Ectopic Pancreas
Ectopic pancreas also called heterotopic pancreas or aberrant pancreas is defined as the presence of healthy pancreatic tissue at a location that is not typical for the pancreas with the pancreatic tissue being separated from the normal pancreas in terms of its blood supply, nerves, and anatomical connection 1, 2, 3, 4, 5, 6, 7. Ectopic pancreas has a genetic make-up, physiologic function, and local environmental exposure similar to that of the pancreas 8.
Ectopic pancreas is a congenital anatomical abnormality that can arise in several locations within the digestive tract and other areas 9. Modern molecular studies have demonstrated that defective Notch signaling pathways and mutations in Neurog3 (neurogenin 3) are responsible for many cases. Several theories such as fusion abnormalities, migration abnormalities, and replication abnormalities have been proposed to explain the pathogenesis and occurrence of ectopic pancreas. The most probable theory implicates that during the development of normal pancreas from several evaginations, originating from the wall of the primitive duodenum, one or more evaginations may remain in the bowel wall 10. Migration of this embryonic remnant along with the development of the gastrointestinal tract gives rise to the ectopic pancreatic tissue 11. Another theory suggests that during embryogenesis pancreatic metaplasia of the endodermal tissues localized in the gastric submucosa may occur 11, 12. Currently, the embryological origin of ectopic pancreas remains uncertain. More recent theory is the dislocation theory, which proposes that pancreatic tissue deposits become detached from the primary pancreas and are then relocated to other developing organs, particularly the gastrointestinal system 1.
Ectopic pancreas is frequently located in the stomach (up to 38%) of which 95% are located in the greater curvature in the antrum 13, 3, followed by the duodenum (25%–35%) 14, jejunum (16%) 15, Meckel diverticulum, the ileum 16, liver, spleen, bile duct, gall bladder 17, omentum, fallopian tube, mediastinum 18 and more 19, 20. Generally, the most common ectopic pancreas site is the stomach (70%), located at the submucosa of the gastric antrum, the prepyloric region on the greater curvature or posterior wall and proximal portion of the duodenum or the jejunum 20, 21, 16. Rarely it can also be found in the ileum, colon, appendix, mesentery, gallbladder or Meckel diverticulum. The ectopic tissue usually measures 0.5-2.0 cm in its larger diameter (rarely up to 5 cm) 22.
There are 4 subtypes of ectopic pancreas based on histological examination 23, 24, 25, 26:
- Type 1 ectopic pancreas consists of typical pancreatic tissue with acini, ducts, and islet cells similar to those seen in normal pancreas
- Type 2 ectopic pancreas is composed of pancreatic ducts only, referred as canalicular variety
- Type 3 ectopic pancreas is characterized by glandular acinar tissue only (exocrine pancreas)
- Type 4 ectopic pancreas is made up of endocrine islet cells only (endocrine pancreas)
According to literature, ectopic pancreas is mainly located in submucosa (54–75% of cases, also, it may span the submucosa and muscularis propria in 23% of cases), following by muscular layer (muscularis propria in 8% of cases) and serous layer (11–13% of cases). Lesions which are located in the stomach and duodenal bulb may involve full thickness of the wall (4% of cases) 27, 28, 29.
The incidence of ectopic pancreas in autopsies ranges 0.5% to 15% in the general population, being more common at the age of 30 to 50 years with a male predominance 5, 30, 21, 11, 31.
Ectopic pancreas is asymptomatic and is usually found incidentally during routine endoscopic or radiographic studies 32. However, ectopic pancreas may become symptomatic when complicated by inflammation, ulceration, bleeding, intussusception, obstruction or malignant transformation 33, 34, 35, 36, 37, 38.
Ectopic pancreas symptoms depending upon the anatomical location, such as gastric outlet obstruction in a pre-pyloric rest or obstructive jaundice in a bile duct focus, may originate from the mass effect of the tumor and are also related to the size of the lesion 39. Lesions greater than 1.5 cm in diameter are more likely to cause symptoms 39. Pain is one of the most common symptoms. The possible explanation is that the pain is due to endrocrine and exocrine function of the ectopic pancreatic tissue, and relates to the secretion of hormones and enzymes, being responsible for inflammation or chemical irritation of the involved tissues 40. Bleeding due to mucosal erosion, ulcer formation and perforation especially localized in the small intestine have also been reported 39.
Rarely malignant transformation into adenocarcinoma occuring in the ectopic pancreatic tissue may be seen 41, 33, 42, 43. In order to be described as arising from ectopic pancreas, the diagnosis of a carcinoma should fulfil three criteria: (1) the tumor must be located within or very close to the ectopic pancreatic tissue, (2) transition between pancreatic structures and carcinoma must be identified and (3) the non-neoplastic pancreatic tissue must comprise fully developed acini and ducts 42. Adenocarcinomas arising from ectopic pancreas seem to have a somewhat better prognosis than those arising from the pancreas itself, probably due to earlier presentation 43.
Generally, it is difficult to make the correct pathological diagnosis from a typical endoscopic mucosal biopsy to distinguish ectopic pancreas from other gastric submucosal diseases 20. At barium studies, ectopic pancreas is usually seen as a smooth, broad-based submucosal lesion in the greater curvature of the gastric antrum or in the proximal duodenum. A diagnostic feature is a central niche or umbilication, representing the orifice of the rudimentary pancreatic duct, containing a small collection of barium, seen in up to 45% of cases 22, 44, 45. Park et al. 46 investigated the morphologic features of 26 patients with ectopic pancreas, and they observed umbilication or central dimpling in 34.6% (9/26 lesions). There are no specific diagnostic features at computed tomography (CT) to differentiate ectopic pancreas from other submucosal masses 32.
Since patients with ectopic pancreas are generally asymptomatic, most ectopic pancreas cases require no treatment 3. However, surgical resection is required in rare instances such as bleeding 47, pancreatitis 48, 49, inflammatory change 50, obstructive jaundice, and malignant transformation 51, 41. The most common malignancy is ductal adenocarcinoma. Other malignant tumors that have been reported are mucinous cystadenocarcinoma, acinar cell carcinoma, islet cell tumor, and solid and papillary neoplasms 28.
Many patients with ectopic pancreas remain completely asymptomatic throughout their lives and without developing any complications 1. Endoscopic treatment has been widely advocated in symptomatic ectopic pancreas 52. For patients with the potential for malignant transformation of ectopic pancreatic tissue (a rare event), surgical resection is the preferred course of treatment 9, 41. In cases with uncertain diagnosis endoscopic cap or band ligation assisted resection or endoscopic submucosal dissection are appropriate and safe treatment procedures, in particular for lesions not involving the muscularis propria 53, 54, 30, 55, 56.
