Abscopal effect
Abscopal effect is an extremely rare phenomenon occurring when radiation therapy (radiotherapy) or treatment of a primary tumor burden not only results in debulking of the targeted site but also reduces tumor size at distant sites in parts of the body that were not the direct target of local therapy 1. The exact underlying mechanism of the abscopal effect remains unclear. It is thought that in the abscopal effect, the immune system is stimulated to fight cancer in the whole body as a result of the local therapy, an immune response mediated by activated CD8+ T cells has been shown to play a central role 2.
Abscopal effect, initially defined by a researcher Dr. R. H. Mole in 1953 3 who showed that radiation could shrink a tumor on one side of a mouse and lead to the regression of an untreated tumor on the other side of the animal. Abscopal means “at a distance from the irradiated volume but within the same organism,” and is derived from ab-, prefix with the meaning “position away from” and scopus (Latin) meaning “mark or target for shooting at”. The abscopal effect occurs infrequently in clinical cases, though it has been reported in different tumor types, renal cell carcinoma, melanoma, lymphomas, hepatocellular carcinoma, and other tumor types 4. Of note, when it does occur, the abscopal effect is associated with prolongation of overall patient survival time 5. For instance, Stamell et al. 6 reported a metastatic melanoma patient who received palliative radiation therapy to the primary tumor also experienced regression of non-irradiated metastases 6. Learning more about the abscopal effect may help in the development of new cancer treatments, especially for metastatic cancer.
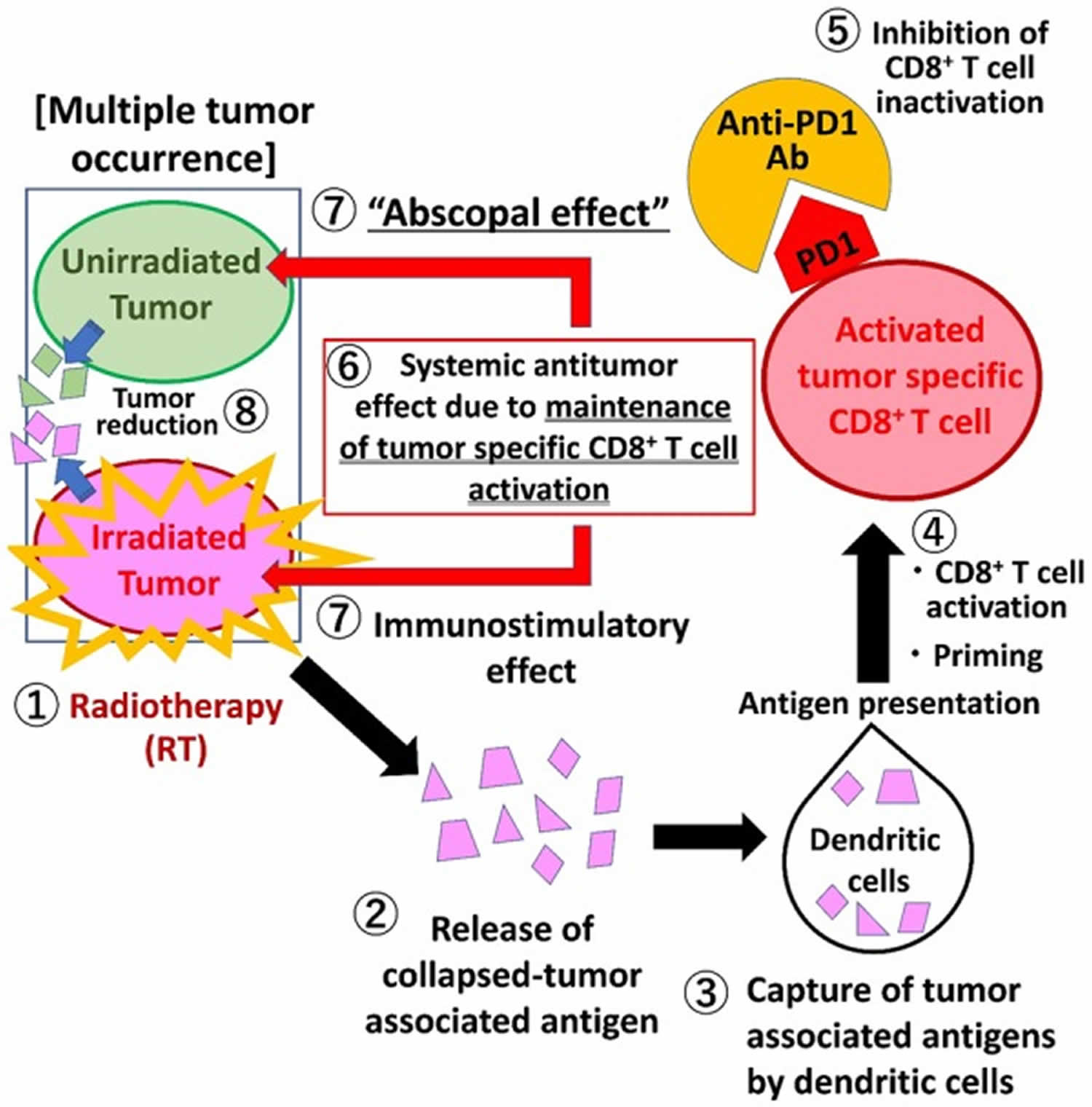
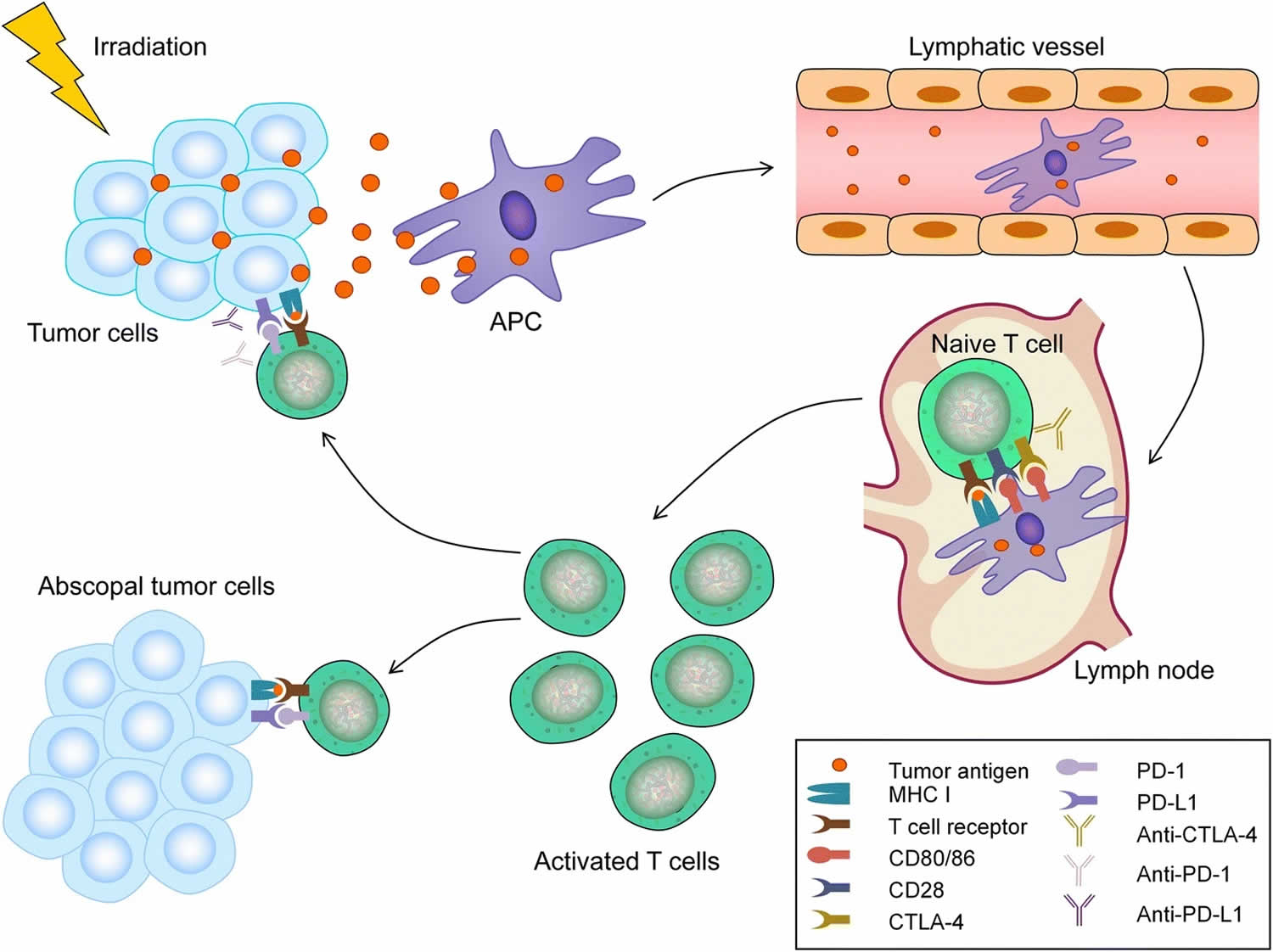
Figure 1. Mechanism of abscopal effect radiotherapy
