelial hyperplasia – an update. J Oral Pathol Med. 2013;42(6):435–442. doi: 10.1111/jop.12009
- Eversole LR. Clinical Outline of Oral Pathology: Diagnosis and Treatment. People’s Medical Publishing House; 2011.
- Archard HO, Heck JW, Stanley HR. Focal epithelial hyperplasia: an unusual oral mucosal lesion found in Indian children. Oral Surg Oral Med Oral Pathol. 1965;20:201-212. doi:10.1016/0030-4220(65)90192-1
- Wu JSA, Florian MC, Rodrigues DA, Tomimori J. Skin diseases in indigenous population: retrospective epidemiological study at Xingu Indigenous Park (XIP) and review of the literature. Int J Dermatol. 2017;56(12):1414-1420. doi:10.1111/ijd.13716
- Said AK, Leao JC, Fedele S, Porter SR. Focal epithelial hyperplasia – an update. J Oral Pathol Med. 2013 Jul;42(6):435-42. doi: 10.1111/jop.12009
- Bascones-Martínez A, Cok S, Bascones-Ilundáin C, Arias-Herrera S, Gomez-Font R, Bascones-Ilundáin J. Multifocal epithelial hyperplasia: A potentially precancerous disease? (Review). Oncol Lett. 2012;3(2):255-258. doi:10.3892/ol.2011.465 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3362355
- Kreuter A, Silling S. Multifocal epithelial hyperplasia (Heck disease) in a 7-year-old boy. CMAJ. 2018;190(50):E1481. doi:10.1503/cmaj.180882 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6291387
- de Castro LA, de Castro JG, da Cruz AD, Barbosa BH, de Spindula-Filho JV, Costa MB. Focal Epithelial Hyperplasia (Heck’s Disease) in a 57-Year-Old Brazilian Patient: A Case Report and Literature Review. J Clin Med Res. 2016;8(4):346-350. doi:10.14740/jocmr2466w https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4780501
- Mansouri Z, Bakhtiari S, Noormohamadi R. Extensive Focal Epithelial Hyperplasia: A Case Report. Iran J Pathol. 2015;10(4):300-305. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4539750
- Bascones-Martínez A, Cok S, Bascones-Ilundáin C, Arias-Herrera S, Gomez-Font R, Bascones-Ilundáin J. Multifocal epithelial hyperplasia: A potentially precancerous disease? (Review). Oncol Lett. 2012 Feb;3(2):255-258. doi: 10.3892/ol.2011.465
- Wood ZC, Bain CJ, Smith DD, Whiteman DC, Antonsson A. Oral human papillomavirus infection incidence and clearance: a systematic review of the literature. J Gen Virol. 2017;98(4):519-526. doi:10.1099/jgv.0.000727
- Antonsson A, Cornford M, Perry S, Davis M, Dunne MP, Whiteman DC. Prevalence and risk factors for oral HPV infection in young Australians. PLoS One. 2014;9(3):e91761. doi:10.1371/journal.pone.0091761
- WITKOP CJ Jr, NISWANDER JD. FOCAL EPITHELIAL HYPERPLASIA IN CENTRAL AND SOUTH AMERICAN INDIANS AND LADINOS. Oral Surg Oral Med Oral Pathol. 1965 Aug;20:213-7. doi: 10.1016/0030-4220(65)90193-3
- Gomez A, Calle C, Arcila G, Pindborg JJ. Focal epithelial hyperplasia in a half-breed family of Colombians. J Am Dent Assoc. 1969 Sep;79(3):663-7. doi: 10.14219/jada.archive.1969.0328
- Premoli-De-Percoco G, Cisternas JP, Ramírez JL, Galindo I. Focal epithelial hyperplasia: human-papillomavirus-induced disease with a genetic predisposition in a Venezuelan family. Hum Genet. 1993 May;91(4):386-8. doi: 10.1007/BF00217363
- García-Corona C, Vega-Memije E, Mosqueda-Taylor A, Yamamoto-Furusho JK, Rodríguez-Carreón AA, Ruiz-Morales JA, Salgado N, Granados J. Association of HLA-DR4 (DRB1*0404) with human papillomavirus infection in patients with focal epithelial hyperplasia. Arch Dermatol. 2004 Oct;140(10):1227-31. doi: 10.1001/archderm.140.10.1227
- Premoli-de-Percoco G, Galindo I, Ramirez JL. In situ hybridization with digoxigenin-labelled DNA probes for the detection of human papillomavirus-induced focal epithelial hyperplasia among Venezuelans. Virchows Arch A Pathol Anat Histopathol. 1992;420(4):295-300. doi: 10.1007/BF01600207
- Ledesma-Montes C, Garcés-Ortíz M, Hernández-Guerrero JC. Clinicopathological and immunocytochemical study of multifocal epithelial hyperplasia. J Oral Maxillofac Surg. 2007 Nov;65(11):2211-7. doi: 10.1016/j.joms.2006.11.035
- Feller L, Khammissa RA, Wood NH, Malema V, Meyerov R, Lemmer J. Focal epithelial hyperplasia (Heck disease) related to highly active antiretroviral therapy in an HIV-seropositive child. A report of a case, and a review of the literature. SADJ. 2010 May;65(4):172-5.
- Marvan E, Firth N. Focal epithelial hyperplasia in an HIV positive man. An illustrated case and review of the literature. Aust Dent J. 1998 Oct;43(5):305-10. doi: 10.1111/j.1834-7819.1998.tb00178.x
- Hashemipour MA, Shoryabi A, Adhami S, Mehrabizadeh Honarmand H. Extensive focal epithelial hyperplasia. Arch Iran Med. 2010 Jan;13(1):48-52.