If ectopic pancreatic tissue is found incidentally during a surgical procedure, excision should be considered due to its potential for becoming symptomatic and malignant. If malignancy is suspected extended oncological surgical resection (e.g., pylorus-preserving pancreatoduodenectomies) is justified 57.
The prognosis of patients with adenocarcinoma arising from ectopic pancreatic tissue seems to be better compared to patients with tumors arising from the pancreas itself, probably due to earlier presentation 58, 43.
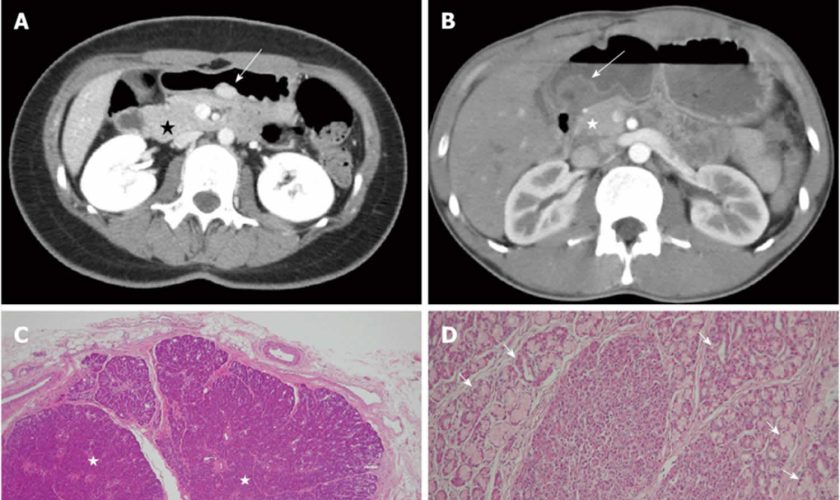
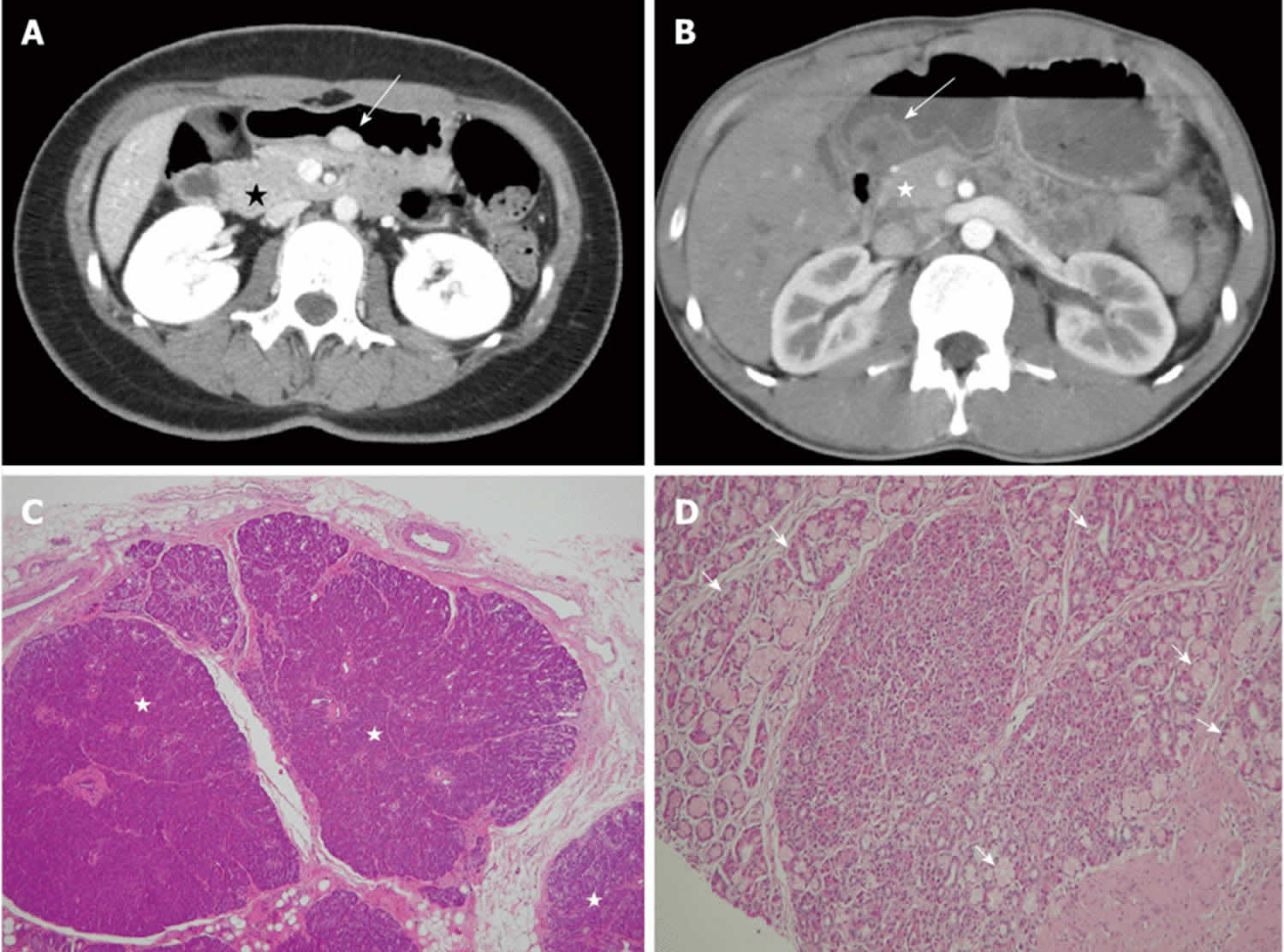
Figure 1. Ectopic pancreas
Footnotes: A 26-year-old woman presented with postprandial epigastric pain for 2 years. (A) Transverse computed tomography (CT) scan showing a small round submucosal lesion with well-defined margins in the wall of the antrum (arrow). Note the contrast material enhancement is higher than that of the normal pancreas (star); (B) Low-power photomicrograph (original magnification, × 20; HE stain) showing that pancreas tissue (star) is predominant in the acinar tissue. A 20-year-old man presented with intermittent epigastralgia for 2 months; (C) Transverse CT scan showing a submucosal round mass (arrow) with necrosis at the gastric antrum. Note the poorly enhancing nodular mass, as compared with the markedly enhancing adjacent normal pancreas (star); (D) Low-power photomicrograph (original magnification, × 200; HE stain) showing ectopic pancreatic tissue, composed primarily of pancreatic ducts (arrow) in the gastric mucosal layer.