Footnote: Radiotherapy (RT) can lead to immunogenic cell death and the release of tumor antigens by irradiated tumor cells. These neoantigens are taken up by antigen-presenting cells (APCs), such as dendritic cells (DCs) and phagocytic cells. The APCs interact with tumor antigens and then migrate to the lymph nodes where they present antigens to T cells, a process that is mediated by the MHC pathway and other co-stimulatory signals, such as CD80 and CD28. After activation by multiple signals, T cells, especially the CD8+ T cells, are activated and begin to propagate. As a result, activated effector T cells exit the lymph nodes and home to tumors, including primary tumors and non-irradiated tumor metastases, to exert their effect of killing tumor cells. However, cytotoxic T lymphocyte-associated antigen 4 (CTLA-4) competitively combines with CD80/86 and inhibits the activation of T cells. Following T cell activation, programmed cell death 1 (PD-1) receptors that are expressed on the T cell surface bind primarily to programmed death-ligand 1 (PD-L1) and inhibit immune responses. The administration of immune checkpoint blockades of CTLA-1, PD-1, and PD-L1 can enhance the anti-tumor immunity of radiotherapy.
[Source 7 ]Abscopal effect of radiotherapy and using immunotherapy to boost the abscopal effect
Radiotherapy is one of the cornerstones of cancer treatment in definitive and the palliative settings with well established local effects. Classical radiobiology postulates that the cytotoxic effects of radiation are the result of direct DNA damage and indirect generation of cell-damaging free radicals 8. This makes this modality precisely a local therapy which is characterized by an impressively high degree of spatial accuracy. Additional research has demonstrated that radiotherapy also causes localized bystander effects, where the treated tumor cells influence neighboring cells 9. Because of all these cytotoxic effects and the effects on circulating lymphocytes radiotherapy is known to be immunosuppressant, but by the definition of the abscopal effect, there might be an immune stimulant nature of radiotherapy 10.
Radiotherapy does not only exerts direct cytotoxic effects on tumor cells but also reprograms the tumor microenvironment to exert a potent antitumor immune response and enhances antitumor immunity 11. It initiates immunogenic cell death causing production and release of the cytokines and chemokines into the tumor microenvironment. This causes chemoattraction and infiltration of dendritic cells (DCs) to the site of the tumor. Activation of Dcs, which are essential antigen presenting cells, and up-regulation of cytotoxic T lymphocytes are responsible for the abscopal effect mechanism 11. On the other hand, the infiltration of Tregs and myeloid-derived suppressor cells (MDSCs) act as a brake mechanism on the immune system.
As a part of the radiotherapy’s nature, which is immunosuppressive dominantly, abscopal is seen as a rare event. Over the years sporadic abscopal effect events were published as case reports. Over the years sporadic abscopal effect events were published as case reports. Abuodeh et al. 12 reviewed the literature and reported that 46 case reports were published between 1969 and 2014 where the abscopal effect was observed from radiotherapy alone. In the reviewed cases, the abscopal effect was observed independently from the treatment dose, fractionation, modality and the characteristic of the target lesion. The dose range was 0,45 Gy to 74,8 Gy, and the abscopal effect was seen at the second week earliest. Moreover, most of the reported cases were on renal cell carcinoma, melanoma, and lymphoma, which are the disease entities traditionally considered immunogenic 12.