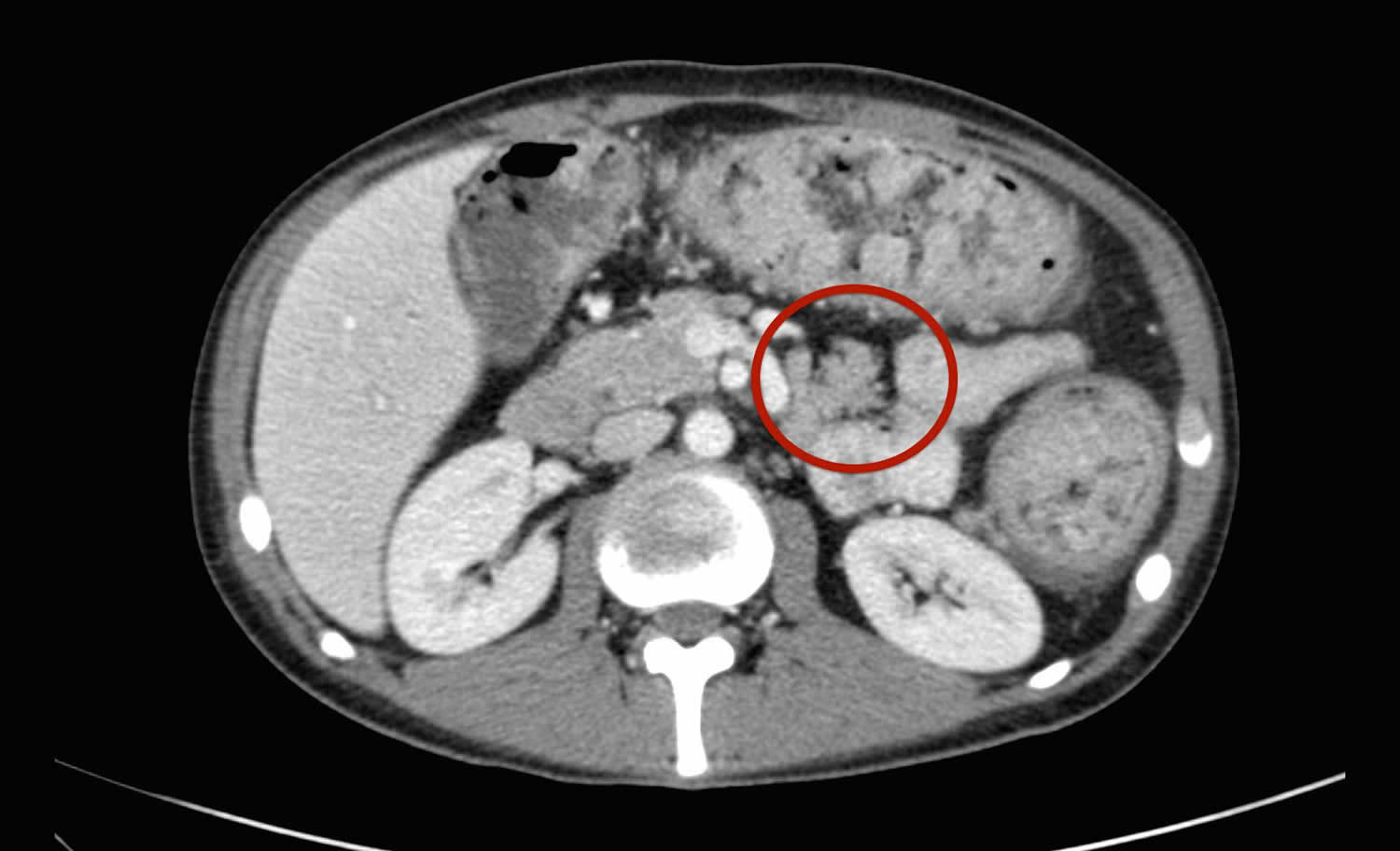
[Source 59 ]Figure 2. Ectopic pancreatic tissue CT
Footnote: Red circle highlights position of ectopic pancreatic tissue on CT.
[Source 60 ]Ectopic pancreas causes
The cause of ectopic pancreas remains unknown. Modern molecular studies have demonstrated that defective Notch signaling pathways and mutations in Neurog3 (neurogenin 3) are responsible for many cases. Ectopic pancreas is a congenital anatomical abnormality that can arise in several locations within the digestive tract and other areas 9. Several theories such as fusion abnormalities, migration abnormalities (misplacement theory), metaplasia theory and replication abnormalities have been proposed to explain the pathogenesis and occurrence of ectopic pancreas.
The most widely held misplacement theory claims that during the period of embryonic rotation of the dorsal and ventral buds deposits of pancreatic tissue migrate from the main body of pancreas and are implanted at various ectopic sites 61, 62, 63, 24.
On the other hand, metaplasia theory suggests that during embryogenesis pancreatic metaplasia of the endodermal tissues localized in the gastric submucosa may occur 11, 12. Currently, the embryological origin of ectopic pancreas remains uncertain. More recent theory is the dislocation theory, which proposes that pancreatic tissue deposits become detached from the primary pancreas and are then relocated to other developing organs, particularly the gastrointestinal system 1.
The most probable theory implicates that during the development of normal pancreas from several evaginations, originating from the wall of the primitive duodenum, one or more evaginations may remain in the bowel wall 10. Migration of this embryonic remnant along with the development of the gastrointestinal tract gives rise to the ectopic pancreatic tissue 11.
Ectopic pancreas most common site
Ectopic pancreas is frequently located in the stomach (up to 38%) of which 95% are located in the greater curvature in the antrum 13, 3, followed by the duodenum (25%–35%) 14, jejunum (16%) 15, Meckel diverticulum, the ileum 16, liver, spleen, bile duct, gall bladder 17, omentum, fallopian tube, mediastinum 18 and more 19, 20.
Generally, the most common ectopic pancreas site is the stomach (up to 38%) of which 95% are found within a distance of 6 cm from the greater curvature of the stomach in the antrum near the pylorus followed by the duodenum (25%–35%) and jejunum (16%) 10, 64, 65, 33, 66. Rarely it can also be found in the ileum, colon, appendix, mesentery, gallbladder or Meckel diverticulum. Ectopic pancreas typically resides within the submucosa or muscularis propria layer 67. The ectopic pancreatic tissue usually measures 0.5 to 2.0 cm in its larger diameter (rarely up to 5 cm) 22.
Ectopic pancreas symptoms
The clinical symptoms of ectopic pancrea depend of location, size and other pathological features that may occasionally coexist, e.g., secretion of pancreatic enzymes that can result in local inflammation or/and secreting the hormones that may exert a whole body effect. In general, ectopic pancreas is asymptomatic and is usually found incidentally on gastroscopy 32. However, ectopic pancreas may become symptomatic when complicated by inflammation, ulceration, bleeding, intussusception, obstruction or malignant transformation 34, 35, 36, 37, 38.
Lesions smaller than 15 mm in diameter remain asymptomatic until they cause local inflammation or obstruction, and are usually detected accidentally. Lesions greater than 15 mm in diameter are more likely to cause symptoms 39.
Ectopic pancreas causing gastric outlet obstruction or obstructive jaundice may originate from the mass effect of the tumor and are also related to the size of the lesion 39.
Pain is one of the most common symptoms. The possible explanation is that the pain is due to endrocrine and exocrine function of the ectopic pancreatic tissue, and relates to the secretion of hormones and enzymes, being responsible for inflammation or chemical irritation of the involved tissues 40. Bleeding due to mucosal erosion, ulcer formation and perforation especially localized in the small intestine have also been reported 39.
Last, but not least, ectopic pancreatic tissue may occasionally turn into adenocarcinoma or a neuroendocrine neoplasm 10, 68, 69.
In children, the clinical picture of ectopic pancreas can be different. Most characteristic are gastrointestional obstructions and intrasuspection that can also be associated with some congenital abnormalities, including granular pancreas, esophageal atresia, Meckel’s diverticulum, malrotation, choledochal cyst and extrahepatic biliary atresia 70.
Ectopic pancreas diagnosis
Ectopic pancreas diagnosis involves determining the location and nature of the condition, typically with the help of medical imaging. However, the findings from imaging tests do not provide particular information, and the diagnosis ultimately depends on a pathological examination. Currently, ectopic pancreas diagnosis can be achieved using biopsy or endoscopic ultrasonography (EUS), which can then be used to direct the appropriate treatment 71. Ectopic pancreas is observed as submucosal masses with intraluminal growth patterns during endoscopic examination 72, 73.
Endoscopic ultrasonography (EUS) examination can visualize the morphological characteristics of ectopic pancreas lesions and pinpoint the location from which the ectopic pancreas originates 74, 75. If necessary, an fine-needle aspiration can be conducted with the use of endoscopic ultrasonography guidance (EUS-FNA) 76, 77. Under ultrasonography gastroscopy, the stomach wall exhibits a distinct five-layer structure 78. Common ultrasonography endoscopic findings of ectopic pancreas include submucosal masses with high echogenicity, low echogenicity, or equiechogenicity. These masses typically originate from the submucosal layer or intrinsic muscle layer 79, 80. The occurrence in particular layers of the stomach wall is as follows: 73% in the submucosal layer, 17% in the muscularis propria layer and 10% in the subserosal layer, sometimes involving through all of these layers 16.
Certain lesions exhibit a combination of echogenicity, which could be indicative of degeneration or the development of cysts 81.
The diagnosis may be sometimes difficult intraoperatively due to the gross similarity of pancreatic heterotopia with gastrointestinal stromal tumour (GIST), gastrointestinal autonomic nerve tumour (GANT), carcinoid, lymphoma or even gastric carcinoma. If in doubt, frozen section is very helpful to establish the diagnosis intraoperatively and to avoid unnecessary extensive operations.
Computed tomography findings are usually non specific. However, multi-slice spiral CT with oral and portovenous phase IV contrast may demonstrate the lesion which enhances similarly with the normal pancreatic tissue. CT can localize lesions with normal pancreatic tissue but cannot distinguish ectopic pancreas from other submucosal tumors 82, 83.
Ectopic pancreas histological type
There are 4 subtypes of ectopic pancreas based on histological examination 23, 24, 25, 26:
- Type 1 ectopic pancreas consists of typical pancreatic tissue with acini, ducts, and islet cells similar to those seen in normal pancreas
- Type 2 ectopic pancreas is composed of pancreatic ducts only, referred as canalicular variety
- Type 3 ectopic pancreas is characterized by glandular acinar tissue only (exocrine pancreas)
- Type 4 ectopic pancreas is made up of endocrine islet cells only (endocrine pancreas)
Ectopic pancreas differential diagnoses
Ectopic pancreas differential diagnoses include gastrointestinal stromal tumors, gastrointestinal autonomic nerve tumors, gastric carcinoids, lymphomas and gastric carcinomas 34, 84, 85.
Ectopic pancreas treatment
Since patients with ectopic pancreas are generally asymptomatic, most ectopic pancreas cases require no treatment 3. However, surgical resection is required in rare instances such as bleeding 47, pancreatitis 48, 49, inflammatory change 50, pseudocyst formation, cyst formation, insulinoma, adenoma, obstructive jaundice, and malignant transformation 41, 33, 51, 86, 87, 43.
With the rapid advancement of endoscopic technology in recent years, there has been widespread promotion of endoscopic treatment 88. Nevertheless, the selection of therapeutic approaches must be dependent on the lesion’s location, size, and its interaction with neighboring organs 89. The existing techniques employed for the endoscopic treatment of ectopic pancreas encompass endoscopic submucosal dissection, submucosaltunnel endoscopic resection (STER), endoscopic high-frequency resection, mucosal resection, and endoscopic full-thickness resection 30, 90, 91. Certain lesions can be eliminated with ligation 92. If the ectopic pancreas arises from the muscular layer or grows through the entire wall of the organ, or if the growth is located outside the reach of endoscopy, it is indicated to have local surgical excision for treatment 93. If there is a possibility of cancer, it is advisable to undertake aggressive surgery for resection 94. Certain individuals with malignant transformation may necessitate adjuvant chemotherapy following surgical intervention. Zheng et al 95 reported that laparoscopic resection is better for large gastric ectopic pancreas with a deep origin, which has added new clue for the surgical treatment in the field of ectopic pancreas. Meanwhile, multicenter large-scale studies are needed to describe its characteristics and evaluate the safety due to the rarity of gastric ectopic pancreas 1.