Although the abscopal effect of radiotherapy alone has been reported by a growing number of trials and cases, the overall occurrence rate was relatively low. This may be explained by the insufficiency of radiotherapy alone to overcome the immunoresistance of malignant tumors. Given that immunotherapy can reduce host’s immune tolerance toward tumors, it is possible that the combination of radiotherapy and immunotherapy can amplify the anti-tumor immune response, which is more likely to cause the occurrence of an abscopal effect 13. In fact, this synergistic anti-tumor effect has been investigated in many clinical studies. Nevertheless, the mechanism of the abscopal effect is not yet completely understood.
Many methods to increase the prevalence of the abscopal effect have been tried up to date. These strategies were based on the combination of radiotherapy with cytokines, stimulation of dendritic cells (DCs) to aggravate tumor antigen-presenting, vaccination with autologous tumor cells, targeting Toll-like receptors and immunotherapy-radiotherapy combination 14. Dewan et al. 15 tried to find the optimal dose and fractionation schemes of radiotherapy to induce the abscopal effect with immunotherapy. They showed that fraction dose of radiotherapy might be a critical determinant of its efficacy to synergize with immunotherapy. In their experimental design, 8 Gy given in three consecutive days (8Gyx3) was the most effective regimen for induction of the abscopal effect in combination with cytotoxic T-lymphocyte–associated antigen 4 (CTLA-4) blockade 15. Later they studied the underlying mechanisms for this dose effect. They showed that the balance between the cytosolic DNA and DNA exonuclease Trex1 activation plays a vital role in the synergistic effect of radiotherapy and immunotherapy. radiotherapy induces double-strand DNA accumulation in the cytosol, and the amount of double-strand DNA is directly proportional to the fraction dose of radiotherapy. Cytosolic DNA has an essential impact on the activation of antitumor immunity, as it activates the DNA sensor cyclic GMP-AMP (cGAMP) synthase (cGAS) and its downstream effector, stimulator of interferon genes (STING). Activation of this cascade results in interferon-b secretion by cancer cells, which causes dendritic cell recruitment and activation. This stimulus is essential for priming of CD8 T cells and antitumor immunity. On the other hand, doses above 12–18 Gy per fraction cause activation of DNA exonuclease Trex1. Activated DNA exonucleases degrade cytosolic DNA and attenuate the immunologic response stimulated by radiotherapy. This delicate balance between cytosolic DNA and activated Trex1 is optimal at 8Gyx3 for the emergence of the !abscopal effect with immunotherapy 16.
With the new modern immunotherapy era, the potential for immune activation by radiation therapy defines an important role for radiation therapy in systemic disease. An increasing number of reports of abscopal effect is seen in cases where radiotherapy is combined with various immunotherapeutic agents that enhance the immune response or remove the inhibitory effect of existing immune checkpoints.
Recently, immune checkpoint inhibitors (e.g., anti-PD1 antibodies) have emerged as powerful therapeutics for the treatment of various metastatic cancer types 17. Anti-PD1 antibodies are especially effective against tumors with high mutational burden 18, which presumably possess large numbers of neoantigens, leading to activation of an anti-tumor immune response 19.
In patients who have undergone radiotherapy, radiation-induced cell deaths increase tumor-associated antigens and consequently promote activation of an immune response 20. Meanwhile, the activated tumor-specific CD8+ T cells express programmed death 1 (PD1) and release interferon-γ (IFN-γ) 21. Moreover, tumor cells express the ligand of PD1 (PD-L1) in cytoplasm and/or plasma membrane, facilitating escape from attack by activated tumor-specific CD8+ T cells 22. Thus, although tumor-specific CD8+ T cells were prepared to defend against attacking tumor cells, adaptive immune tolerance is induced in a significant proportion of patients after irradiation, as a function of the PD1/PD-L1 pathway, which inhibits the radiotherapy-mediated systemic anti-tumor effects 21.