- Li CF, Li QR, Bai M, Lv YS, Jiao Y. Overview of ectopic pancreas. World J Gastrointest Surg. 2024 Feb 27;16(2):284-288. doi: 10.4240/wjgs.v16.i2.284[↩][↩][↩][↩][↩]
- Rezvani M, Menias C, Sandrasegaran K, Olpin JD, Elsayes KM, Shaaban AM. Heterotopic pancreas: histopathologic features, imaging findings, and complications. Radiographics. (2017) 37:484–99. 10.1148/rg.2017160091[↩]
- Iwamuro M, Tsuzuki T, Ohya S, Okada H, Tanaka T, Hori K, Kita M, Kawano S, Kawahara Y, Yamamoto K. Ectopic pancreas in the stomach successfully resected by endoscopic submucosal dissection. Case Rep Med. 2015;2015:147927. doi: 10.1155/2015/147927[↩][↩][↩][↩][↩]
- Borghei P, Sokhandon F, Shirkhoda A, Morgan DE. Anomalies, anatomic variants, and sources of diagnostic pitfalls in pancreatic imaging. Radiology. 2013 Jan;266(1):28-36. doi: 10.1148/radiol.12112469[↩]
- Mulholland KC, Wallace WD, Epanomeritakis E, Hall SR. Pseudocyst formation in gastric ectopic pancreas. JOP. 2004 Nov 10;5(6):498-501.[↩][↩]
- Breunig M, Merkle J, Wagner M, Melzer MK, et al. Modeling plasticity and dysplasia of pancreatic ductal organoids derived from human pluripotent stem cells. Cell Stem Cell. 2021 Jun 3;28(6):1105-1124.e19. doi: 10.1016/j.stem.2021.03.005[↩]
- Ehrhardt JD, Gomez F. Embryology, Pancreas. [Updated 2023 Aug 14]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books[↩]
- Zhang L, Sanderson SO, Lloyd RV, Smyrk TC. Pancreatic intraepithelial neoplasia in heterotopic pancreas: evidence for the progression model of pancreatic ductal adenocarcinoma. Am J Surg Pathol. 2007 Aug;31(8):1191-5. doi: 10.1097/PAS.0b013e31806841e1[↩]
- Mundackal N, Arslan ME, Decker C, Lee H, Nigam A. The removal of ectopic pancreas to prevent carcinoma development. Am J Surg. 2021 Dec;222(6):1196-1197. doi: 10.1016/j.amjsurg.2021.07.002[↩][↩][↩]
- Christodoulidis G, Zacharoulis D, Barbanis S, Katsogridakis E, Hatzitheofilou K. Heterotopic pancreas in the stomach: a case report and literature review. World J Gastroenterol. 2007 Dec 7;13(45):6098-100. doi: 10.3748/wjg.v13.45.6098[↩][↩][↩][↩]
- Chandan VS, Wang W. Pancreatic heterotopia in the gastric antrum. Arch Pathol Lab Med. 2004 Jan;128(1):111-2. doi: 10.5858/2004-128-111-PHITGA[↩][↩][↩][↩][↩]
- Skandalakis LJ, Rowe JS Jr, Gray SW, Skandalakis JE. Surgical embryology and anatomy of the pancreas. Surg Clin North Am. 1993 Aug;73(4):661-97. doi: 10.1016/s0039-6109(16)46080-9[↩][↩]
- Seneviratne SA, Ramanayaka IT, Samarasekera DN. Heterotopic pancreas in the body of the stomach. Ceylon Med J. 2009 Jun;54(2):57-8. doi: 10.4038/cmj.v54i2.869[↩][↩]
- Ginori A, Vassallo L, Butorano MA, Bettarini F, Di Mare G, Marrelli D. Pancreatic adenocarcinoma in duodenal ectopic pancreas: a case report and review of the literature. Pathologica. 2013 Apr;105(2):56-8.[↩][↩]
- Serrano JS, Stauffer JA. Ectopic Pancreas in the Wall of the Small Intestine. J Gastrointest Surg. 2016 Jul;20(7):1407-8. doi: 10.1007/s11605-016-3104-4[↩][↩]
- Christodoulidis G, Zacharoulis D, Barbanis S, Katsogridakis E, Hatzitheofilou K. Heterotopic pancreas in the stomach: a case report and literature review. World J Gastroenterol. 2007;13(45):6098–6100. doi: 10.3748/wjg.v13.45.6098[↩][↩][↩][↩]
- Sanchiz Cárdenas EM, Soler Humanes R, Lavado Fernández AI, Díaz Nieto R, Suárez Muñoz MA. Ectopic pancreas in gallbladder. Clinical significance, diagnostic and therapeutic implications. Rev Esp Enferm Dig. 2015 Nov;107(11):701-3. doi: 10.17235/reed.2015.3627/2014[↩][↩]
- Wu L, Lin L, Jiang L, Jiang G. Ectopic Pancreas in Mediastinum Presenting as Hemoptysis. Ann Thorac Surg. 2015 Dec;100(6):2368. doi: 10.1016/j.athoracsur.2015.08.064[↩][↩]
- Matsumoto T, Tanaka N, Nagai M, Koike D, Sakuraoka Y, Kubota K. A case of gastric heterotopic pancreatitis resected by laparoscopic surgery. Int Surg. 2015 Apr;100(4):678-82. doi: 10.9738/INTSURG-D-14-00182.1[↩][↩]
- Liu X, Wu X, Tuo B, Wu H. Ectopic pancreas appearing as a giant gastric cyst mimicking gastric lymphangioma: a case report and a brief review. BMC Gastroenterol. 2021 Apr 6;21(1):151. doi: 10.1186/s12876-021-01686-9[↩][↩][↩][↩]
- DeBord JR, Majarakis JD, Nyhus LM. An unusual case of heterotopic pancreas of the stomach. Am J Surg. 1981 Feb;141(2):269-73. doi: 10.1016/0002-9610(81)90172-0[↩][↩]
- Yu J, Turner MA, Fulcher AS, Halvorsen RA. Congenital anomalies and normal variants of the pancreaticobiliary tract and the pancreas in adults: part 2, Pancreatic duct and pancreas. AJR Am J Roentgenol. 2006 Dec;187(6):1544-53. doi: 10.2214/AJR.05.0774[↩][↩][↩]
- Akbari AH, Putra J. Type 1 Choledochal Cyst with Ectopic Pancreas and Septate Gallbladder. Fetal Pediatr Pathol. 2022 Apr;41(2):334-337. doi: 10.1080/15513815.2020.1797962[↩][↩]
- Trifan A, Târcoveanu E, Danciu M, Huţanaşu C, Cojocariu C, Stanciu C. Gastric heterotopic pancreas: an unusual case and review of the literature. J Gastrointestin Liver Dis. 2012 Jun;21(2):209-12.[↩][↩][↩]
- Matsuki M, Gouda Y, Ando T, Matsuoka H, Morita T, Uchida N, et al. Adenocarcinoma arising from aberrant pancreas in the stomach. J Gastroenterol. 2005;40(6):652–656. doi: 10.1007/s00535-004-1601-9[↩][↩]
- Gaspar Fuentes A, Campos Tarrech JM, Fernández Burgui JL, Castells Tejón E, Ruíz Rossello J, Gómez Pérez J, Armengol Miró J. Ectopias pancreáticas [Pancreatic ectopias]. Rev Esp Enferm Apar Dig. 1973 Feb 1;39(3):255-68. Spanish.[↩][↩]
- Alqahtani A, Aljohani E, Almadi F, Billa S, Alqahtani M, Alkhaldi H. Heterotopic pancreatic tissue in the gastric antrum an incidental finding during bariatric surgery: A case report and literature review. Int J Surg Case Rep. 2020;67:39-41. doi: 10.1016/j.ijscr.2019.12.040[↩]
- Rezvani M, Menias C, Sandrasegaran K, Olpin JD, Elsayes KM, Shaaban AM. Heterotopic pancreas: histopathologic features, imaging findings, and complications. RadioGraphics. 2017;37(2):484–499. doi: 10.1148/rg.2017160091[↩][↩]
- Wang QY, Yang XY, Zhang Z. A stubborn anemia caused by ectopic pancreas bleeding in the jejunum revealed by capsule endoscopy. Quant Imaging Med Surg. 2015 Oct;5(5):783-6. doi: 10.3978/j.issn.2223-4292.2015.01.09[↩]
- Ryu DY, Kim GH, Park DY, Lee BE, Cheong JH, Kim DU, Woo HY, Heo J, Song GA. Endoscopic removal of gastric ectopic pancreas: an initial experience with endoscopic submucosal dissection. World J Gastroenterol. 2010 Sep 28;16(36):4589-93. doi: 10.3748/wjg.v16.i36.4589[↩][↩][↩]
- Jiang LX, Xu J, Wang XW, Zhou FR, Gao W, Yu GH, Lv ZC, Zheng HT. Gastric outlet obstruction caused by heterotopic pancreas: A case report and a quick review. World J Gastroenterol. 2008 Nov 21;14(43):6757-9. doi: 10.3748/wjg.14.6757[↩]
- Türkvatan A, Erden A, Türkoğlu MA, Yener Ö. Congenital variants and anomalies of the pancreas and pancreatic duct: imaging by magnetic resonance cholangiopancreaticography and multidetector computed tomography. Korean J Radiol. 2013 Nov-Dec;14(6):905-13. doi: 10.3348/kjr.2013.14.6.905[↩][↩][↩]
- Cazacu IM, Luzuriaga Chavez AA, Nogueras Gonzalez GM, Saftoiu A, Bhutani MS. Malignant Transformation of Ectopic Pancreas. Dig Dis Sci. 2019 Mar;64(3):655-668. doi: 10.1007/s10620-018-5366-z[↩][↩][↩][↩]
- Kim JY, Lee JM, Kim KW, Park HS, Choi JY, Kim SH, et al. Ectopic pancreas: CT findings with emphasis on differentiation from small gastrointestinal stromal tumor and leiomyoma. Radiology. 2009;252(1):92–100. doi: 10.1148/radiol.2521081441[↩][↩][↩]
- Cazacu IM, Luzuriaga Chavez AA, Nogueras Gonzalez GM, Saftoiu A, Bhutani MS. Malignant transformation of ectopic pancreas. Dig Dis Sci. 2019;64(3):655–668. doi: 10.1007/s10620-018-5366-z[↩][↩]
- Emerson L, Layfield LJ, Rohr LR, Dayton MT. Adenocarcinoma arising in association with gastric heterotopic pancreas: a case report and review of the literature. J Surg Oncol. 2004;87(1):53–57. doi: 10.1002/jso.20087[↩][↩]
- Papaziogas B, Koutelidakis I, Tsiaousis P, Panagiotopoulou K, Paraskevas G, Argiriadou H, et al. Carcinoma developing in ectopic pancreatic tissue in the stomach: a case report. Cases J. 2008;1(1):249. doi: 10.1186/1757-1626-1-249[↩][↩]
- Kaneda M, Yano T, Yamamoto T, Suzuki T, Fujimori K, Itoh H, Mizumoto R. Ectopic pancreas in the stomach presenting as an inflammatory abdominal mass. Am J Gastroenterol. 1989 Jun;84(6):663-6.[↩][↩]
- Armstrong CP, King PM, Dixon JM, Macleod IB. The clinical significance of heterotopic pancreas in the gastrointestinal tract. Br J Surg. 1981 Jun;68(6):384-7. doi: 10.1002/bjs.180068060[↩][↩][↩][↩][↩][↩]
- Ormarsson OT, Gudmundsdottir I, Mårvik R. Diagnosis and treatment of gastric heterotopic pancreas. World J Surg. 2006 Sep;30(9):1682-9. doi: 10.1007/s00268-005-0669-6[↩][↩]
- Betzler A, Mees ST, Pump J, Schölch S, Zimmermann C, Aust DE, et al. Clinical impact of duodenal pancreatic heterotopia – is there a need for surgical treatment? BMC Surg. 2017;17:53. doi: 10.1186/s12893-017-0250-x[↩][↩][↩][↩]
- Emerson L, Layfield LJ, Rohr LR, Dayton MT. Adenocarcinoma arising in association with gastric heterotopic pancreas: A case report and review of the literature. J Surg Oncol. 2004 Jul 15;87(1):53-7. doi: 10.1002/jso.20087[↩][↩]
- Eisenberger CF, Gocht A, Knoefel WT, Busch CB, Peiper M, Kutup A, Yekebas EF, Hosch SB, Lambrecht W, Izbicki JR. Heterotopic pancreas–clinical presentation and pathology with review of the literature. Hepatogastroenterology. 2004 May-Jun;51(57):854-8.[↩][↩][↩][↩]
- Alexander LF. Congenital pancreatic anomalies, variants, and conditions. Radiol Clin North Am. 2012 May;50(3):487-98. doi: 10.1016/j.rcl.2012.03.006[↩]
- Sabiston Textbook of Surgery: The Biological Basis of Modern Surgical Practice. 15th ed. In: Sabiston DC, editor. Philadelphia: WB Saunders Company; 1997. pp. 872–873.[↩]
- Park S. H., Kim G. H., Park D. Y., et al. Endosonographic findings of gastric ectopic pancreas: a single center experience. Journal of Gastroenterology and Hepatology. 2011;26(9):1441–1446. doi: 10.1111/j.1440-1746.2011.06764.x[↩]
- Teke Z., Kabay B., Kelten C., Yilmaz M., Duzcan E. Ectopic pancreas of the gastric antrum contiguous to a gastrointestinal stromal tumor manifesting as upper gastrointestinal bleeding: report of a case. Surgery Today. 2007;37(1):74–77. doi: 10.1007/s00595-006-3340-4[↩][↩]
- Hirasaki S., Tanimizu M., Moriwaki T., Nasu J. Acute pancreatitis occurring in gastric aberrant pancreas treated with surgery and proved by histological examination. Internal Medicine. 2005;44(11):1169–1173. doi: 10.2169/internalmedicine.44.1169[↩][↩]