Postow et al. 23 were the first to report an abscopal effect in a metastatic melanoma case treated with ipilimumab (a monoclonal antibody that inhibits an immune checkpoint on T cells, the cytotoxic T lymphocyte-associated antigen 4) and radiotherapy. The patient had progressive disease during maintenance treatment with ipilimumab. radiotherapy was delivered to a painful paraspinal mass with a palliative intent, which led to a systemic response 23. Reynders et al. 24 published an overview of the abscopal effect of radiotherapy in 2015. They inclueded studies that used immmune modifying therapy but excluded studies using total body irradiation and/or cytotoxic agents. They reviewed 26 clinical reports, 23 case reports, and one retrospective study. The median time to abscopal response was five months and was maintained for three to 39 months 24. Even further, Poleszczuk and Enderling suggested a mathematical model that can be used to predict and dissect the complexity of the immune-mediated response at multiple tumor sites after applying focal irradiation and systemic immunotherapy 25.
In a recently published article by Yazici et al. 26, the abscopal effect is demonstrated in a sinonasal carcinoma patient. Irradiation the target lesion with concurrently pembrolizumab resulted in resolution of all the metastatic lesions. In this patient, pembrolizumab was used as second-line therapy, and tumor progression was observed under pembrolizumab. When radiotherapy came into the picture and combined with immunotherapy, complete treatment response was achieved 26.
Figure 2. Abscopal effect with radiotherapy (RT) and anti-PD1 antibody (immune checkpoint inhibitors) combination therapy
Footnote: Activated tumor-specific CD8+ T cells play a central role in inducing the abscopal effect. RT causes tumor collapse, which releases tumor-specific antigen. Dendritic cells then capture the tumor-specific antigen and activate tumor-specific CD8+ T cells. Activated tumor-specific CD8+ T cells show anti-tumor effects in irradiated tumors (i.e., immunostimulatory effect) as well as non-irradiated tumors (i.e., abscopal effect). PD1 on activated CD8+ T cells inactivates T cells when it binds to PD-L1 on tumor cells. Just as anti-PD1 antibody inhibits the binding of PD1 to PD-L1, anti-PD1 antibody contributes to maintaining tumor-specific CD8+ T cell activation. Thus, RT and anti-PD1 antibody combination therapy co-operate to maintain activated tumor-specific CD8+ cells and enhance systemic anti-tumor effects.
[Source 27 ] References- A Peculiar Case of the Abscopal Effect: Radioactive Iodine Therapy Incidentally Palliating Marginal Zone Lymphoma. Case Reports in Oncological MedicineVolume 2015, Article ID 281729, 4 pages https://doi.org/10.1155/2015/281729
- Park SS, Dong H, Liu X, Harrington SM, Krco CJ, Grams MP, Mansfield AS, Furutani KM, Olivier KR, Kwon ED. PD-1 restrains radiotherapy-induced abscopal effect. Cancer Immunol Res. 2015;3:610–9.
- Mole RH. Whole body irradiation—radiobiology or medicine? Br J Radiol. 1953;26:234–41.
- Poleszczuk JT, Luddy KA, Prokopiou S, Robertson-Tessi M, Moros EG, Fishman M, et al. Abscopal benefits of localized radiotherapy depend on activated T-cell trafficking and distribution between metastatic lesions. Cancer Res. 2016;76:1009–18.
- Grimaldi AM, Simeone E, Giannarelli D, Muto P, Falivene S, Borzillo V, Giugliano FM, Sandomenico F, Petrillo A, Curvietto M, Esposito A, Paone M, Palla M, Palmieri G, Caraco C, Ciliberto G, Mozzillo N, Ascierto PA. Abscopal effects of radiotherapy on advanced melanoma patients who progressed after ipilimumab immunotherapy. Oncoimmunology. 2014;3:e28780
- Stamell EF, Wolchok JD, Gnjatic S, Lee NY, Brownell I. The abscopal effect associated with a systemic anti-melanoma immune response. Int J Radiat Oncol Biol Phys. 2013;85:293–5.
- Liu, Y., Dong, Y., Kong, L. et al. Abscopal effect of radiotherapy combined with immune checkpoint inhibitors. J Hematol Oncol 11, 104 (2018). https://doi.org/10.1186/s13045-018-0647-8
- Opportunities and challenges of radiotherapy for treating cancer. Schaue D, McBride WH. Nat Rev Clin Oncol. 2015;12:527–540.