- Fujii M., Kawamoto H., Nagahara T., et al. A case of gastric aberrant pancreas diagnosed with EUS-guided FNA. Gastrointestinal Endoscopy. 2011;74(2):407–408. doi: 10.1016/j.gie.2011.03.1124[↩][↩]
- Kishiki T., Abe N., Sugiyama M., et al. Inflammatory mass formation caused by gastric ectopic pancreas: report of a case. Surgery Today. 2013;43(12):1448–1451. doi: 10.1007/s00595-012-0317-3[↩][↩]
- Matsuki M., Gouda Y., Ando T., et al. Adenocarcinoma arising from aberrant pancreas in the stomach. Journal of Gastroenterology. 2005;40(6):652–656. doi: 10.1007/s00535-004-1601-9[↩][↩]
- Ormarsson OT, Gudmundsdottir I, Marvik R. Diagnosis and treatment of gastric heterotopic pancreas. World J Surg. 2006;30(9):1682–1689. doi: 10.1007/s00268-005-0669-6[↩]
- He G, Wang J, Chen B, Xing X, Wang J, Chen J, He Y, Cui Y, Chen M. Feasibility of endoscopic submucosal dissection for upper gastrointestinal submucosal tumors treatment and value of endoscopic ultrasonography in pre-operation assess and post-operation follow-up: a prospective study of 224 cases in a single medical center. Surg Endosc. 2016 Oct;30(10):4206-13. doi: 10.1007/s00464-015-4729-1[↩]
- Zhang Y, Huang Q, Zhu LH, Zhou XB, Ye LP, Mao XL. Endoscopic excavation for gastric heterotopic pancreas: an analysis of 42 cases from a tertiary center. Wien Klin Wochenschr. 2014 Sep;126(17-18):509-14. doi: 10.1007/s00508-014-0563-z[↩]
- Bain AJ, Owens DJ, Tang RS, Peterson MR, Savides TJ. Pancreatic rest resection using band ligation snare polypectomy. Dig Dis Sci. 2011 Jun;56(6):1884-8. doi: 10.1007/s10620-011-1669-z[↩]
- Liu X, Wang G, Ge N, Wang S, Guo J, Liu W, Sun S. Endoscopic removal of symptomatic gastric heterotopic pancreas: a report of nine cases. Surg Innov. 2013 Dec;20(6):NP40-6. doi: 10.1177/1553350613499453[↩]
- Betzler A, Mees ST, Pump J, Schölch S, Zimmermann C, Aust DE, Weitz J, Welsch T, Distler M. Clinical impact of duodenal pancreatic heterotopia – Is there a need for surgical treatment? BMC Surg. 2017 May 8;17(1):53. doi: 10.1186/s12893-017-0250-x[↩]
- Okamoto H, Kawaoi A, Ogawara T, et al. Invasive ductal carcinoma arising from an ectopic pancreas in the gastric wall: a long-term survival case. Case Rep Oncol. 2012;5:69–73. doi: 10.1159/000335870[↩]
- Lin YM, Chiu NC, Li AF, Liu CA, Chou YH, Chiou YY. Unusual gastric tumors and tumor-like lesions: Radiological with pathological correlation and literature review. World J Gastroenterol. 2017 Apr 14;23(14):2493-2504. https://www.wjgnet.com/1007-9327/full/v23/i14/2493.htm[↩]
- Ectopic pancreatic tissue. https://radiopaedia.org/cases/ectopic-pancreatic-tissue[↩]
- Chen H-L, Chang W-H, Shih S-C, Bair M-J, Lin S-C. Changing pattern of ectopic pancreas: 22 years of experience in a medical center. J Formos Med Assoc. 2008;107(12):932–936. doi: 10.1016/S0929-6646(09)60016-4[↩]
- Chen H-L, Lin S-C, Chang W-H, Yang T-L, Chen Y-J. Identification of ectopic pancreas in the ileum by capsule endoscopy. J Formos Med Assoc. 2007;106(3):240–243. doi: 10.1016/S0929-6646(09)60246-1[↩]
- Monier A, Awad A, Szmigielski W, Muneer M, Alrashid A, Darweesh A, et al. Heterotopic pancreas: a rare cause of Ileo-Ileal intussusception. Pol J Radiol. 2014;79:349–351. doi: 10.12659/PJR.890913[↩]
- Filip R, Walczak E, Huk J, Radzki RP, Bieńko M. Heterotopic pancreatic tissue in the gastric cardia: a case report and literature review. World J Gastroenterol. 2014 Nov 28;20(44):16779-81. doi: 10.3748/wjg.v20.i44.16779[↩]
- Kim JY, Lee JM, Kim KW, Park HS, Choi JY, Kim SH, Kim MA, Lee JY, Han JK, Choi BI. Ectopic pancreas: CT findings with emphasis on differentiation from small gastrointestinal stromal tumor and leiomyoma. Radiology. 2009 Jul;252(1):92-100. doi: 10.1148/radiol[↩]
- Qian L, Yang J, Zhang J. Gastric ectopic pancreas adenocarcinoma: A case report and literature review. Saudi Med J. 2023 Nov;44(11):1174-1179. doi: 10.15537/smj.2023.44.11.20220914[↩]
- Ourô S, Taré F, Moniz L. Ectopia pancreática [Pancreatic ectopia]. Acta Med Port. 2011 Mar-Apr;24(2):361-6. Portuguese.[↩]
- Agale SV, Agale VG, Zode RR, Grover S, Joshi S. Heterotopic pancreas involving stomach and duodenum. J Assoc Physicians India. 2009 Sep;57:653-4.[↩]
- Gurocak B, Gokturk HS, Kayacetin S, Bakdik S. A rare case of heterotopic pancreas in the stomach which caused closed perforation. Neth J Med. 2009 Jul-Aug;67(7):285-7.[↩]
- Ogata H, Oshio T, Ishibashi H, Takano S, Yagi M. Heterotopic pancreas in children: review of the literature and report of 12 cases. Pediatr Surg Int. 2008 Mar;24(3):271-5. doi: 10.1007/s00383-007-2098-0[↩]
- Matsushita M, Hajiro K, Okazaki K, Takakuwa H. Gastric aberrant pancreas: EUS analysis in comparison with the histology. Gastrointest Endosc. 1999 Apr;49(4 Pt 1):493-7. doi: 10.1016/s0016-5107(99)70049-0[↩]
- Wei R, Wang QB, Chen QH, Liu JS, Zhang B. Upper gastrointestinal tract heterotopic pancreas: findings from CT and endoscopic imaging with histopathologic correlation. Clin Imaging. 2011 Sep-Oct;35(5):353-9. doi: 10.1016/j.clinimag.2010.10.001[↩]
- Gottschalk U, Dietrich CF, Jenssen C. Ectopic pancreas in the upper gastrointestinal tract: Is endosonographic diagnosis reliable? Data from the German Endoscopic Ultrasound Registry and review of the literature. Endosc Ultrasound. 2018 Jul-Aug;7(4):270-278. doi: 10.4103/eus.eus_18_17[↩]
- Flores A, Papafragkakis C, Uberoi AS, Thaiudom S, Bhutani MS. EUS of an atypical ectopic pancreas. Endosc Ultrasound. 2018 May-Jun;7(3):216-217. doi: 10.4103/eus.eus_111_17[↩]
- Endo S, Saito R, Ochi D, Yamada T, Hirose M, Hiroshima Y, Yamamoto Y, Ueno T, Hasegawa N, Moriwaki T, Narasaka T, Kaneko T, Fukuda K, Suzuki H, Mizokami Y, Hyodo I. Effectiveness of an endoscopic biopsy procedure using EUS-FNA and EMR-C for diagnosing adenocarcinoma arising from ectopic pancreas: two case reports and a literature review. Intern Med. 2014;53(10):1055-62. doi: 10.2169/internalmedicine.53.1420[↩]
- Kida M, Kawaguchi Y, Miyata E, Hasegawa R, Kaneko T, Yamauchi H, Koizumi S, Okuwaki K, Miyazawa S, Iwai T, Kikuchi H, Watanabe M, Imaizumi H, Koizumi W. Endoscopic ultrasonography diagnosis of subepithelial lesions. Dig Endosc. 2017 May;29(4):431-443. doi: 10.1111/den.12854[↩]
- Attwell A, Sams S, Fukami N. Diagnosis of ectopic pancreas by endoscopic ultrasound with fine-needle aspiration. World J Gastroenterol. 2015 Feb 28;21(8):2367-73. doi: 10.3748/wjg.v21.i8.2367[↩]
- Pellicano R, Bruno M, Fagoonee S, Ribaldone DG, Fasulo R, De Angelis C. Endoscopic ultrasound in the preoperative staging of gastric cancer: key messages for surgeons. Minerva Chir. 2015 Dec;70(6):417-27.[↩]
- Park SH, Kim GH, Park DY, Shin NR, Cheong JH, Moon JY, Lee BE, Song GA, Seo HI, Jeon TY. Endosonographic findings of gastric ectopic pancreas: a single center experience. J Gastroenterol Hepatol. 2011 Sep;26(9):1441-6. doi: 10.1111/j.1440-1746.2011.06764.x[↩]
- Zinkiewicz K, Juśkiewicz W, Zgodziński W, Szumiło J, Cwik G, Furtak J, Maciejewski R, Wallner G. Ectopic pancreas: endoscopic, ultrasound and radiological features. Folia Morphol (Warsz). 2003;62(3):205-9.[↩]
- Shanbhogue AK, Fasih N, Surabhi VR, Doherty GP, Shanbhogue DK, Sethi SK. A clinical and radiologic review of uncommon types and causes of pancreatitis. Radiographics. 2009 Jul-Aug;29(4):1003-26. doi: 10.1148/rg.294085748[↩]
- Rodriguez FJ, Abraham SC, Allen MS, Sebo TJ. Fine-needle aspiration cytology findings from a case of pancreatic heterotopia at the gastroesophageal junction. Diagn Cytopathol. 2004 Sep;31(3):175-9. doi: 10.1002/dc.20066[↩]
- Cho JS, Shin KS, Kwon ST, Kim JW, Song CJ, Noh SM, Kang DY, Kim HY, Kang HK. Heterotopic pancreas in the stomach: CT findings. Radiology. 2000 Oct;217(1):139-44. doi: 10.1148/radiology.217.1.r00oc09139[↩]
- Diaconu C, Ciocirlan M, Jinga M, Costache RS, Constantinescu G, Ilie M, et al. Ectopic pancreas mimicking gastrointestinal stromal tumor in the stomach fundus. Endoscopy. 2018;50(7):E186–e187. doi: 10.1055/a-0605-2996[↩]
- Subasinghe D, Sivaganesh S, Perera N, Samarasekera DN. Gastric fundal heterotopic pancreas mimicking a gastrointestinal stromal tumour (GIST): a case report and a brief review. BMC Res Notes. 2016;9:185. doi: 10.1186/s13104-016-1995-5[↩]
- Claudon M, Verain AL, Bigard MA, Boissel P, Poisson P, Floquet J, Regent D. Cyst formation in gastric heterotopic pancreas: report of two cases. Radiology. 1988 Dec;169(3):659-60. doi: 10.1148/radiology[↩]
- Ura H, Denno R, Hirata K, Saeki A, Hirata K, Natori H. Carcinoma arising from ectopic pancreas in the stomach: endosonographic detection of malignant change. J Clin Ultrasound. 1998 Jun;26(5):265-8. doi: 10.1002/(sici)1097-0096(199806)26:5<265::aid-jcu7>3.0.co;2-a[↩]
- Hawes RH. Endoscopic innovations. Gastrointest Endosc. 2013 Sep;78(3):410-3. doi: 10.1016/j.gie.2013.07.005[↩]
- Zhou Y, Zhou S, Shi Y, Zheng S, Liu B. Endoscopic submucosal dissection for gastric ectopic pancreas: a single-center experience. World J Surg Oncol. 2019 Apr 16;17(1):69. doi: 10.1186/s12957-019-1612-x[↩]
- Vitiello GA, Cavnar MJ, Hajdu C, Khaykis I, Newman E, Melis M, Pachter HL, Cohen SM. Minimally Invasive Management of Ectopic Pancreas. J Laparoendosc Adv Surg Tech A. 2017 Mar;27(3):277-282. doi: 10.1089/lap.2016.0562[↩]
- Matsushita M, Hachimine D, Nishio A, Seki T, Okazaki K. Endoscopic and/or laparoscopic full-layer resection of gastric ectopic pancreas arising from submucosal and muscular layers. Gastrointest Endosc. 2016 Sep;84(3):547. doi: 10.1016/j.gie.2016.02.035[↩]
- Wu Y, Xu W, Chen C, Chai H, Sun C, Liu J. Ligation-assisted endoscopic mucosal resection for duodenal neuroendocrine tumor. Rev Esp Enferm Dig. 2023 Oct 26. doi: 10.17235/reed.2023.9985/2023[↩]
- Paolucci P, Brasesco OE, Rosin D, Saber AA, Avital S, Berho M, Rosenthal RJ. Laparoscopic resection of ectopic pancreas in the gastric antrum: case report and literature review. J Laparoendosc Adv Surg Tech A. 2002 Apr;12(2):139-41. doi: 10.1089/10926420252939691[↩]
- Chang TK, Huang CW, Ma CJ, Su WC, Tsai HL, Wang JY. Ectopic pancreas mimicking gastric submucosal tumour treated using robotic surgery. J Minim Access Surg. 2020 Apr-Jun;16(2):179-181. doi: 10.4103/jmas.JMAS_1_19[↩]
- Zheng HD, Huang QY, Hu YH, Ye K, Xu JH. Laparoscopic resection and endoscopic submucosal dissection for treating gastric ectopic pancreas. World J Gastrointest Surg. 2023 Dec 27;15(12):2799-2808. doi: 10.4240/wjgs.v15.i12.2799[↩]