- Contribution of the immune system to bystander and non-targeted effects of ionizing radiation. Rodel F, Frey B, Multhoff G, Gaipl U. Cancer Lett. 2015;356:105–113.
- Whole body irradiation; radiobiology or medicine? Mole RH. Br J Radiol. 1953;26:234–241.
- Radiotherapy and immunotherapy: a beneficial liaison? Weichselbaum RR, Liang H, Deng L, Fu YX. Nat Rev Clin Oncol. 2017;14:365–379.
- Systematic review of case reports on the abscopal effect. Abuodeh Y, Venkat P, Kim S. Curr Probl Cancer. 2016;40:25–37.
- Vatner RE, Cooper BT, Vanpouille-Box C, Demaria S, Formenti SC. Combinations of immunotherapy and radiation in cancer therapy. Front Oncol. 2014;4:325.
- Combining radiotherapy and immunotherapy: a revived partnership. Demaria S, Bhardwaj N, McBride WH, Formenti SC. Int J Radiat Oncol Biol Phys. 2005;63:655–666.
- Fractionated but not single-dose radiotherapy induces an immune-mediated abscopal effect when combined with anti-CTLA-4 antibody. Dewan MZ, Galloway AE, Kawashima N, Dewyngaert JK, Babb JS, Formenti SC, Demaria S. Clin Cancer Res. 2009;15:5379–5388.
- DNA exonuclease Trex1 regulates radiotherapy-induced tumour immunogenicity. Vanpouille-Box C, Alard A, Aryankalayil MJ, et al. Nat Commun. 2017;8:15618.
- Alsaab HO, Sau S, Alzhrani R, Tatiparti K, Bhise K, Kashaw SK, Iyer AK. PD-1 and PD-L1 checkpoint signaling inhibition for cancer immunotherapy: mechanism, combinations, and clinical outcome. Front Pharmacol. 2017;8:561.
- Le DT, Durham JN, Smith KN, et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science. 2017;357(6349):409-413. doi:10.1126/science.aan6733 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5576142
- Yi M, Qin S, Zhao W, Yu S, Chu Q, Wu K. The role of neoantigen in immune checkpoint blockade therapy. Exp Hematol Oncol. 2018;7:28.
- Hodge JW, Sharp HJ, Gameiro SR. Abscopal regression of antigen disparate tumors by antigen cascade after systemic tumor vaccination in combination with local tumor radiation. Cancer Biother Radiopharm. 2012;27:12–22.
- Taube JM, Anders RA, Young GD, Xu H, Sharma R, McMiller TL, Chen S, Klein AP, Pardoll DM, Topalian SL, Chen L. Colocalization of inflammatory response with B7-h1 expression in human melanocytic lesions supports an adaptive resistance mechanism of immune escape. Sci Transl Med. 2012;4:127ra37
- Qu QX, Xie F, Huang Q, Zhang XG. Membranous and cytoplasmic expression of PD-L1 in ovarian cancer cells. Cell Physiol Biochem. 2017;43:1893–906.
- Immunologic correlates of the abscopal effect in a patient with melanoma. Postow MA, Callahan MK, Barker CA, et al. N Engl J Med. 2012;366:925–931.
- The abscopal effect of local radiotherapy: using immunotherapy to make a rare event clinically relevant. Reynders K, Illidge T, Siva S, Chang JY, De Ruysscher D. Cancer Treat Rev. 2015;41:503–510.
- The optimal radiation dose to induce robust systemic antI-tumor immunity. Poleszczuk J, Enderling H. Int J Mol Sci. 2018;19:3377
- The synergistic effect of immune checkpoint blockade and radiotherapy in recurrent/metastatic sinonasal cancer. Yazici G, Gullu I, Cengiz M, et al. Cureus. 2018;10:0
- Baba K, Nomura M, Ohashi S, et al. Experimental model for the irradiation-mediated abscopal effect and factors influencing this effect. Am J Cancer Res. 2020;10(2):440-453. Published 2020 Feb 1. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7061743